Abstract
Background
Laparoscopic sleeve gastrectomy (LSG) is a favorable bariatric procedure. This study evaluated the status of LSG in China.
Methods
During the 4th International Forum of Bariatric and Metabolic Surgery in May 2016, Nanjing China, an on-the-spot questionnaire was filled out by 105 attending surgeons with experience of LSG. The feedback data was collected and analyzed.
Results
For preoperative preparations, surgeons preferred blood glucose control with insulin (61.0%), bowel cleaning (33.3%), and fasting and water deprivation (75.2%). For surgical techniques, surgeons preferred 36/38F bougie (86.7%), greater curvature mobilization with ultrasonic energy device (89.5%), direct transection of short gastric vessels (80%), antrum resection within 2–6 cm to the pylorus (84.8%) with 4.8 mm height stapler (72.4%), and 3.5 mm for corpus (94.3%). Whole stapler-line reinforcement, gastric sleeve fixation, leaking test, and abdominal drainage were preferred by 48.6, 62.9, 39, and 47.6% surgeons. For postoperative managements, surgeons preferred nasogastric tube insertion (33.3%), early liquid diet (69.6%), 4 weeks of liquid diet (55.2%), 2000 ml daily water intake before discharge (79%), 4 weeks of PPI (69.5%), and multi-vitamin supplementation 1 week after operation (77.1%). For postoperative complications, preferences were tachycardia as the onset of leak (81.0%) and oral contrast radiography for leak diagnosis (72.4%). Leak managements include US-guided percutaneous drainage (68.6%), nasogastric tube (87.6%), and parenteral nutrition (61%). For prolonged leak, enteral nutrition (87.6%) and Roux-en-Y bypass (84.8%) as the salvage procedure were preferred. 95.2% preferred endoscopic dilation for stricture.
Conclusions
LSG is gaining its wide application in China, but standardization of LSG is urgently needed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Laparoscopic sleeve gastrectomy (LSG) was initially described as a part of biliopancreatic diversion with duodenal switch (BPS-DS) for weight loss in 2000 [1]. In 2003, LSG was used as the first-stage procedure followed by laparoscopic Roux-en-Y gastric bypass (LRYGB) for super super obese patients to reduce the perioperative risk [2]. Gradually, LSG evolved as an independent bariatric procedure because of its effective excessive weight loss (EWL), safety, and simplicity. Subsequently, multiple randomized controlled trials (RCT) confirmed its efficacy, safety, and feasibility in clinical practice [3,4,5]. Therefore, in 2012, American Society for Metabolic and Bariatric Surgery (ASMBS) recognized LSG as an acceptable option as a primary bariatric procedure [6]. Since then, LSG has been widely accepted as an important procedure of bariatric surgery and gained its robust growth worldwide.
LSG was first introduced in China mainland in 2006 and prevailed since 2011 [7]. In 2014, the Chinese Society for Metabolic and Bariatric Surgery (CSMBS) recommended that LSG could be an independent procedure for the patients with obesity and type 2 diabetes mellitus (T2DM). Meanwhile, CSMBS issued the indications of LSG which were followed by most of the Chinese bariatric surgeons, including (1) BMI ≥ 32.5 kg/m2 with or without T2DM; (2) 27.5 kg/m2 < BMI < 32.5 kg/m2 with T2DM but failed conservative treatment and combined with at least two metabolic diseases or comorbidities; (3) Duration of T2DM ≤15 years with fasting C-peptide ≥ 50% of normal lower limit; (4) Waist circumference: male ≥ 90 cm, female ≥ 85 cm; and (5) Age within 16~65 years old. It is estimated that about 5000 cases of bariatric surgery were performed in China mainland in 2015, and LSG accounted for half of them. Chinese surgeons prefer LSG to other bariatric procedures because of the following advantages: (1) Effective EWL after LSG; (2) Less perioperative complications; (3) Less cost; and (4) Relatively less surgical difficulty.
During the 4th International Forum of Bariatric and Metabolic Surgery on May 5th, 2016, in Nanjing, China, most of the Chinese laparoscopic bariatric surgeons attended the meeting and shared their experience of LSG. The objective of this study is to evaluate the usage of LSG, including the surgical techniques and perioperative management, in China.
Methods
This was a prospective survey with an on-spot questionnaire to evaluate the application of LSG in China. Most of the surgeons, who were practicing LSG or intended to practice it in China mainland, attended the meeting, with a total of 120 surgeons. The survey included the following sections: (1) Surgeons’ character; (2) Preoperative preparations; (3) Surgical technique; and (4) Postoperative management.
