Abstract
Purpose
This study aims to characterize complications, metabolic improvement, and change in ambulation status for patients with impaired mobility undergoing bariatric surgery.
Material and Methods
Individuals undergoing primary sleeve gastrectomy (SG) or Roux-en-Y gastric bypass (RYGB) from February 2008 to December 2015 were included. Impaired mobility (WC) was defined as using a wheelchair or motorized scooter for at least part of a typical day. The WC group was propensity score matched to ambulatory patients (1:5 ratio). Comparisons were made for 30-day morbidity and mortality and 1-year improvement in weight-related comorbidities.
Results
There were 93 patients in the WC group matched to 465 ambulatory controls. The median operative time (180 vs 159 min, p = 0.003) and postoperative length of stay (4 vs 3 days, p ≤ 0.001) was higher in the WC group. There were no differences in readmission or all-cause morbidity within 30 days. The median percent excess weight loss (%EWL) at 1 year was similar (WC group, 65% available, 53% EWL vs AMB group, 73% available, 54% EWL); however, patients with impaired mobility were less likely to experience improvement in diabetes (76 vs 90%, p = 0.046), hypertension (63 vs 82%, p < 0.005), and obstructive sleep apnea (53 vs 71%, p < 0.001). Within the WC group, 62% had improvement in their mobility status, eliminating dependence on wheelchair or scooter assistance.
Conclusion
Patients with both obesity and impaired mobility experience similar rates of perioperative morbidity and weight loss at 1 year compared to ambulatory controls. However, improvement in weight-related comorbidities may be less likely with impaired mobility.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Morbid obesity contributes to the development of arthritis of weight bearing joints with subsequent impairment of physical activity and walking limitations [1, 2]. Obese patients are more likely to develop both osteoarthritis and arthralgia resulting in difficulty walking long distances compared to non-obese controls [3]. Possible mechanisms involved in the development of obesity-associated arthritis include both increased joint loading as well as a systemic low-grade pro-inflammatory state mediated by altered lipid metabolism and an increased in macrophages present in obese individuals [4,5,6,7]. Further confounding impaired mobility in obese patients is the fact that obese patients are at an increased risk for weight-related comorbidities, including impaired cardiorespiratory function, which may physically restrict their ability to participate in weight-reducing activities. Concomitant emotional disorders, such as depression which can negatively impact motivation to lose weight, also contribute to impairment of physical activity [8,9,10,11].
Bariatric surgery has proven durable and effective for sustained weight loss and resolution of weight-related comorbidities [12]. Furthermore, bariatric surgery has been shown to improve the physical functioning of patients postoperatively. For example, a recent multi-institutional observational cohort study of 2458 participants showed improvement in physical function, pain, and walk time compared to baseline over a 3-year follow-up period after bariatric surgery [13]. Furthermore, significant literature exists suggesting symptomatic and clinical improvement of osteoarthritis in patients undergoing bariatric surgery [14, 15]. Nevertheless, these studies have primarily included patients who were fully ambulatory prior to bariatric surgery.
It is currently unknown whether the benefits of bariatric surgery outweigh the risks in patients with impaired mobility. It has been postulated that higher perioperative complications may exist and could include a higher risk of venous thromboembolism and suboptimal weight loss due to impaired ambulation [16]. On the other hand, however, several small case series exist that demonstrate an improvement in ambulation and quality of life for wheelchair-bound patients following bariatric surgery [16,17,18,19,20,21]. Of the few studies performed, wheelchair-bound patients have not been compared to those without limitations to mobility. Therefore, the purpose of this study is to evaluate and compare the perioperative and short-term (1 year) outcomes following bariatric surgery for patients with impaired mobility preoperatively to an ambulatory cohort. The primary endpoint was defined as the improvement in the ambulation and metabolic outcomes at 1 year after bariatric surgery in patients with impaired mobility. Secondary endpoints included comparison of the postoperative length of stay, short-term surgical outcomes, and improvement in weight-related comorbidities at 1 year among patients with impaired mobility versus the ambulatory cohort.
