Abstract
Background
This study assessed eating disorder pathology in persons with obesity before and after Roux-en-Y gastric bypass (RYGB) and biliopancreatic diversion with duodenal switch (DS), in a 5-year follow-up study.
Methods
Sixty participants with BMI 50–60 kg/m2 were randomly assigned to RYGB (n = 31) or DS (n = 29). The participants completed the Eating Disorder Examination-Questionnaire (EDE-Q) before and 6 months, 1 year, 2 years, and 5 years after surgery.
Results
Before surgery, the prevalence of objective bulimic episodes was 29 % in the RYGB group and 32 % in the DS group. The prevalence improved during the first 12 months after surgery in both groups. After 5 years, the prevalence of objective bulimic episodes was 22 % in the RYGB group and 7 % in the DS group. The difference between groups throughout follow-up was non-significant (logistic regression model). A linear mixed model showed that global EDE-Q score was not a significant predictor for weight loss after surgery, but participants reporting objective bulimic episodes before surgery had significantly lower BMI than those with no episodes after 2 years (p = 0.042) and 5 years (p = 0.013). Global EDE-Q score was significantly lower in the DS group after 5 years (p = 0.009) (linear mixed model).
Conclusions
Objective bulimic episodes but not global EDE-Q score before surgery predicted greater weight loss after RYGB and DS. The DS group had a significantly lower global EDE-Q score than the RYGB group 5 years after surgery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bariatric surgery is currently the most viable weight loss intervention in persons with obesity. Postoperative weight loss, however, varies considerably. Some patients experience suboptimal weight loss and some regain a substantial portion of the initial weight lost [1, 2]. Psychological factors [3], changes in gut hormones and appetite [4], and pre- and postsurgical eating patterns [5–8] may all contribute to explain this variation in weight loss outcome. A recent meta-analysis showed that preoperative weight loss might be positively associated with postbariatric weight loss, while preoperative BMI, super obesity, and personality disorders were negatively associated with weight loss after surgery [9]. While some studies suggest that preoperative eating behavior does not predict postoperative weight loss [9–14], postoperative binge eating, loss of control (LOC) eating, and grazing have consistently predicted poorer outcome in regard to weight loss and psychosocial measures after surgery [5–8, 15, 16]. Reports on binge eating as predictor for postoperative weight loss are numerous [8, 17–19], but controversy still remains regarding the role of preoperative binge eating as predictor of treatment success [7]. Furthermore, few studies have evaluated effects of different surgical procedures in regard to eating habits. In general, more research is needed to investigate whether presurgical factors can predict weight loss after bariatric surgery, as substantiated knowledge may help target interventions and improve outcomes [9]. Our longitudinal randomized controlled study with RYGB and DS patients may shed more light on the complex relationship between pre- and postoperative eating disorder behavior and cognitions and weight loss after bariatric surgery.
The aim of this study was to report and compare the prevalence of eating disorder pathology after RYGB and DS and to investigate if preoperative eating disorder symptoms predict postoperative weight loss after these two surgical procedures in a super obese population.
Materials and Methods
This report is part of a randomized controlled trial conducted in two Scandinavian university hospitals. The primary endpoint of the trial was change in BMI after surgery as reported on previously [20]. The design of the trial has previously been described [20–23]. Individuals with BMI (kg/m2) 50 to 60, age 20–50 years, and failed attempts of sustained weight loss by medical and life style measures were included. The enrollment period was from February 2006 to August 2007. Participants were randomized to either laparoscopic RYGB or laparoscopic DS [20, 22]. The RYGB group had a 25-ml gastric pouch, 150-cm alimentary limb, and 50-cm biliopancreatic limb. The DS group received a sleeve gastrectomy along a nasogastric tube of 30–32 F, a 200-cm alimentary limb, and 100-cm common channel [20]. All participants were informed about possible benefits, risks, and side effects of respective surgery. Informed consent was obtained from all individual participants included in the study. The Regional Ethics Committees for Medical Research in Eastern Norway and Gothenburg, Sweden, approved the study which was registered in Clinicaltrials.gov (NCT00327912).
