Abstract
Background
Bariatric surgery is a safe and established treatment option of morbid obesity. Mere percentage of excess weight loss (%EWL) should not be the only goal of treatment.
Methods
One hundred seventy-three obese patients were included in the study. They underwent either Roux-en-Y gastric bypass (RYGB; n = 127, mean body mass index (BMI) 45.7 ± 5.7 kg/m2) or sleeve gastrectomy (SG; n = 46, mean BMI 55.9 ± 7.8 kg/m2) for weight reduction. Body weight and body composition were assessed periodically by bioelectrical impedance analysis.
Results
After 1 year of observation, %EWL was 62.9 ± 18.0 % in RYGB and 52.3 ± 15.0 % in SG (p = 0.0024). Body fat was reduced in both procedures with a slight preference for SG, and lean body mass was better preserved in the RYGB group. Due to significant differences in the initial BMI between the two groups, an analysis of covariance was performed, which demonstrated no significant differences in the %EWL as well as in the other parameters of body composition 1 year after surgery. Using percentage of total weight loss to evaluate the outcomes between the two procedures, no significant difference was found (31.7 ± 8.4 % in RYGB and 30.5 ± 7.6 % in SG patients, p > 0.4).
Conclusions
Excess weight loss is highly influenced by the initial BMI. Total weight loss seems to be a better measurement tool abolishing initial weight differences. SG and RYGB do not differ in terms of body composition and weight loss 1 year after surgery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Among various therapeutic strategies for morbid obesity, bariatric surgery has evolved as an effective and safe option that achieves sustained weight reduction with improvement of comorbidities [1–3]. In the past, many published studies compared the results of the most commonly performed operative procedures: Roux-en-Y gastric bypass (RYGB) and sleeve gastrectomy (SG). There is still a weak evidence of superiority for either of the surgical techniques regarding weight loss and improvement of comorbidities [4, 5]. Early results of the Swiss Multicenter Bypass or Sleeve Study (SM-BOSS) showed that SG and RYGB have almost the same efficacy in terms of excessive body mass index loss 1 year after the operation (72.3 vs. 76.6 %, p = 0.2), increased quality of life, as well as reduction of comorbidities, particularly diabetes mellitus (57.7 vs. 67.9 %) 1 year after surgery [6]. On the other hand, results published by Strain et al. favor RYGB with percentage of excess weight loss (%EWL) reaching 70.4 % in comparison to only 49 % in SG patients (p > 0.0001) 1 year after surgery [7]. Moreover, it has not been investigated if changes in the body composition differ between the two surgical procedures.
Post bariatric surgery weight loss should be targeted towards reduction of body fat with a maximum preservation of body cell mass; otherwise, metabolic and physical capacities of the patient may be impacted negatively [8]. Changes in body composition after bariatric surgery such as a sustained loss of body fat are associated with an inevitable loss of lean body mass. This misbalance leads to an undesirable disturbance in resting metabolic rate, body temperature, and even weight maintenance [9]. To evaluate the efficacy of SG and RYGB, it is important, apart from the amount of weight loss, to investigate the quality of the remaining body composition. But different initial weight seems to be a relevant confounding variable in terms of %EWL and body composition when comparing surgical procedures in bariatric surgery [10]. Favoring SG for the treatment of super obese patients by many bariatric surgeons leads to large differences of the initial body mass index (BMI). Therefore, we conducted a prospective study comparing RYGB and SG, regarding their effects on the status of body composition using an analysis of covariance in order to adjust for the initial BMI. Additionally we tested if %EWL is a good parameter to characterize bariatric success or if percentage of total weight loss (%TWL) is a superior parameter.
Materials and Methods
Subjects
This open, single-center, prospective study was applied on 173 obese patients from January 2007 to February 2012. Approval from the local institutional review board (IRB) and written informed consent of all individuals were obtained.
Patients were selected for surgery according to the German guidelines, namely body mass index (BMI) over 40 or 35 kg/m2 with one or more comorbidities such as diabetes mellitus or obstructive sleep apnea [11].
Operative Technique
All operations were done laparoscopically using a standard technique with no conversion to open surgery. RYGB was performed with a 150-cm antecolic Roux–limb with a linear stapled gastrojejunostomy and a 50-cm long biliopancreatic limb.