Materials and Methods
Study Cohort
After approval by our Institutional Review Board, a retrospective review of a prospectively maintained database was completed to identify patients who underwent primary bariatric surgery at our institution from February 2008 to December 2015. Patients included in this study underwent either a Roux-en-Y gastric bypass (RYGB) or a sleeve gastrectomy (SG). Patients who underwent adjustable gastric banding (AGB) procedures and patients who underwent revisional bariatric surgery were excluded.
Definitions
Patients with impaired mobility were defined as using a wheelchair (WC group) or motorized scooter for at least part of a typical day. Patients with impaired mobility were further subclassified as those totally dependent on wheelchair (WC-D) and those using both wheelchair and an assist device for part of a typical day (WC-A). For the WC-D group, improvement in ambulation was defined as eliminating the use of a wheelchair or scooter either to walking unaided or with an assist device. For the WC-A group, improvement in ambulation status was defined as walking unaided or with assist device alone, not requiring a wheelchair. Patients without improvement were classified as unchanged. Ambulatory patients (AMB group) were defined as patients who did not require assistance with walking, either with a wheelchair, motorized scooter, or with an assist device. The early postoperative surgical outcomes (≤ 30 days) and metabolic outcomes at 1 year after bariatric surgery were defined as per the American Society for Metabolic and Bariatric Surgery (ASMBS) reporting standards [22].
Study Endpoints and Data Collection
Study data was retrospectively collected and managed using REDCap (Research Electronic Data Capture) hosted at our institution and customized for this study [23]. Collected data included baseline patient demographics: age at the time of procedure; gender; preoperative body mass index (BMI); comorbidities including diabetes mellitus, hypertension, dyslipidemia, sleep apnea, gastroesophageal reflux disease (GERD), venous thromboembolism (VTE), chronic obstructive pulmonary disease (COPD), myocardial infarction, heart failure, chronic kidney disease, and osteoarthritis; and duration of wheelchair use prior to bariatric surgery.
Laboratory data and medication use were collected preoperatively at the preoperative-anesthetic visit closest to bariatric procedure and postoperatively at 1 year office visit to the department of bariatric surgery.
Perioperative variables included American Society of Anesthesiologist (ASA) score, date of procedure, duration, type (RYGB and SG), and technique (laparoscopic, open or laparoscopic converted to open). In addition, concurrent procedures performed, intraoperative complications, estimated intraoperative blood loss, date of discharge, and postoperative length of hospital stay and discharge destination were also collected.
Postoperative complications were categorized as major and minor as per ASMBS outcome reporting standards [22]. Major complications included 30-day mortality, reoperation, hospital readmissions, venous thromboembolism events, gastrointestinal hemorrhage requiring blood transfusion or surgical or endoscopic intervention, bowel obstruction requiring reoperation, anastomotic leak requiring reoperation or percutaneous drainage, myocardial infarction, new-onset heart failure, renal failure requiring the initiation of dialysis, respiratory failure requiring intubation, chronic nausea/vomiting requiring total parenteral nutrition, surgical site infection (SSI) (including superficial, deep, and organ space SSIs) requiring debridement, washout, or percutaneous drainage, and gastric sleeve stenosis or obstruction requiring revision to RYGB. Minor complications included marginal ulcer formation, anastomotic stricture, respiratory infection, urinary tract infection requiring antibiotics, trocar site SSI managed with drainage or local wound care, acute renal failure managed with intravenous (IV) fluids, nausea/vomiting or dehydration requiring IV fluids as an inpatient, postoperative ileus managed non-operatively, vitamin/mineral deficiency requiring IV supplementation, anemia, symptomatic cholelithiasis, new-onset atrial fibrillation, and diabetic ketoacidosis. Composite morbidity was defined as the unique count of all the patients who developed one or more early postoperative complication.
Outcomes of interest at 1 year postoperatively included percentage excess weight lost and changes in preoperative weight-related comorbidities, including obstructive sleep apnea (OSA), hypertension (HTN), dyslipidemia, gastroesophageal reflux disease (GERD), and diabetes mellitus (DM). Changes in these comorbidities were categorized as improved, unchanged, or unavailable. Improvement in these comorbidities was defined as per the ASMBS standardized reporting of outcomes. Ambulatory status among the patients with preoperative impaired mobility was noted as either walking unaided, walking with partial assistance (walker/cane) without using a wheelchair or motorized scooter, or no change from the preoperative status.