Eating Disorder Examination (EDE-Q)
All participants filled out a Swedish or Norwegian translation of The Eating Disorder Examination-Questionnaire (EDE-Q) at their baseline examination and subsequently at 6 months, 1 year, 2 years, and 5 years of follow-up. EDE-Q has well-established psychometric properties [24]. A non-validated version of EDE-Q was used in this study. A later Norwegian and Swedish version of EDE-Q (version 6.0) with minor changes has, however, been validated [25–27]. The EDE-Q focuses on the previous 28 days and measures key eating disorder behavior- and cognitive symptoms. The questionnaire distinguishes between three types of binge eating: objective bulimic episodes (eating an unusually large amount of food with a sense of having lost control over eating), subjective bulimic episodes (eating not a large amount of food but have a sense of having lost control over eating), and objective overeating (eating an unusually large amount of food without a sense of having lost control over eating). In addition, questions about self-induced vomiting, use of laxatives or diuretics, and intensive exercise to control shape or weight are included. Exercise is defined as intensive when it is “driven” or “compulsive” to control weight, shape or amount of fat, or to burn off calories. The questions about eating disorders cognitions are rated on a 7-point Likert scale from 0–6, where a higher score indicates increased eating disorder symptoms. The EDE-Q consists of one global score and four subscales (restriction and eating-, weight-, and shape concern). Cut-off is usually defined in normal weight populations as mean global EDE-Q score plus one standard deviation which is about 2.5–2.8 depending on different normative samples [25, 27, 28]. Owing to scant evidence supporting the original four-factor subscale structure [29], we report only the global EDE-Q scores as indicator of overall eating disorder cognitive symptoms as proposed by others [30, 31].
Statistics
Data were analyzed using SPSS 22.0 and SAS 9.3. Demographic and clinical characteristics were presented as means and standard deviations (SD) or frequencies and percentages. Prevalence rates for objective and subjective bulimic episodes, objective overeating, self-induced vomiting, and excessive exercise are based on those who answered that such behaviors were present. Logistic regression models with random intercepts were estimated to test whether there were significant differences in occurrences of these behaviors in the two surgery groups throughout the follow-up. Models with fixed effects for time component (up to second order where significant) and dummy identifying surgery group were estimated. Interaction term between the time and dummy was included if significant.
A linear mixed model (LMM) with random effects for intercepts and time was estimated to assess the BMI development after surgery. To account for non-linear trend in time, the fixed effects for time components up to third order were included. Next, a LMM with fixed effects for global EDE-Q score and bulimic episodes at baseline was estimated. Interaction terms between the two and the time were considered but excluded as non-significant. The results were presented graphically as estimated mean BMI with 95 % confidence intervals (CI) at each follow-up point among those with and without objective bulimic episodes preoperatively.
Another LMM with random effects for intercepts and time was estimated to assess the development in global EDE-Q score. The model contained fixed effects for time components up to second order, a dummy identifying surgery group and the interaction term between the dummy and time. The results of this model were presented as estimated mean global EDE-Q scores with 95 % CI at each follow-up point within two surgical groups.
Results
Sixty participants, 42 women and 18 men, were included. Patient demographics at baseline is described in Table 1.
The prevalence of self-reported objective bulimic episodes dropped from 29 % (RYGB) and 32 % (DS) preoperatively to 3 % (RYGB) and 0 % (DS) after 12 months, then increased to 17 % in both groups after 2 years, and at a 5-year follow-up was 22 % in the RYGB group and 7 % in the DS group. The prevalence of subjective bulimic episodes was stable at around 30 % throughout the follow-up period in the RYGB group, while it was 19, 8, and 25 % after 12 months, 2 years, and 5 years, respectively, in the DS group. The prevalence of objective overeating in the GBP group was 42, 24, 7, 17, and 18 % at baseline, 6 months, 12 months, 2 years, and 5 years. Corresponding numbers in the DS group were 48, 14, 4, 17, and 18 %. Self-induced vomiting was reported by one participant in each surgery group at baseline, one participant (RYGB group) at 6 months, and no participants at later time-points. Finally, the prevalence of intense exercise in the RYGB group was 10, 19, 3, 10, and 26 % at baseline, 6 months, 12 months, 2 years, and 5 years. Corresponding numbers in the DS group were 14, 32, 23, 10, and 14 %, respectively. None of the eating disorder behavior symptoms showed significant differences between surgical groups throughout the follow-up.