For SG, a 42-Fr bougie was inserted along the lesser curvature for calibration of the gastric sleeve starting the linear stapling 5 cm proximal to the pylorus up to the angle of Hiss. Oversewing of staple line was only performed in case of bleeding or insecure appearing staples.
Follow-Up
Baseline body measurements were taken 1 day before the operation. Postoperative follow-up was done in the outpatient clinic after 6 weeks, 18 weeks, 6 months, 9 months, and 12 months. On each visit, body weight as well as body composition were assessed by bioelectrical impedance analysis using Nutriguard-M (Data Input GmbH, Darmstadt, Germany).
Additionally, all patients received individualized nutritional counseling at each visit from a nutritionist specialized in bariatric surgery.
The bioelectrical impedance analysis (BIA) is a common and frequently used tool for postbariatric evaluation of body composition. It provides accurate values comparable to those obtained by dual-energy X-ray absorptiometry (DXA) at low cost [12].
All patients were asked to rest for 30 min; after which, the BIA was performed by applying four silver electrodes, with two detecting electrodes placed at the ulnar aspect of the right wrist and the right medial malleolus. Body cell mass (BCM), extracellular mass (ECM), ECM/BCM, lean body mass, phase angle, fat, and total body water were analyzed using the software Nutri-Plus.
Statistical Analysis
For qualitative parameters, absolute and relative figures are given. Quantitative variables are presented by mean values together with standard deviations. In order to compare differences between two time points (e. g., preintervention and 12 months after intervention), a t test for two paired samples was used. Furthermore, for each variable, an analysis of covariance was performed in order to adjust for the initial BMI value. All statistical calculations were done using SAS software, release 9.3 (SAS Institute Inc., Cary, NC, USA).
Results
One hundred seventy-three patients between 19 and 67 years were included in this study. Patients were offered RYGB as the standard procedure (n = 127, mean BMI 45.6 ± 5.7 kg/m2). Only patients with a BMI more than 60 kg/m2 or a history of previous operative procedures involving the small intestine were offered SG as an alternative (n = 46, mean BMI 55.9 ± 7.8 kg/m2) (Table 1).
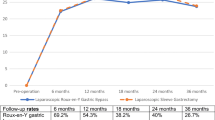
Our overall follow-up rate in the first year after surgery reached 98 %. However, we only included in our study data from patients who showed up exactly at all given six time points. Of the patients, 73.4 % successfully completed the first year follow-up with analyzable data sets (78.2 % in SG patients vs. 67.7 % in RYGB).
Weight Loss
%EWL was 62.9 ± 18 % in RYGB and 52.3 ± 15.0 % in SG 1 year after the operation (p = 0.0024). The largest difference in %EWL between the two groups—in favor of RYGB—was observed 9 months after surgery (+13.2 %) and thereafter decreased (+10.6 %) at 1 year (Fig. 1, Tables 2 and 3).
To compensate for the selection bias, an analysis of covariance was performed in order to adjust for the initial BMI value. The adjusted %EWL was no more significantly different between the two groups (p = 0.86).
In contrast to the %EWL results, no significant differences between the two procedures in terms of %TWL were found even without BMI adjustment (31.7 ± 8.4 % in RYGB and 30.48 ± 7.6 % in SG patients, p > 0.4) (Fig. 2).
Body Fat
After SG, body fat was reduced from 80.7 ± 19.4 kg to 43.1 ± 16.3 kg, while after RYGB, fat decreased from 58.6 ± 14.2 kg to 28.4 ± 12.7 kg. There was no significant difference between the two groups. Body fat loss was most significant during the first 6 weeks (11.1 kg in RYGB and 13.1 kg in SG) (Fig. 3, Tables 2 and 3).
Lean Body Mass
Lean body mass is defined as the fat-free body mass and is the sum of body cell mass (BCM) and extracellular mass (ECM). Lean body mass was better preserved in the RYGB group decreasing from 71.4 ± 15.9 kg to 61.7 ± 12 kg while showing a more pronounced reduction in the SG group from 83.5 ± 20.5 kg to 68.8 ± 13.7 kg (Tables 2 and 3).
Body Cell Mass
Loss of body cell mass, defined as the total mass loss of cellular body elements that are metabolically active [13], was similar in both groups, decreasing from 38.5 ± 9.7 kg to 30.0 ± 7 kg after RYGB, and in SG from 43.7 ± 10.6 kg to 33.1 ± 8 kg.