The surgeons who never performed LSG were excluded from the survey, and the survey included 105 surgeons. The surgeons gave their feedback of the questionnaire through the voters. The data was collected and analyzed.
Results
Surgeons’ Character
All attendees were male surgeons from 25 of 34 provinces in China. 97.1% surgeons were from university hospitals. The remaining 2.9% were from tertiary referral hospitals. On the basis of annual surgical volume of LSG among 105 surgeons, the percentages were 51.4, 36.2, and 12.4% for 1–10, 11–50, and >50 procedures/year, respectively.
Preoperative Preparations
For patients with diabetes mellitus (DM), all the surgeons accepted that the preoperative fasting blood glucose should be <10 mmol/l. Sixty-one percent chose subcutaneous insulin injection as a drug of choice to lower the blood glucose levels, and 5.7% chose hypoglycemic agents, while 33.3% accepted both of them.
33.3% performed bowel cleaning with laxative before the operation, while 66.7% considered it unnecessary. 75.2% of the surgeons preferred preoperative fasting and water deprivation for 6–8 h, and 16.2% accepted that the preoperative fasting was necessary while water deprivation was not. 8.6% surgeons chose to follow the anesthesiologist’s advice.
Surgical Techniques
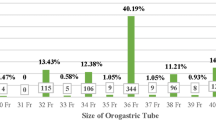
86.7% surgeons used 36F or 38F bougie for the intraoperative resection guidance, 11.4% preferred gastroscope, while 1.9% used nothing.
Mobilization of the greater curvature was typically done with the ultrasonic energy device (89.5%), followed by feedback-controlled bipolar sealing device (8.6%), and the bipolar (1.9%). Eighty percent of surgeons directly transected the short gastric vessels with the energy device, 14.3% chose titanium clips or hemolock to reinforce the hemostasis, while 5.7% used absorbable clips.
84.8% of surgeons preferred to start transection of antrum within 2–6 cm proximal to the pylorus, 12.4% chose to start beyond 6 cm, while 2.9% began transection at less than 2 cm. 72.4% preferred to transect the antrum with the 4.8 mm height stapler (green load), 8.6% used the 3.5 mm height stapler (blue load), 10.5% chose the multiple-height staple, and 8.6% used two 4.8 mm height stapler for first two fires. Gastric corpus was resected with 3.5 mm height stapler by 94.3% surgeons, with 4.8 mm height stapler by 1.9% surgeons, while 3.8% surgeons were uncertain. For fundus, 98.1% used 3.5 mm height stapler, while 1.9% were uncertain.
After the resection of stomach, 48.6% surgeons used to reinforce the whole staple line of the gastric sleeve, 33.3% did the reinforcement by only oversewing of the area between staple firings, while the rest (18.1%) considered the reinforcement unnecessary. 35.2% surgeons fixed the gastric sleeve to the surrounding tissue to prevent it from kinking or twisting, 62.9% did not, and 1.9% did it when it was necessary. Leak test was done by 39% surgeons. Among those who did the test, surgeons preferred bougie to do intraoperative air leak test (51.2%) and methylene blue test (29.3%), while gastroscopic air leak test was preferred by 19.5% surgeons.
Eighty-one percent surgeons directly removed the resected stomach from 12 mm port site, 17.1% used specimen bag, and the rest (1.9%) cut the specimen into pieces and removed them without using an endobag. An abdominal drain was put in by 47.6% surgeons, equal percentage did not place the drain, and 4.8% did it when it was necessary.
Postoperative Management
Placement of nasogastric tube was regarded as a routine procedure by 33.3% surgeons, while 53.3% surgeons did not insert it, and the rest (13.3%) placed it sometimes. For the urinary catheters, 23.8% considered keeping it for a longer duration, 52.4% would remove it when the patients regained consciousness, and 23.8% made decision on a case to case basis.
Liquid diet was prescribed by 69.6% surgeons when patients gained their consciousness, 15.2% did it only on resumption of gastrointestinal function (passing of flatus), and 15.2% allowed it the next day, after excluding of gastric leak by contrast radiography.
Seventy-nine percent of surgeons discharged the patients when the oral water intake was more than 2000 ml, 10.5% discharged the patients after suture removal of the incisions, and the rest (10.5%) discharged the patients 1 week after operation.