Mean global EDE-Q score in the RYGB group was 2.6 (SD 0.9) at baseline, 1.5 (SD 0.9) at 12 months, and 2.3 (SD 1.5) after 5 years, while corresponding numbers in the DS group were 2.8 (SD 0.9), 1.4 (SD 1.1), and 1.5 (SD 1.0), respectively. Overall, the score increased throughout follow-up in the RYGB group and remained stable in the DS group (Table 2).
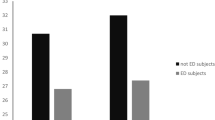
According to the LMM, there was a significant non-linear (third-order) trend in time for BMI for all participants (p < 0.001). Baseline global EDE-Q score was not a significant predictor for change in BMI after surgery (p = 0.599). BMI was not significantly different between those with and without preoperative objective bulimic episodes the first year after surgery. However, the estimated mean BMI was significantly lower in the group with preoperative objective bulimic episodes after 2 years (p = 0.042) and 5 years (p = 0.013) (Fig. 1).
There was a significant second-order time trend in global EDE-Q score as estimated by the linear mixed model. The DS group had a significantly lower estimated global EDE-Q score than the RYGB group after 5 years (p = 0.009) (Fig. 2).
Discussion
Our observations suggest that eating disorder cognitions as measured by global EDE-Q score is not a significant predictor for weight loss after RYGB and DS, whereas patients with objective bulimic episodes before surgery had significantly greater weight loss after 2 and 5 years independently of the type of surgery. We also found that patients in the DS group had a significantly lower global EDE-Q score than the RYGB group at a 5-year follow-up.
The prevalence of self-reported objective bulimic episodes before surgery in our sample was 30 %, while studies in the review article by Meany et al. reported a binge eating prevalence before bariatric surgery ranging from 14 to 55.9 % [8]. In our study, objective bulimic episodes presurgery predicted a greater weight loss after 2- and 5-year follow-up. Recent studies have found unclear or no associations between preoperative eating behaviors such as sweet eating, emotional eating, hyperphagia or gorging, and postoperative weight loss [9, 14], while Konttinen et al. [13] showed that neither preoperative restraint, disinhibition, or hunger measured with TFEQ predicted weight loss postsurgery. Others have found that preoperative restraint [32, 33] and disinhibition [32, 34] was negatively associated with postoperative weight loss. Meanwhile, studies evaluating the specific effect of binge eating on weight loss after bariatric surgery report mixed results. Some studies have demonstrated that preoperative binge eating positively influence weight loss after bariatric surgery [35, 36]. In contrast, others have found a negative effect [17, 37, 38] or no association between postoperative weight loss and binge eating [39–41]. The discrepancy in results may be due to different methods in assessing and defining binge eating and different surgical procedures used.
Subjective bulimic episodes (sense of losing control but normal amount of food) were more prevalent than objective bulimic episodes in the RYGB group at all times of follow-up and at all times except at a 2-year follow-up for the DS group. In a recent cross-sectional study by Conceição et al., looking at loss of control (LOC) eating before bariatric surgery, then at short- and long-term follow-up, objective bulimic episodes were present in only 0.9 %, while subjective bulimic episodes were present in 10.9 % of participants at short-term (<2 years) follow-up [7]. In light of this, researchers have questioned whether binge eating criteria should be re-evaluated when applied to patients after bariatric surgery since many report a lack of control even when the amount of food consumed is small [5], and many patients who reported binge eating before surgery resort to grazing [6]. Another study by Conceição et al. showed that “picking and nibbling” was the most commonly reported maladaptive eating behavior after RYGB and gastric band surgery and was significantly associated with weight regain [42].