Similar to body fat, the loss in BCM was at its maximum during the first 6 weeks (5.8 kg in RYGB vs. 6.1 kg in SG) and then decreases with time except for a small increase in BCM loss in the SG group at 9 months after the operation (1.7 kg) (Fig. 4, Tables 2 and 3).
Cell Proportion
Cell proportion is defined as the percentage of cells within BCM [13]. This parameter shows a more pronounced reduction in the RYGB group (from 53.8 ± 4.7 % to 48.5 ± 5.4 %) than in the SG group (51 ± 5.1 % to 48.1 ± 5.1 %). The maximum decrease in cell proportion was also observed during the first 6 weeks after surgery (4.2 % after RYGB vs. 3.3 % after SG) (Table 2 and 3).
Discussion
Sleeve gastrectomy is the bariatric procedure with the highest increase in the world [14]. From its introduction as a stand-alone procedure in 2003 to date, number of sleeve resections has risen to more than 100,000 per year [15]. In the past, sleeve resection was recommended mainly for super obese patients and special medical conditions like previous abdominal interventions and/or chronic diseases. Nevertheless, gastric bypass was and is still considered to be the gold standard in bariatric surgery. Due to its marked effects on weight loss and comorbidities, it is currently under debate if sleeve resection is still the second best opinion for bariatric patients or if it has to be considered as a comparable alternative to the gastric bypass.
Current available data from the Swiss Multicentre Bypass or Sleeve Study (SM-BOSS) by Peterli et al. comparing SG and RYGB show that sleeve gastrectomy was associated with shorter operation time with a trend towards fewer complications than gastric bypass. However, both procedures were almost equally efficient regarding weight loss, improvement of comorbidities, and quality of life within the first 12 months after surgery. Due to its randomized study design, the mean BMI in the study population was 44 ± 11.1 kg/m in both arms [6]. Considering the gastric bypass as the gold standard in bariatric surgery and based on national and international recommendations, our institutions has suggested sleeve resection in patients with a BMI above 60 or other medical reasons. Our primary recommendation in any other patients was the laparoscopic gastric bypass. Therefore, like in most other studies, the intervention groups in our study differed significantly in terms of initial weight and BMI right from the beginning of our observation.
As expected, our subjective impression and first statistical analysis showed superiority of the bypass group. %EWL in the RYGB group was 62.9 ± 18 %, whereas a relatively low %EWL in the SG group was observed 12 month after surgery (52.3 ± 15.0 %).
Similar weight loss in extremely obese patients after sleeve resection has been reported in other studies. The University of Pittsburgh Medical Centre’s study showed that %EWL for super obese patients (mean BMI 65.3 ± 0.8) 12 months after SG was 46 % [10].
A confounding parameter which may have affected the %EWL in the SG Group is the used bougie size. All SG patients in this study were operated with a 42-Fr bougie, which is perhaps bigger than other surgeons would use. Based on the recent published data comparing outcomes after SG using different bougies, the size seems to have no influence on %EWL [16, 17]. Parikh et al. concluded in his meta-analysis that no difference regarding %EWL between bougie <40 Fr and bougie ≥40 Fr up to 36 months after SG could be detected [18].
Besides the pure excess weight loss, our first impression in changes in body composition showed better results in the RYGB group. If these results allow the conclusion that the RYGB is superior to the sleeve resection is still unclear.
Our study showed that the lean body mass is better preserved, in gastric bypass group—the lean body mass as the sum of body cell mass (BCM) and extracellular mass (ECM) can stay stable even if the BCM drops, which would be an unfavorable effect if the ECM rises to the same extent. This case would be even an indicator for a state of malnutrition. However, the better preservation of body composition in the RYGB group are in accordance with the publication of Strain et al. who also found different outcomes after both procedures. Nevertheless, these findings are in contrast to the early results of the SM-BOSS study published last year that show no significant difference in terms of percentage of excess BMI loss [6].
This discrepancy of the current literature is obviously contributed to the fact that most studies compare different populations of patients. The results from the SM-BOSS study strongly suggest that SG and RYGB lead to comparable results when they are offered to patients in the same BMI range.
That is why we used an analysis of covariance in order to adjust for the initial BMI value to validly compare our results to both above studies. The adjusted %EWL was not more significantly different between the two groups and neither were the other body composition parameters. The adjusted results are in agreement with the findings of the SM-BOSS study in terms of weight loss and at the same time provide a satisfactory explanation as to why the statistical results comparing the two procedures differ from the daily experience of many bariatric surgeons.