Statistical Analysis
Continuous variables were summarized by medians and quartiles, and categorical variables were summarized by counts and percentages. Prior to analysis, any missing values were singly imputed by their conditional medians or modes using the transcan function from the Hmisc package in R [24]. A 1:5 (WC:AMB patients) propensity score-matched analysis was performed. Patients were matched based on factors that have been previously shown to be associated with the risk of perioperative morbidity and mortality following bariatric surgery as well as weight-related outcomes more commonly seen in non-ambulatory patients [25,26,27,28,29]. These factors included age, gender, body mass index (BMI), type of bariatric procedure performed, history of venous thromboembolism, OSA, HTN, dyslipidemia, GERD, and DM. The propensity scores were estimated using a logistic regression model involving these predictors, resulting in a score on the scale of the linear predictor. Using these scores, matching of group A to similar group B patients proceeded using functionality in the Matching package [30]. Balance was assessed using absolute standardized mean differences, which are the absolute value of the difference in means between groups, expressed as a percentage of the standard deviation within treated patients. Once the groups were well matched, comparisons were made between the WC group and the AMB group and also between WC-D and WC-A groups with respect to 30-day and 1-year outcomes using chi-square analysis, Fisher’s exact test, or two-sample t tests. Perioperative outcomes and metabolic outcomes at 1 year were also stratified by procedure type. All statistical analyses were performed using R version 3.3.1, and p < 0.05 was considered statistically significant [31].
Results
Patient Demographics
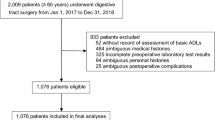
A total of 3643 patients met inclusion criteria; 93 patients (2.6%) had impaired mobility. Following propensity score matching (1:5), a total of 558 patients remained for analysis; 465 patients (80%) were ambulatory while 93 patients (20%) had impaired mobility. Table 1 details the patient demographics of the two groups. Following propensity score matching, there was no statistically significant difference between the two groups with respect to age, gender, BMI, type of procedure performed, OSA, HTN, hyperlipidemia, GERD, or venous thromboembolism events. Among the unmatched patient comorbidities, patients with impaired mobility were more likely to have chronic obstructive pulmonary disease (COPD), a history of myocardial infarction, chronic renal disease, osteoarthritis, and previous abdominal surgeries in comparison to the ambulatory cohort (p < 0.05). For the WC group, the median duration of mobility impairment prior to bariatric surgery was 24 months (IQR 12–57).
Perioperative Details
Table 2 details the perioperative variables for the WC and AMB groups. Significant differences existed between the two groups with respect to American Society of Anesthesiology (ASA) score, procedure time, and intraoperative blood loss, all of which were higher in patients with impaired mobility.
30-Day Postoperative Outcomes
Table 3 details the 30-day outcomes for the WC and AMB groups and a subgroup analysis within the WC cohort according to the procedure performed (RYGB and SG). Median postoperative length of stay (LOS) was higher in patients with impaired mobility versus ambulatory cohort (p ≤ 0.001). With respect to discharge destinations, patients in WC groups were less likely to be discharged to home in comparison to the patients in AMB group (p ≤ 0.001). There was no statistically significant difference between the two groups with respect to composite morbidity, major complications, minor complications, 30-day mortality, 30-day unplanned reoperations, or 30-day unplanned hospital readmissions. Incidence of venous thromboembolism was similar in both groups (1%; p > 0.99). The RYGB group within the WC cohort experienced higher incidence of composite morbidity, major and minor complications, reoperations, readmissions, and mortality. Table 4 details the 30-day outcomes for the patients that were completely wheelchair dependent (WC-D) and those with the ability to ambulate with assistive devices (WC-A). There were no differences between the WC-D and WC-A groups with respect to postoperative LOS, discharge destination, composite morbidity, mortality, reoperation, and readmission rate.