After discharge, 69.5% prescribed the intake of PPI for 4 weeks, 15.2% prescribed it for 2 weeks, 10.4% for 1 week, and 4.8% considered it unnecessary. All the surgeons agreed that postoperative liquid diet was indispensable, but the duration varied. 55.2% insisted that 4 weeks of liquid diet was necessary, 29.5% preferred 2 weeks, while 15.3% believed that 1 week was enough.
77.1% surgeons preferred to prescribe oral multi-vitamin 1 week after the operation, 17.1% prescribed it only if there were any fall in their levels or on the appearance of clinical signs of deficiency on follow-up visits, and 5.8% considered it unnecessary. Most of the surgeons (79.0%) preferred centrum (Wyeth Pharmaceuticals). The rest (21.0%) respected the patients’ choice.
For patients with poorly controlled diabetes after operation (no remission/improvement), 72.4% surgeons pointed out that the endocrinologists prescribed oral hypoglycemic agents (metformin or sulfonylurea), and 27.6% surgeons reported that endocrinologist prescribed insulin injection for DM control.
Postoperative Complications
LSG is considered as a relatively safe procedure, but complications like gastric leak or sleeve obstruction are always a major concern.
In this survey, most of the surgeons (71.4%) were uncertain about the cause of the leak, while 14.3, 9.5, and 4.8% believed that inappropriate stapler choice, stapling close to the esophagus, and no reinforcement of staple line may be responsible for the leak. For patients with the confirmed leak, tachycardia was regarded as the initial manifestation by 81.0% surgeons, fever by 8.6%, abdominal pain by 8.6%, and shoulder and back pain by 1.9%. Twenty-one percent surgeons believed that peritonitis was the major abdominal sign, while 79.0% believed that there was no specific sign when the leak happened.
In the patients with the suspicion of leak, oral contrast radiography was used by 72.4% surgeons for diagnosing it, 7.6% preferred ultrasound, 12.4% used enhanced CT scanning, while 7.6% preferred multiple radiological investigations. For the patients with abdominal drainage, 61.0% regarded drainage would be helpful to diagnose the leak, 24.8% regarded it as useless, while the rest (14.3%) were uncertain about it.
When the leak happened, 66.7% believed that the abdominal drainage would be helpful for the treatment, while 25.7% denied its usefulness, and 7.6% were uncertain about it. When the fluid collection was detected, 68.6% preferred US-guided percutaneous drainage, and 20% preferred CT guided drainage. 4.8% surgeons considered re-operation as the first treatment option, and 6.7% insisted of conservative therapy and observation if the patient was in stable condition. Nasogastric tube would be placed as a routine procedure by 87.6% surgeons, while 4.8% would place it based on the severity of the infection, 4.8% based on the postoperative duration, and 2.9% would not place it. For nutrition support, 39% preferred enteral nutrition, while 61% used parenteral nutrition. If the duration of leak prolonged, all the surgeons agreed that the fasting should be obligatory, but the percentages of enteral and parenteral nutrition were 87.6 and 12.4%, respectively. On failure of conservative therapy, 84.8% would consider Roux-en-Y bypass as a salvage procedure, 11.4% preferred total gastrectomy, while only 3.8% would perform laparoscopic repair of the leak.
Postoperative obstruction or stricture of gastric sleeve is another major complication of LSG. The possible etiology includes stapling too tight along the bougie (65.7%) and stapling too close to the gastric angle (21.9%), torsion (7.6%), and leak (4.8%). To diagnose the obstruction or stricture, 87.6% preferred upper gastrointestinal radiology, the others (12.4%) used gastroscope. For treatment of stricture, 95.2% would consider endoscopic dilation, 1.9% prefer laparoscopic gastroplasty, and 2.9% consider Roux-en-Y bypass.
Conclusion
During the past three decades, the rapid economic development dramatically increased the prevalence of obesity in China mainland [8, 9]. Though behavioral interventions and long-term drug treatments could offer benefits on weight loss maintenance, bariatric surgery results in greater body weight loss and higher remission of obesity-related diseases [10,11,12]. Among the bariatric procedures, LSG is relatively simple and offers favorable control of the body weight and the comorbidities [13]. Furthermore, LSG is associated with fewer early major and minor complications, compared with LRYGB [14]. Thus, LSG has become the most popular bariatric procedure in China mainland to treat obesity and accounts for more than 50% of all bariatric surgeries now. However, due to the short time of its application (10 years) in China mainland, some problems have come into notice based on the current on-the-spot survey.