To our knowledge, few previous studies have investigated the relationship between global EDE-Q score and weight development after bariatric surgery. Our data showed that both eating disorder behavior and cognitive symptoms seemed to increase from the same turning point as weight [20, 21]. Factors explaining this co-variation in weight development and eating disorder symptoms may well be psychological. Colles et al. showed that recurrence of distorted eating behavior after surgery was associated with elevated psychological distress and a poorer excess weight loss (EWL) [6], while in a study by Thonney et al., EWL and a lower BMI also improved psychological outcomes such as depression, anxiety, and eating disorders 2 years after surgery [43]. A study by Castellini et al. investigated the relationship between bariatric surgery, weight loss, and psychological outcomes before and 12 months after surgery in 83 subjects categorized according to the type of surgery (laparascopic adjustable gastric band, RYGB, and biliopancreatic diversion). They found that while all three groups experienced significant improvements in anxiety, depression, general psychopathology, and binge eating symptomatology, the patients operated with biliopancreatic diversion had the greatest reduction in weight and in binge eating psychopathology, similar to our findings. At a 1-year follow-up, the total EDE-Q score was 1.2 after biliopancreatic diversion and 2.2 after GBS [44], a larger difference than was found in our study. The co-variation in global EDE-Q score and weight corresponds to studies showing that increased BMI in normal populations is positively correlated to global EDE-Q [26].
The reappearance of binge eating after surgery and a subsequent negative influence on weight loss have been demonstrated previously. In an 8-year follow-up study by Kalarchian et al., almost half the patients reported recurrent objective or subjective bulimic episodes, which was associated with a less favorable outcome and greater weight regain [17]. While some claim that extremely obese patients with binge eating should be referred to preoperative behavioral counseling [45, 46], preoperative binge eating disorder is not, according to expert guidelines, considered an absolute contradiction to bariatric surgery [47]. Practitioners are, however, advised to assess the severity and consequences of the condition both pre- and postsurgery [47, 48].
The strengths of this study include a relatively high participation rate, a randomized design, and the longitudinal perspective. The main limitation was a small number of participants in each surgery group. The sample was also predominantly female, which is common in studies reporting on bariatric surgery outcome measures [49]. Another limitation was that the Norwegian and Swedish translated version of the EDE-Q form has not been validated. However, the subsequent version (6.0) which has been validated [25, 26] was almost identical to the one used here. Further, the EDE-Q may not adequately address the difficulties for postbariatric patients in identifying “objectively large” amounts of food [50]. Neither does it identify new maladaptive eating behaviors after surgery, such as grazing. There is thus a need for validated questionnaires in this patient population. Finally, the occurrence of eating disordered behavior are based on self-reported data. Binge eating episodes are complex behavior including an evaluation of amount of food and sense of loss of control and the validity of the results of such behavior in self-reporting in overweight persons is uncertain [51].
Conclusion
Objective bulimic episodes but not global EDE-Q score before surgery predicted increased weight loss after RYGB and DS. The DS group had a significantly lower global EDE-Q score than the RYGB group 5 years after surgery.
References
Beck NN, Mehlsen M, Stoving RK. Psychological characteristics and associations with weight outcomes two years after gastric bypass surgery: postoperative eating disorder symptoms are associated with weight loss outcomes. Eat Behav. 2012;13(4):394–7. doi:10.1016/j.eatbeh.2012.06.001.
Sarwer DB, Dilks RJ, West-Smith L. Dietary intake and eating behavior after bariatric surgery: threats to weight loss maintenance and strategies for success. Surg Obes Relat Dis. 2011;7(5):644–51. doi:10.1016/j.soard.2011.06.016.
Wimmelmann CL, Dela F, Mortensen EL. Psychological predictors of weight loss after bariatric surgery: a review of the recent research. Obes Res Clin Pract. 2014;8(4):e299–313. doi:10.1016/j.orcp.2013.09.003.
Pournaras DJ, Le Roux CW. The effect of bariatric surgery on gut hormones that alter appetite. Diabetes Metab. 2009;35(6 Pt 2):508–12. doi:10.1016/S1262-3636(09)73457-3.