1-Year Ambulatory and Cardiometabolic Outcomes
At 1 year, 66% (n = 61/93) and 73% (n = 338/465) were available for follow-up in the WC and AMB groups. Table 5 details the changes in ambulation status at 1 year postoperatively. In terms of ambulation, 38/61 patients (62%) experienced improvement in their mobility status; 13 patients (21%) were able to walk unaided while 25 patients (41%) required only partial support with the use of a cane or walker, all eliminating the use of a wheelchair or scooter. With respect to individual groups, WC-D patients experienced lower improvement in their ambulation compared to WC-A patients (39 vs 72%) at 1 year.
Table 6 details the cardiometabolic outcomes at 1 year postoperatively for the WC and AMB groups. The two groups were equivalent with respect to BMI and percentage excess weight loss. Nevertheless, patients with impaired mobility were significantly less likely to improve in their cardiometabolic comorbidities, including OSA, HTN, and DM compared to the ambulatory cohort (p < 0.05). Among patients with impaired mobility who underwent RYGB had greater mean %EWL and were more likely to show improvement in diabetes and dyslipidemia outcomes compared to those undergoing SG. At 1 year postoperatively, there were no differences between the WC-D and WC-A groups with respect to BMI, %EWL, improvement in DM, HTN, dyslipidemia, GERD, and OSA.
Discussion
This study aimed to evaluate and compare the short-term surgical outcomes, both with respect to ambulatory status and weight-related metrics, following bariatric surgery in patients with impaired mobility, to the patients with no limitations to ambulation. The primary findings of this study are that through 1 year of follow-up after bariatric surgery, (1) among the patients available for follow-up at 1 year, 62% patients experienced improvement in ambulation, and (2) although there was a significant improvement in the metabolic outcomes of WC group patients at 1 year, the rate of improvement was lower than AMB group with respect to diabetes, hypertension, and sleep apnea (p < 0.05). Additionally, the results of this study suggest that patients with impaired mobility were more likely to have a longer postoperative hospitalization compared to an ambulatory cohort (p < 0.05). There were no statistical differences between the two cohorts with respect to early postoperative outcomes: composite morbidity, major complications, minor complications, mortality, reoperations, or readmissions.
Literature on the surgical outcomes after bariatric surgery in patients with impaired mobility is limited to few case series [16,17,18,19,20,21]. This may be the result of resistance among surgeons to operate on this population due to increased risk of postoperative VTE, multiple associated comorbidities, and inadequate weight loss postoperatively due to limited ambulation. Another important factor contributory to the paucity of literature may be the patient selection. Previous studies suggest that younger age, older age, limited functional status, poor social support, self-pay, and public insurance are associated with decreased odds of being offered bariatric procedures [32]. However, preoperative impaired ambulation and disability in morbidly obese subjects has been widely reported. Surgical intervention with a bariatric procedure in this patient population may help prevent further morbidity [13, 17, 33]. Possible mechanisms underlying improvement in physical functioning after bariatric surgery can be attributed to changes in joint loads, alterations in regional body fat distribution, and reductions in inflammation [34].
Several studies have reported an improvement in ambulation or physical functioning following bariatric surgery. A recent observational cohort study of 2221 individuals undergoing bariatric surgery showed a large percentage of patients experienced improvement compared with baseline, in physical function (77%) and walking time (60%) at 1 year postoperatively [13]. Similarly, a prospective study in obese patients, who underwent either bariatric surgery (n = 25) or medical management (n = 20) for weight loss, showed significant improvements in walking speeds and quality of life in the surgical cohort [35]. A few studies have also reported early improvements, with increase in 6-min walking distance at 3 months and 1 year postoperatively [34, 36,37,38].
Williams et al. reported early complications in 13% patients and improvement in ambulation in 71% patients in a cohort undergoing either RYGB or AGB operations [17]. Another study, including 6 patients (LRYGB, n = 5 and AGB, n = 1), reported mean percentage excess weight loss of 51% at 1 year and improvement in mobility in 2/6 patients. No patients developed early postoperative complications [16]. Case reports published on the outcomes of bariatric surgery in non-ambulatory patients with lower limb paralysis showed significant improvement in weight-related comorbidities and quality of life [19,20,21].