The survey indicated that LSG is gaining its wide application in China mainland. Most of the provinces in China have started to perform LSG, though the distribution of surgical volume varies significantly. The surgeons in eastern part of China have performed 51.1% of overall LSG [7]. Only 12.4% Chinese surgeons reached the annual surgical volume of over 50 cases. Multiple reasons are responsible for this dilemma. In the long history, Chinese believe that overweight or obesity is the symbol of health, but not a disease. Some people, especially the elder Chinese population, also hold the concept that it is unacceptable to receive bariatric surgery for the obese people. Meanwhile, the concept of the bariatric and metabolic surgery among most of the Chinese physicians is not updated. Most of the Chinese patients with obesity first attend the outpatient clinic of endocrinology for medical consultation, but only a very few of them are advised to seek the benefit of bariatric surgery even though it is necessary. Moreover, bariatric surgery is not covered by the national medical insurance. Some patients with obesity, especially with morbid obesity, may not afford the expense of the bariatric surgery.
Based on this survey, we find the perioperative managements of LSG in China exhibits heterogeneity as listed in Table 1 and demands urgent improvement. It is widely accepted that the preoperative use of all hypoglycemic agents should be discontinued and replaced by insulin injection to achieve favorable blood glucose control to avoid postoperative hypoglycemia [15]. However, around 40% Chinese surgeons still prescribe oral hypoglycemic agents to control the blood glucose. The implementation of an enhanced recovery after bariatric surgery (ERABS) was confirmed to be safe, feasible, well-tolerated, and could significantly reduce the length of hospital stay with low readmission rates [16, 17]. It includes a series of perioperative evidence-based interventions to reduce the perioperative surgical stress by maintaining physiological function, enhancing mobilization, reducing pain, and facilitating postoperative early oral nutrition [18]. The protocol of ERABS for LSG was first proposed by Lemanu in 2013 [19]. In 2016, the guidelines of ERABS application for perioperative care in bariatric surgery were recommended by Enhanced Recovery After Surgery (ERAS) Society for clinical practice [18]. Based on the survey of postoperative length of hospital stay, placement of nasogastric tube, and duration of urinary catheter removal, we found that ERABS was not widely accepted by Chinese bariatric surgeons. Radical antral resection was reported to be associated with higher postoperative EWL and low incidence of reflux symptoms (2 versus 6 cm from the pylorus), thus, guarantee greater and better maintained weight loss [20]. However, 15.3% of Chinese surgeons did not transect the antrum within 2–6 cm from the pylorus. The indications for bariatric and metabolic surgery, followed by most of the Chinese bariatric surgeons, were issued by CSMBS in 2014, but the detailed managements of LSG remain missing in China. The protocol of bariatric surgery could be divided into three types: north (Shenyang), middle (Nanjing), and south (Guangzhou). These three cities provide the training for the most of the new bariatric surgeons. It can be speculated that the training would have influence on the surgeons’ decision of the practice. Other factors that contribute to the surgeons’ practice still require further investigation.
The laparoscopic bariatric surgery is a new emerging discipline in China mainland, with overall history of less than 15 years. The application of LSG also remains at the early stage. Current outlined guidelines or expert consensus on bariatric surgery are adapted from the Western guidelines, but not based on Chinese clinical data. As a matter of fact, solid clinical evidence, regarding the therapeutic effectiveness on obesity and T2DM in China mainland remains missing. Though key points of LSG have reached expert consensus, the practical procedure varies from surgeons to surgeons based on this survey [21, 22]. Some Chinese bariatric surgeons modify the procedure to decrease the intraoperative difficulty and postoperative complications. We believe that the main reason for this dilemma is due to the absence of national guidelines. In view of ethnic and diet difference from the Western population, more clinical trials involving Chinese patients should be carried out to validate the effectiveness of this procedure. Moreover, updating the Chinese version of guideline is urgently required.
Another major problem is the training of the new practicing bariatric surgeons. In China mainland, most of the bariatric surgeons are general surgeons, and the doing laparoscopic operation is not a tough challenge for them. As a matter of course, these surgeons assume that sleeve gastrectomy is just a simple procedure to form the gastric sleeve, but neglect the minor but important technical points and the value of postoperative follow-up. LSG is relatively simple procedure; however, it still requires strict preoperative evaluation, standard intraoperative procedure, and regular postoperative follow-up to secure its therapeutic effect and decrease the LSG-related complications. It is reported that the technical skill of the practicing bariatric surgeons varies widely, and it is negatively correlated with the postoperative complications [23]. The known best-practice techniques warrant the better surgical outcomes and guarantee the patients’ safety. The training of the new practicing bariatric surgeons should aim to improve the quality of LSG in forms of proper patient selection, standard surgical procedure, and scientific management of the postoperative complications. Meanwhile, the international and domestic communications should be included in the training to exchange and share the better understanding of LSG.