Because of the influence of initial BMI to %EWL, Ivan de Laar suggested the use of percentage of total weight loss (%TWL)—not %EWL—as an absolute assessment tool for bariatric outcomes and goals. With no need to set an initial reference point for the patient, %TWL can abolish initial BMI variations among different patients or non-randomized groups. Reflecting our results, showing major changes after BMI adjustment in terms of %EWL and a nearly identical %TWL along the six time points in the first year after operation, %TWL is the better choice to compare groups with different initial BMIs [19].
The maximum decrease in body fat, BCM, as well as the cell proportion was observed during the first 6 weeks after surgery. This negative body cell mass balance can be attributed to initial restriction or intolerance of food intake postoperatively that leads to increased muscle mass catabolism [20]. This negative balance may persist in spite of periodic nutritional assessment offered to all patients by our nutritionist recommending increased protein intake. Moreover, we encouraged all patients to follow an active physical fitness program aiming to preserve LBM despite increased postoperative catabolism.
Patients deemed to be chronically protein deficient postoperatively were advised to consume additional protein supplements via protein shakes, drinks, and/or powders.
According to the clinical practice guidelines for the perioperative nutritional support of the bariatric surgery patient published by The Obesity Society, and American Society for Metabolic and Bariatric Surgery, we recommended a minimal protein intake of 60 g/day and up to 1.5 g/kg ideal body weight per day in order to reduce the loss of lean body mass [21]. The results of our study shows that SG as well as RYGB leads to a decrease in body cell mass and cell proportion during the first 6 weeks and, following our results, both operations should be handled in the same way in terms of protein supplementation.
Limitations
Beside the small study population, a randomization is necessary to achieve a higher level of evidence. In addition, BIA estimates and does not actually measure the body cell mass. BIA devices are still accepted by many authors for monitoring changes in body composition within individuals over time [22, 23], but other studies demonstrated poor agreement between BIA and more advanced and precise techniques like dual X-ray absorptiometry, specific bioelectrical vector analysis, or air displacement plethysmography [24–26].
Conclusion
The lack of weight balance between the groups at baseline makes it hard to interpret the findings and conclusion, but proper statistical tools reveal that there seems to be no significant difference between SG and RYGB regarding excess body weight loss and body composition changes 1 year after the operation.
%TWL is a valuable measurement tool which can be used to assess and compare bariatric outcomes and abolish initial BMI differences.
A long-term prospective randomized study is required to support these results.
References
Buchwald H, Avidor Y, Braunwald E, et al. Bariatric surgery: a systematic review and meta-analysis. JAMA. 2004;292:1724–37.
Maggard MA, Shugarman LR, Suttorp M, et al. Meta-analysis: surgical treatment of obesity. Ann Intern Med. 2005;142:547–59.
Sjöström L, Lindroos A, Peltonen M, et al. Lifestyle, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med. 2004;351:2683–93.
Kehagias I, Karamanakos SN, Argentou M, et al. Randomized clinical trial of laparoscopic Roux-en-Y gastric bypass versus laparoscopic sleeve gastrectomy for the management of patients with BMI <50 kg/m2. Obes Surg. 2011;21:1650–6.
Woelnerhanssen B, Peterli R, Steinert RE, et al. Effects of postbariatric surgery weight loss on adipokines and metabolic parameters: comparison of laparoscopic Roux-en-Y gastric bypass and laparoscopic sleeve gastrectomy—a prospective randomized trial. Surg Obes Relat Dis. 2011;7:561–8.
Peterli R, Borbély Y, Kern B, et al. Early results of the Swiss Multicentre Bypass or Sleeve Study (SM-BOSS). A prospective randomized trial comparing laparoscopic sleeve gastrectomy and Roux-en-Y gastric bypass. Ann Surg. 2013;258:690–5.
Strain GW, Gagner M, Pomp A, et al. Comparison of weight loss and body composition changes with four surgical procedures. Surg Obes Relat Dis. 2009;5(5):582–7.
Zalesin KC, Franklin BA, Lillystone MA, et al. Differential loss of fat and lean mass in the morbidly obese after bariatric surgery. Metab Syndr Relat Disord. 2010;8(1):15–20.
Faria SL, Faria OP, Buffington C, et al. Dietary protein intake and bariatric surgery patients: a review. Obes Surg. 2011;21:1798–805.