Niego SH, Kofman MD, Weiss JJ, et al. Binge eating in the bariatric surgery population: a review of the literature. Int J Eat Disord. 2007;40(4):349–59. doi:10.1002/eat.20376.
Colles SL, Dixon JB, O'Brien PE. Grazing and loss of control related to eating: two high-risk factors following bariatric surgery. Obesity (Silver Spring). 2008;16(3):615–22. doi:10.1038/oby.2007.101.
Conceicao E, Bastos AP, Brandao I, et al. Loss of control eating and weight outcomes after bariatric surgery: a study with a Portuguese sample. Eat Weight Disord. 2014;19(1):103–9. doi:10.1007/s40519-013-0069-0.
Meany G, Conceicao E, Mitchell JE. Binge eating, binge eating disorder and loss of control eating: effects on weight outcomes after bariatric surgery. Eur Eat Disord Rev. 2014;22(2):87–91. doi:10.1002/erv.2273.
Livhits M, Mercado C, Yermilov I, et al. Preoperative predictors of weight loss following bariatric surgery: systematic review. Obes Surg. 2012;22(1):70–89. doi:10.1007/s11695-011-0472-4.
Burgmer R, Grigutsch K, Zipfel S, et al. The influence of eating behavior and eating pathology on weight loss after gastric restriction operations. Obes Surg. 2005;15(5):684–91. doi:10.1381/0960892053923798.
Fischer S, Chen E, Katterman S, et al. Emotional eating in a morbidly obese bariatric surgery-seeking population. Obes Surg. 2007;17(6):778–84.
Lang T, Hauser R, Buddeberg C, et al. Impact of gastric banding on eating behavior and weight. Obes Surg. 2002;12(1):100–7. doi:10.1381/096089202321144667.
Konttinen H, Peltonen M, Sjostrom L, et al. Psychological aspects of eating behavior as predictors of 10-y weight changes after surgical and conventional treatment of severe obesity: results from the Swedish obese subjects intervention study. Am J Clin Nutr. 2015;101(1):16–24. doi:10.3945/ajcn.114.095182.
Facchiano E, Scaringi S, Quartararo G, et al. Do preoperative eating behaviors influence weight loss after biliopancreatic diversion? Obes Surg. 2013;23(12):2080–5. doi:10.1007/s11695-013-0940-0.
White MA, Kalarchian MA, Masheb RM, et al. Loss of control over eating predicts outcomes in bariatric surgery patients: a prospective, 24-month follow-up study. J Clin Psychiatry. 2010;71(2):175–84. doi:10.4088/JCP.08m04328blu.
Saunders R. “Grazing”: a high-risk behavior. Obes Surg. 2004;14(1):98–102. doi:10.1381/096089204772787374.
Kalarchian MA, Marcus MD, Wilson GT, et al. Binge eating among gastric bypass patients at long-term follow-up. Obes Surg. 2002;12(2):270–5. doi:10.1381/096089202762552494.
Alger-Mayer S, Rosati C, Polimeni JM, et al. Preoperative binge eating status and gastric bypass surgery: a long-term outcome study. Obes Surg. 2009;19(2):139–45. doi:10.1007/s11695-008-9540-9.
Bocchieri-Ricciardi LE, Chen EY, Munoz D, et al. Pre-surgery binge eating status: effect on eating behavior and weight outcome after gastric bypass. Obes Surg. 2006;16(9):1198–204. doi:10.1381/096089206778392194.
Sovik TT, Aasheim ET, Taha O, et al. Weight loss, cardiovascular risk factors, and quality of life after gastric bypass and duodenal switch: a randomized trial. Ann Intern Med. 2011;155(5):281–91. doi:10.7326/0003-4819-155-5-201109060-00005.
Risstad H, Sovik TT, Engstrom M, et al. Five-year outcomes after laparoscopic gastric bypass and laparoscopic duodenal switch in patients with body mass index of 50 to 60: a randomized clinical trial. JAMA Surg. 2015. doi:10.1001/jamasurg.2014.3579.