It is estimated that a total of 168 Chinese hospitals were practicing bariatric surgery in 2015, including those performs open bariatric surgery for T2DM, and the number is still increasing. However, a national registry system for bariatric surgery has not been established yet, and the data with regard to bariatric surgery remains unknown. The registry system offers the major source of data on the effectiveness and comprehensive trend of the bariatric surgery. It may reflect the flaws and defects within the bariatric surgery, which will be of great help to the further improvement. Meanwhile, the registry system may be helpful to influence the medical resource allocation and provide public education. Besides the registry system, more attention should be paid on clinical trials. Though laparoscopic bariatric surgery has been practiced in China for 15 years, RCTs with good quality remain missing to demonstrate its effectiveness in Chinese ethics. We still cannot provide solid evidence to convince the Chinese physicians and patients with obesity to accept the bariatric surgery. With the data retrieving from the registry system or RCTs , the bariatric surgery may gain its wider acceptance in China mainland.
It is worth noting that there are some limitations of this survey. First, this on-the-spot-survey is mainly focused on the technical aspects and perioperative managements of the LSG, while patient’s data and postoperative outcomes are not included. Second, not all the Chinese bariatric surgeons attended the conference, thus, current data may not reflect the true status of LSG within China mainland. Third, data in terms of postoperative follow-up is not included. For bariatric surgery, follow-up is an important part to monitor the therapeutic effect and manage the LSG-related complications. Fourth, the cost of LSG is not included. Bariatric surgery is not covered by the medical insurance in China. Analysis of cost and effect of bariatric surgery in patients with obesity may influence the patient’s acceptance. Thus, the data retrieving from the survey should be interpreted carefully.
The survey indicated that the techniques of LSG in China mainland exhibited great variation, though most Chinese surgeons followed the international guidelines and expert consensus. And the LSG in China mainland is at an early stage of development with small amount of annual surgical volume. To improve the quality of LSG in China mainland, the following steps should be done: standardization of the surgical procedure and perioperative management, training and certifying the surgeons, establishment of national registry system, and updating the national guidelines. Moreover, more domestic data in terms of RCTs or clinical trials with large patients sample to validate the effectiveness of LSG is needed.
References
Ren CJ, Patterson E, Gagner M. Early results of laparoscopic biliopancreatic diversion with duodenal switch: a case series of 40 consecutive patients. Obes Surg. 2000;10(6):514–523; discussion 24. doi:10.1381/096089200321593715.
Regan JP, Inabnet WB, Gagner M, et al. Early experience with two-stage laparoscopic Roux-en-Y gastric bypass as an alternative in the super-super obese patient. Obes Surg. 2003;13(6):861–4. doi:10.1381/096089203322618669.
Kehagias I, Karamanakos SN, Argentou M, et al. Randomized clinical trial of laparoscopic Roux-en-Y gastric bypass versus laparoscopic sleeve gastrectomy for the management of patients with BMI < 50 kg/m2. Obes Surg. 2011;21(11):1650–6. doi:10.1007/s11695-011-0479-x.
Karamanakos SN, Vagenas K, Kalfarentzos F, et al. Weight loss, appetite suppression, and changes in fasting and postprandial ghrelin and peptide-YY levels after Roux-en-Y gastric bypass and sleeve gastrectomy: a prospective, double blind study. Ann Surg. 2008;247(3):401–7. doi:10.1097/SLA.0b013e318156f012.
Lee WJ, Chong K, Ser KH, et al. Gastric bypass vs sleeve gastrectomy for type 2 diabetes mellitus: a randomized controlled trial. Arch Surg. 2011;146(2):143–8. doi:10.1001/archsurg.2010.326.
Updated position statement on sleeve gastrectomy as a bariatric procedure. Surgery for obesity and related diseases: official journal of the American Society for Bariatric Surgery. 2012;8(3):e21–6. doi:10.1016/j.soard.2012.02.001.
Du X, Dai R, Zhou HX, et al. Bariatric surgery in China: how is this new concept going? Obes Surg. 2016; doi:10.1007/s11695-016-2204-2.
Reynolds K, Gu D, Whelton PK, et al. Prevalence and risk factors of overweight and obesity in China. Obesity (Silver Spring). 2007;15(1):10–8. doi:10.1038/oby.2007.527.