The findings of our study reinforce the improvement in ambulation demonstrated in the existing reported literature. Sixty-two percent patients (n = 38/61) experienced improvement in ambulation at 1 year postoperatively. However, the noted improvement may have been influenced by the fact that majority of patients who experienced improvement (n = 31/38) were able to ambulate for at least part of a normal day out of a wheelchair (WC-A group) and thus likely had a better chance of improvement of ambulation when compared to wheelchair-dependent individuals (WC-D group; n = 7/38). However, there was a lower percentage of patients in the WC-D group with follow-up available at 1 year (58 vs 70% in WC-A group), which may have resulted in an underestimate of actual improvement in the WC-D group. King et al. reported improvement in physical functioning decreased after 1 year, so a longer follow-up may help provide a better picture of the improvement in ambulation long term [13]. In terms of perioperative outcomes, there were statistically significant differences between the impaired mobility and ambulatory cohorts, including procedure time, intraoperative blood loss, postoperative length of stay, and improvement in diabetes, improvement in hypertension, and improvement in sleep apnea (p ≤ 0.05). Although propensity score matching was performed between the two groups, WC group had more comorbid disease preoperatively, based on greater proportion of patients with COPD, MI, chronic renal disease, osteoarthritis, and past surgical history for abdominal procedures, which may have been responsible for the statistically significant outcomes. A greater proportion of patients with impaired mobility were discharged to skilled nursing or rehabilitation facilities (p ≤ 0.001), which has been shown to influence hospital length of stay [39]. With respect to type of bariatric procedure performed within the WC cohort, RYGB was associated with a higher perioperative morbidity but also higher resolution of weight-related comorbidities (diabetes mellitus and dyslipidemia) when compared to SG. Procedure selection in this high risk cohort should be individualized and should be offered with an adequate balance between the risks of perioperative morbidity and resolution of cardiometabolic outcomes.
Our experience demonstrates bariatric surgery can improve ambulation and associated weight-related comorbidities in patients with impaired mobility. Subsequently, improved ambulation may have a positive impact on the chances for patients to return to work, limiting health care costs and increasing economy in the long run. As noted by the follow-up at 1 year (65.6%, n = 61/93), compliance may be an important factor for patients with impaired mobility undergoing bariatric surgery. Objective measures for improvement in physical activity, formalized physiotherapy, or coached exercise program individualized to patient needs should be incorporated as a part of closer follow-up for this patient population.
This study has one of the largest sample sizes of patients with impaired mobility who underwent bariatric surgery reported in the literature to date. This is one of the few studies comparing short-term metabolic outcomes of non-ambulatory patients to an ambulatory cohort. The study limitations include retrospective nature, use of single academic institution, non-standardized nature of index procedure, missing follow-up data at 1 year, and selection bias with respect to patients with limited ambulation who had higher chances of improvement when compared to wheelchair-bound patients.
Conclusion
Bariatric surgery is safe and feasible in higher risk patients with impaired mobility. Compared to ambulatory controls, patients with impaired mobility experience similar rates of perioperative morbidity. Improvement in weight-related comorbidities is less likely in the short term for patients with impaired mobility, but bariatric surgery is positively associated with more independent mobility.
References
Garver MJ, Focht BC, Dials J, et al. Weight status and differences in mobility performance, pain symptoms, and physical activity in older, knee osteoarthritis patients. Arthritis. 2014;2014:1–7. https://doi.org/10.1155/2014/375909.
Hergenroeder AL, Wert DM, Hile ES, et al. Association of body mass index with self-report and performance-based measures of balance and mobility. Phys Ther. 2011;91(8):1223–34. https://doi.org/10.2522/ptj.20100214.
Peytremann-Bridevaux I, Santos-Eggimann B. Health correlates of overweight and obesity in adults aged 50 years and over: results from the Survey of Health, Ageing and Retirement in Europe (SHARE). Obesity and health in Europeans aged ≥ or = 50 years. Swiss Med Wkly. 2008;138(17–18):261–6.
Oliveria SA, Felson DT, Cirillo PA, et al. Body weight, body mass index, and incident symptomatic osteoarthritis of the hand, hip, and knee. Epidemiology. 1999;10(2):161–6. https://doi.org/10.1097/00001648-199903000-00013.
Johnson VL, Hunter DJ. The epidemiology of osteoarthritis. Best Pract Res Clin Rheumatol. 2014;28(1):5–15. https://doi.org/10.1016/j.berh.2014.01.004.