Aasheim ET, Bjorkman S, Sovik TT, et al. Vitamin status after bariatric surgery: a randomized study of gastric bypass and duodenal switch. Am J Clin Nutr. 2009;90(1):15–22. doi:10.3945/ajcn.2009.27583.
Sovik TT, Taha O, Aasheim ET, et al. Randomized clinical trial of laparoscopic gastric bypass versus laparoscopic duodenal switch for superobesity. Br J Surg. 2010;97(2):160–6. doi:10.1002/bjs.6802.
Berg KC, Peterson CB, Frazier P, et al. Psychometric evaluation of the eating disorder examination and eating disorder examination-questionnaire: a systematic review of the literature. Int J Eat Disord. 2012;45(3):428–38. doi:10.1002/eat.20931.
Ro O, Reas DL, Lask B. Norms for the Eating Disorder Examination Questionnaire among female university students in Norway. Nord J Psychiatry. 2010;64(6):428–32. doi:10.3109/08039481003797235.
Ro O, Reas DL, Rosenvinge J. The impact of age and BMI on Eating Disorder Examination Questionnaire (EDE-Q) scores in a community sample. Eat Behav. 2012;13(2):158–61. doi:10.1016/j.eatbeh.2011.12.001.
Welch E, Birgegard A, Parling T, et al. Eating disorder examination questionnaire and clinical impairment assessment questionnaire: general population and clinical norms for young adult women in Sweden. Behav Res Ther. 2011;49(2):85–91. doi:10.1016/j.brat.2010.10.010.
Mond JM, Hay PJ, Rodgers B, et al. Eating Disorder Examination Questionnaire (EDE-Q): norms for young adult women. Behav Res Ther. 2006;44(1):53–62. doi:10.1016/j.brat.2004.12.003.
Franko DL, Jenkins A, Roehrig JP, et al. Psychometric properties of measures of eating disorder risk in Latina college women. Int J Eat Disord. 2012;45(4):592–6. doi:10.1002/eat.20979.
Becker AE, Thomas JJ, Bainivualiku A, et al. Validity and reliability of a Fijian translation and adaptation of the Eating Disorder Examination Questionnaire. Int J Eat Disord. 2010;43(2):171–8. doi:10.1002/eat.20675.
Fairburn CG, Cooper Z, Doll HA, et al. Transdiagnostic cognitive-behavioral therapy for patients with eating disorders: a two-site trial with 60-week follow-up. Am J Psychiatry. 2009;166(3):311–9. doi:10.1176/appi.ajp.2008.08040608.
Miras AD, Al-Najim W, Jackson SN, et al. Psychological characteristics, eating behavior, and quality of life assessment of obese patients undergoing weight loss interventions. Scand J Surg. 2015;104(1):10–7. doi:10.1177/1457496914543977.
Sarwer DB, Wadden TA, Moore RH, et al. Preoperative eating behavior, postoperative dietary adherence, and weight loss after gastric bypass surgery. Surg Obes Relat Dis. 2008;4(5):640–6. doi:10.1016/j.soard.2008.04.013.
Livhits M, Mercado C, Yermilov I, et al. Patient behaviors associated with weight regain after laparoscopic gastric bypass. Obes Res Clin Pract. 2011;5(3):e169–266. doi:10.1016/j.orcp.2011.03.004.
Latner JD, Wetzler S, Goodman ER, et al. Gastric bypass in a low-income, inner-city population: eating disturbances and weight loss. Obes Res. 2004;12(6):956–61. doi:10.1038/oby.2004.117.
Malone M, Alger-Mayer S. Binge status and quality of life after gastric bypass surgery: a one-year study. Obes Res. 2004;12(3):473–81. doi:10.1038/oby.2004.53.
Mitchell JE, Lancaster KL, Burgard MA, et al. Long-term follow-up of patients’ status after gastric bypass. Obes Surg. 2001;11(4):464–8. doi:10.1381/096089201321209341.
de Zwaan M, Hilbert A, Swan-Kremeier L, et al. Comprehensive interview assessment of eating behavior 18–35 months after gastric bypass surgery for morbid obesity. Surg Obes Relat Dis. 2010;6(1):79–85. doi:10.1016/j.soard.2009.08.011.