Xi B, Liang Y, He T, et al. Secular trends in the prevalence of general and abdominal obesity among Chinese adults, 1993–2009. Obesity reviews: an official journal of the International Association for the Study of Obesity. 2012;13(3):287–96. doi:10.1111/j.1467-789X.2011.00944.x.
Gloy VL, Briel M, Bhatt DL, et al. Bariatric surgery versus non-surgical treatment for obesity: a systematic review and meta-analysis of randomised controlled trials. BMJ. 2013;347:f5934. doi:10.1136/bmj.f5934.
Yanovski SZ, Yanovski JA. Long-term drug treatment for obesity: a systematic and clinical review. JAMA. 2014;311(1):74–86. doi:10.1001/jama.2013.281361.
Dombrowski SU, Knittle K, Avenell A, et al. Long term maintenance of weight loss with non-surgical interventions in obese adults: systematic review and meta-analyses of randomised controlled trials. BMJ. 2014;348:g2646. doi:10.1136/bmj.g2646.
Sakran N, Raziel A, Goitein O, et al. Laparoscopic sleeve gastrectomy for morbid obesity in 3003 patients: results at a high-volume bariatric center. Obes Surg. 2016; doi:10.1007/s11695-016-2063-x.
Osland E, Yunus RM, Khan S, et al. Postoperative early major and minor complications in laparoscopic vertical sleeve gastrectomy (LVSG) versus laparoscopic Roux-en-Y gastric bypass (LRYGB) procedures: a meta-analysis and systematic review. Obes Surg. 2016; doi:10.1007/s11695-016-2101-8.
Mechanick JI, Youdim A, Jones DB, et al. Clinical practice guidelines for the perioperative nutritional, metabolic, and nonsurgical support of the bariatric surgery patient—2013 update: cosponsored by American Association of Clinical Endocrinologists, The Obesity Society, and American Society for Metabolic & Bariatric Surgery. Obesity (Silver Spring). 2013;21(Suppl 1):S1–27. doi:10.1002/oby.20461.
Awad S, Carter S, Purkayastha S, et al. Enhanced recovery after bariatric surgery (ERABS): clinical outcomes from a tertiary referral bariatric centre. Obes Surg. 2014;24(5):753–8. doi:10.1007/s11695-013-1151-4.
Barreca M, Renzi C, Tankel J, et al. Is there a role for enhanced recovery after laparoscopic bariatric surgery? Preliminary results from a specialist obesity treatment center. Surgery for obesity and related diseases: official journal of the American Society for Bariatric Surgery. 2016;12(1):119–26. doi:10.1016/j.soard.2015.03.008.
Thorell A, MacCormick AD, Awad S, et al. Guidelines for perioperative care in bariatric surgery: Enhanced Recovery After Surgery (ERAS) society recommendations. World J Surg. 2016;40(9):2065–83. doi:10.1007/s00268-016-3492-3.
Lemanu DP, Singh PP, Berridge K, et al. Randomized clinical trial of enhanced recovery versus standard care after laparoscopic sleeve gastrectomy. The British journal of surgery. 2013;100(4):482–9. doi:10.1002/bjs.9026.
Obeidat F, Shanti H, Mismar A, et al. The magnitude of antral resection in laparoscopic sleeve gastrectomy and its relationship to excess weight loss. Obes Surg. 2015;25(10):1928–32. doi:10.1007/s11695-015-1642-6.
Rosenthal RJ, Diaz AA, Arvidsson D, et al. International sleeve gastrectomy expert panel consensus statement: best practice guidelines based on experience of >12,000 cases. Surgery for obesity and related diseases: official journal of the American Society for Bariatric Surgery. 2012;8(1):8–19. doi:10.1016/j.soard.2011.10.019.
Gagner M, Deitel M, Erickson AL, et al. Survey on laparoscopic sleeve gastrectomy (LSG) at the fourth international consensus summit on sleeve gastrectomy. Obes Surg. 2013;23(12):2013–7. doi:10.1007/s11695-013-1040-x.
Birkmeyer JD, Finks JF, O'Reilly A, et al. Surgical skill and complication rates after bariatric surgery. N Engl J Med. 2013;369(15):1434–42. doi:10.1056/NEJMsa1300625.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval
For this type of study, formal consent is not required.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Lin, S., Guan, W., Hans, P. et al. Status of Laparoscopic Sleeve Gastrectomy in China: A National Survey. OBES SURG 27, 2968–2973 (2017). https://doi.org/10.1007/s11695-017-2727-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-017-2727-1