Sellam J, Berenbaum F. Is osteoarthritis a metabolic disease? Joint Bone Spine. 2013;80(6):568–73. https://doi.org/10.1016/j.jbspin.2013.09.007.
Ratneswaran A, LeBlanc EA, Walser E, et al. Peroxisome proliferator-activated receptor δ promotes the progression of posttraumatic osteoarthritis in a mouse model. Arthritis Rheumatol (Hoboken, NJ). 2015;67(2):454–64. https://doi.org/10.1002/art.38915.
Hulens M, Vansant G, Lysens R, et al. Exercise capacity in lean versus obese women. Scand J Med Sci Sports. 2001;11(5):305–9. https://doi.org/10.1034/j.1600-0838.2001.110509.x.
Vincent HK, Heywood K, Connelly J, et al. Obesity and weight loss in the treatment and prevention of osteoarthritis. PM&R. 2012;4(5):S59–67. https://doi.org/10.1016/j.pmrj.2012.01.005.
de Souza SAF, Faintuch J, Valezi AC, et al. Gait cinematic analysis in morbidly obese patients. Obes Surg. 2005;15(9):1238–42. https://doi.org/10.1381/096089205774512627.
Vincent H, Zdziarski L, Wasser J. Chronic pain management in the obese patient: a focused review of key challenges and potential exercise solutions. J Pain Res. 2015;8:63. https://doi.org/10.2147/JPR.S55360.
Courcoulas AP, Christian NJ, Belle SH, et al. Weight change and health outcomes at 3 years after bariatric surgery among individuals with severe obesity. JAMA. 2013;310:2416–25. https://doi.org/10.1001/jama.2013.280928.
King WC, Chen J-Y, Belle SH, et al. Change in pain and physical function following bariatric surgery for severe obesity. JAMA. 2016;315(13):1362–71. https://doi.org/10.1001/jama.2016.3010.
Abu-Abeid S, Wishnitzer N, Szold A, et al. The influence of surgically-induced weight loss on the knee joint. Obes Surg. 2005;15(10):1437–42. https://doi.org/10.1381/096089205774859281.
Hooper MM, Stellato TA, Hallowell PT, et al. Musculoskeletal findings in obese subjects before and after weight loss following bariatric surgery. Int J Obes. 2007;31(1):114–20. https://doi.org/10.1038/sj.ijo.0803349.
Bairdain S, Flint RS, Lien C, et al. A case report of the early results of laparoscopic bariatric surgery in six completely nonambulatory patients. Surg Obes Relat Dis. 2013;9(5):e74–6. https://doi.org/10.1016/j.soard.2013.06.007.
Williams GJ, Georgiou PA, Cocker DM, et al. The safety and efficacy of bariatric surgery for obese, wheelchair bound patients. Ann R Coll Surg Engl. 2014;96(5):373–6. https://doi.org/10.1308/003588414X13946184901083.
Lutrzykowski M. Bariatric surgery in morbidly obese patients in wheelchairs. Obes Surg. 2008;18(12):1647–8. https://doi.org/10.1007/s11695-008-9466-2.
Miyano G, Kalra M, Inge TH. Adolescent paraplegia, morbid obesity, and pickwickian syndrome: outcome of gastric bypass surgery. J Pediatr Surg. 2009;44(3):e41–4. https://doi.org/10.1016/j.jpedsurg.2008.12.014.
Alaedeen DI, Jasper J. Gastric bypass surgery in a paraplegic morbidly obese patient. Obes Surg. 2006;16(8):1107–8. https://doi.org/10.1381/096089206778026389.
Perreault JR, Geigle PR, Gorman PH, et al. Improvement in weight loss and ambulation outcomes after gastric sleeve surgery for a person with chronic motor-incomplete tetraplegia: clinical case report. Spinal Cord. 2016;54(9):750–3. https://doi.org/10.1038/sc.2016.22.
Brethauer SA, Kim J, Chaar M, et al. Standardized outcomes reporting in metabolic and bariatric surgery. Surg Obes Relat Dis. 2015;11(3):489–506. https://doi.org/10.1016/j.soard.2015.02.003.