Busetto L, Segato G, De Luca M, et al. Weight loss and postoperative complications in morbidly obese patients with binge eating disorder treated by laparoscopic adjustable gastric banding. Obes Surg. 2005;15(2):195–201. doi:10.1381/0960892053268327.
Powers PS, Perez A, Boyd F, et al. Eating pathology before and after bariatric surgery: a prospective study. Int J Eat Disord. 1999;25(3):293–300.
de Man LJ, Ghaderi A, Norring C. Binge eating in surgical weight-loss treatments. Long-term associations with weight loss, health related quality of life (HRQL), and psychopathology. Eat Weight Disord. 2011;16(4):e263–9.
Conceicao E, Mitchell JE, Vaz AR, et al. The presence of maladaptive eating behaviors after bariatric surgery in a cross sectional study: importance of picking or nibbling on weight regain. Eat Behav. 2014;15(4):558–62. doi:10.1016/j.eatbeh.2014.08.010.
Thonney B, Pataky Z, Badel S, et al. The relationship between weight loss and psychosocial functioning among bariatric surgery patients. Am J Surg. 2010;199(2):183–8. doi:10.1016/j.amjsurg.2008.12.028.
Castellini G, Godini L, Amedei SG, et al. Psychological effects and outcome predictors of three bariatric surgery interventions: a 1-year follow-up study. Eat Weight Disord. 2014;19(2):217–24. doi:10.1007/s40519-014-0123-6.
Ashton K, Drerup M, Windover A, et al. Brief, four-session group CBT reduces binge eating behaviors among bariatric surgery candidates. Surg Obes Relat Dis. 2009;5(2):257–62. doi:10.1016/j.soard.2009.01.005.
Rusch MD, Andris D. Maladaptive eating patterns after weight-loss surgery. Nutr Clin Pract. 2007;22(1):41–9.
Mechanick JI, Kushner RF, Sugerman HJ, et al. American Association of Clinical Endocrinologists, The Obesity Society, and American Society for Metabolic & Bariatric Surgery medical guidelines for clinical practice for the perioperative nutritional, metabolic, and nonsurgical support of the bariatric surgery patient. Obesity (Silver Spring). 2009;17 Suppl 1:S1–70. doi:10.1038/oby.2009.28.
Sarwer DB. Comment on: brief, four session group CBT reduces binge eating behaviors among bariatric surgery candidates (Ashton K, et al. 2009;5:257–62). Surg Obes Relat Dis. 2009;5(3):407–8. doi:10.1016/j.soard.2009.02.006.
Mazzeo SE, Saunders R, Mitchell KS. Gender and binge eating among bariatric surgery candidates. Eat Behav. 2006;7(1):47–52. doi:10.1016/j.eatbeh.2005.05.008.
de Zwaan M. Type 1 diabetes mellitus and eating disorders. An over- or underestimated comorbidity? Wien Klin Wochenschr. 2004;116(7–8):215–6.
Bartholome LT, Peterson RE, Raatz SK, et al. A comparison of the accuracy of self-reported intake with measured intake of a laboratory overeating episode in overweight and obese women with and without binge eating disorder. Eur J Nutr. 2013;52(1):193–202. doi:10.1007/s00394-012-0302-z.
Conflict of Interest
Authors Hanvold and Mala report grants from the South-Eastern Norway Regional Health Authority. Authors Morseth, Hanvold, Rø, Risstad, Šaltytė Benth, Engström, Olbers, and Henjum declare that they have no conflict of interest.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Grants
The South-Eastern Norway Regional Health Authority, Oslo University Hospital, and Sahlgrenska University Hospital supported the trial financially. The funding source had no role in the design of the study; collection, analysis, and interpretation of the data; or drafting of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Morseth, M.S., Hanvold, S.E., Rø, Ø. et al. Self-Reported Eating Disorder Symptoms Before and After Gastric Bypass and Duodenal Switch for Super Obesity—a 5-Year Follow-Up Study. OBES SURG 26, 588–594 (2016). https://doi.org/10.1007/s11695-015-1790-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-015-1790-8