Cottam D, Qureshi FG, Mattar SG, et al. Laparoscopic sleeve gastrectomy as an initial weight-loss procedure for high-risk patients with morbid obesity. Surg Endosc. 2006;20(6):859–63.
Runkel N, Colombo-Benkmann M, Hüttl TP, et al. Evidence-based German guidelines for surgery for obesity. Int J Color Dis. 2011;26(4):397–404.
Thomson R, Brinkworth GD, Buckley JD, et al. Good agreement between bioelectrical impedance and dual-energy X-ray absorptiometry for estimating changes in body composition during weight loss in overweight young women. Clin Nutr. 2007;26(6):771–7.
Kyle UG, Bosaeus I, De Lorenzo AD, et al. Bioelectrical impedance analysis part I: review of principles and methods. Clin Nutr. 2004;5:1226–43.
Buchwald H, Oien DM. Metabolic/bariatric surgery worldwide 2011. Obes Surg. 2013;23(4):427–36.
Gagner M, Buchwald JN. Comparison of laparoscopic sleeve gastrectomy leak rates in four staple-line reinforcement options: a systematic review. Surg Obes Relat Dis. 2014;10(4):713–23.
Hawasli A, Jacquish B, Almahmeed T, et al. Early effects of bougie size on sleeve gastrectomy outcome. Am J Surg. 2015;209(3):473–7.
Yuval JB, Mintz Y, Cohen MJ, et al. The effects of bougie caliber on leaks and excess weight loss following laparoscopic sleeve gastrectomy. Is there an ideal bougie size? Obes Surg. 2013;23(10):1685–91.
Parikh M, Issa R, McCrillis A, et al. Surgical strategies that may decrease leak after laparoscopic sleeve gastrectomy: a systematic review and meta-analysis of 9991 cases. Ann Surg. 2013;257(2):231–7.
Van de Laar A, de Caluwé L, Dillemans B. Relative outcome measures for bariatric surgery. Evidence against excess weight loss and excess body mass index loss from a series of laparoscopic Roux-en-Y gastric bypass patients. Obes Surg. 2011;21(6):763–7.
Friedrich AE, Damms-Machado A, Meile T, et al. Laparoscopic sleeve gastrectomy compared to a multidisciplinary weight loss program for obesity—effects on body composition and protein status. Obes Surg. 2013;23:1957–65.
Mechanick JI, Youdim A, Jones DB, et al. Clinical practice guidelines for the perioperative nutritional, metabolic, and nonsurgical support of the bariatric surgery patient—2013 update: cosponsored by American Association of Clinical Endocrinologists, The Obesity Society, and American Society for Metabolic and Bariatric Surgery. Obesity. 2013;21:1–27.
Buchholz AC, Bartok C, Schoeller DA. The validity of bioelectrical impedance models in clinical populations. Nutr Clin Pract. 2004;19(5):433–46.
Bosy-Westphal A, Later W, Hitze B, et al. Accuracy of bioelectrical impedance consumer devices for measurement of body composition in comparison to whole body magnetic resonance imaging and dual X-ray absorptiometry. Obes Facts. 2008;1(6):319–24.
Frisard MI, Greenway FL, Delany JP. Comparison of methods to assess body composition changes during a period of weight loss. Obes Res. 2005;13:845–54.
Deurenberg P. Limitations of the bioelectrical impedance method for the assessment of body fat in severe obesity. Am J Clin Nutr. 1996;64(3 suppl):449S–52.
Segal KR, Van Loan M, Fitzgerald PI, et al. Lean body mass estimation by bioelectrical impedance analysis: a four-site cross-validation study. Am J Clin Nutr. 1988;47:7–14.
Conflict of Interest
The authors declare that they have no competing interests.
Ethical Approval
Approval from the local institutional review board (IRB) was obtained and is in agreement with the Helsinki declaration.
Statement of Informed Consent
Informed consent was obtained from all individual participants included in the study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Mirko Otto and Mohamad Elrefai contributed equally to this work.
Rights and permissions
About this article
Cite this article
Otto, M., Elrefai, M., Krammer, J. et al. Sleeve Gastrectomy and Roux-en-Y Gastric Bypass Lead to Comparable Changes in Body Composition after Adjustment for Initial Body Mass Index. OBES SURG 26, 479–485 (2016). https://doi.org/10.1007/s11695-015-1792-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-015-1792-6