Harris PA, Taylor R, Thielke R, et al. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377–81. https://doi.org/10.1016/j.jbi.2008.08.010.
Harrell FE Jr. Hmisc: Harrell Miscellaneous. R package version 3.17-0. 2015. https://CRAN.R-project.org/package=Hmisc.
O’Rourke RW, Andrus J, Diggs BS, et al. Perioperative morbidity associated with bariatric surgery. Arch Surg. 2006;141(3):262–8. https://doi.org/10.1001/archsurg.141.3.262.
Livingston EH, Huerta S, Arthur D, et al. Male gender is a predictor of morbidity and age a predictor of mortality for patients undergoing gastric bypass surgery. Ann Surg. 2002;236(5):576–82. https://doi.org/10.1097/01.SLA.0000033321.22614.D6.
Still CD, Wood GC, Chu X, et al. Clinical factors associated with weight loss outcomes after Roux-en-Y gastric bypass surgery. Obesity (Silver Spring). 2014;22(3):888–94. https://doi.org/10.1002/oby.20529.
Stroh C, Michel N, Luderer D, et al. Risk of thrombosis and thromboembolic prophylaxis in obesity surgery: data analysis from the German Bariatric Surgery Registry. Obes Surg. 2016;26(11):2562–71. https://doi.org/10.1007/s11695-016-2182-4.
Cawley J, Sweeney MJ, Kurian M, et al. Predicting complications after bariatric surgery using obesity-related co-morbidities. Obes Surg. 2007;17(11):1451–6. https://doi.org/10.1007/s11695-008-9422-1.
Sekhon JS. Multivariate and propensity score matching software with automated balance optimization: the matching package for R. J Stat Softw. 2011;42(7). https://doi.org/10.18637/jss.v042.i07.
R Core Team. R: a language and environment for statistical computing. Vienna: R Foundation for Statistical Computing; 2015.
Santry HP, Lauderdale DS, Cagney KA, et al. Predictors of patient selection in bariatric surgery. Ann Surg. 2007;245(1):59–67. https://doi.org/10.1097/01.sla.0000232551.55712.b3.
King WC, Engel SG, Elder KA, et al. Walking capacity of bariatric surgery candidates. Surg Obes Relat Dis. 2012;8(1):48–59. https://doi.org/10.1016/j.soard.2011.07.003.
Miller GD, Nicklas BJ, You T, et al. Physical function improvements after laparoscopic Roux-en-Y gastric bypass surgery. Surg Obes Relat Dis. 2009;5(5):530–7. https://doi.org/10.1016/j.soard.2008.11.003.
Vincent HK, Ben-David K, Conrad BP, et al. Rapid changes in gait, musculoskeletal pain, and quality of life after bariatric surgery. Surg Obes Relat Dis. 2012;8(3):346–54. https://doi.org/10.1016/j.soard.2011.11.020.
Tompkins J, Bosch PR, Chenowith R, et al. Changes in functional walking distance and health-related quality of life after gastric bypass surgery. Phys Ther. 2008;88(8):928–35. https://doi.org/10.2522/ptj.20070296.
Josbeno DA, Jakicic JM, Hergenroeder A, et al. Physical activity and physical function changes in obese individuals after gastric bypass surgery. Surg Obes Relat Dis. 2010;6(4):361–6. https://doi.org/10.1016/j.soard.2008.08.003.
Vargas CB, Picolli F, Dani C, et al. Functioning of obese individuals in pre- and postoperative periods of bariatric surgery. Obes Surg. 2013;23(10):1590–5. https://doi.org/10.1007/s11695-013-0924-0.
Picone G, Mark Wilson R, Chou S-Y. Analysis of hospital length of stay and discharge destination using hazard functions with unmeasured heterogeneity. Health Econ. 2003;12(12):1021–34. https://doi.org/10.1002/hec.800.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval
For this type of study, formal consent is not required
Informed Consent
This study does not require informed consent.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Sharma, G., Nor-Hanipah, Z., Haskins, I.N. et al. Comparative Outcomes of Bariatric Surgery in Patients with Impaired Mobility and Ambulatory Population. OBES SURG 28, 2014–2024 (2018). https://doi.org/10.1007/s11695-018-3132-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-018-3132-0