Abstract
Background
Bioelectrical impedance analysis (BIA) is a safe and easy method of assessing body composition. Its accuracy to predict fat mass (FM) in obesity and the change in FM following weight loss is questioned. Our objective was to compare leg-to-leg BIA to dual-energy X-ray absorptiometry (DXA) in the assessment of FM in a large population, the changes in FM after Roux-en-Y gastric bypass (RYGB) and to estimate between-method differences (bias) and limits of agreement.
Methods
BIA (Tanita BC-420MA) and DXA (Hologic Discovery W) were used in 5,740 consecutive patients (mean BMI, 37.7 ± 8.2 kg/m2) examined in a clinical nutrition department and in 72 women undergoing RYGB (BMI, 47.2 ± 7.2 kg/m2). Analyses included correlations between methods and Bland Altman analysis.
Results
In the entire population, BIA significantly overestimated FM in comparison with DXA (1.1 ± 6.1 kg, 0.8 ± 5.6%). FM estimates by each method were significantly correlated in absolute value (kg; r 2 = 0.9 in the whole population), and in percentage (r 2 = 0.6). However, wide limits of agreement were observed. In surgery patients, BIA significantly overestimated FM both before and 12 months after bypass. BIA significantly overestimated changes in FM after RYGB at 3 months (2.9 ± 5.0 kg) and at 12 months (1.9 ± 3.9 kg) but not at 6 months (0.9 ± 5.0 kg; p = 0.08). Estimates of changes in FM by each method were significantly correlated (r 2 = 0.4, 0.6, and 0.9, respectively).
Conclusion
According to the wide limits of agreement, BIA seems more interesting for epidemiological rather than individual use to evaluate body FM and FM changes in obese women undergoing RYGB.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Assessment of body composition in obese subjects and assessment of changes in body composition following weight loss, especially massive surgery-induced weight loss, remain challenging tasks [1–6].
Body composition methods that are currently available differ in terms of precision, cost, complexity, and patient acceptability [5]. Dual-energy X-ray absorptiometry (DXA) is increasingly considered as a reference method for assessment of body composition in various settings [7]. There is also some evidence that DXA accurately assesses changes in body composition in obese subjects following weight loss intervention [1, 3, 6]. However, major disadvantages of this method include the cost of equipment and the irradiation, which makes its use difficult for large population studies, clinical practice and repeated measures. Bioelectrical impedance analysis (BIA) is a rapid, easy, and safe method of assessing body composition but is criticized for being inaccurate in comparison with reference measures, especially when applied to obese subjects [4, 5, 8, 9]. Many interference effects may limit its validity: body geometry, underestimation of resistance and reactance when thighs are in contact, and significant fluid abnormalities [4, 5, 8, 9]. For these reasons, further validation of BIA in subjects at extremes of BMI ranges is required.
However during weight loss, BIA better predicts FM than BMI and very few studies have examined the interest in BIA in the longitudinal follow-up of patients after bariatric surgery [2, 4, 6].
The aim of the study was to compare a recently developed leg-to-leg BIA device (Tanita BC-420MA) and DXA as a criterion method in assessing FM in a large population of patients seen in a nutrition department, including a majority of overweight and obese patients, and in assessing changes in FM during massive weight loss following Roux-en-Y gastric Bypass (RYGB).
Subjects and Methods
Subjects
Between July 2007 and September 2009, 5,740 patients were successively seen in the Nutrition Department of La Pitié Salpêtrière Hospital (Paris, France) for the assessment of body composition (mean age, 44.5 ± 13.4 years; mean BMI, 37.7 ± 8.2 kg/m2).
Among this population were 72 women (mean age, 39.0 ± 10.7 years; mean BMI, 44.8 ± 4.5 kg/m2) who were admitted for surgical treatment (RYGB) of obesity and underwent repeated measurements of body composition 3 (n = 72), 6 (n = 72) and 12 (n = 41) months after RYGB. Suitability for RYGB was assessed by a multidisciplinary team following standard guidelines for the management of obese patients [10]. The same surgical procedure was performed laparoscopically by one surgeon in all patients, creating a loop length of about 1 m [11]. The follow-up study protocol was approved by the Ethics Committee of Hotel Dieu Hospital with subjects signing informed written consent.
Body Composition Measurements
Anthropometric and body composition assessment was performed by two investigators. For each patient, these were performed on the same day and by the same investigator. Subjects had not eaten for the preceding 12 h and rested for 20 min before BIA.
Anthropometric Data
Height was measured to the nearest 5 mm, without shoes, using a wall-mounted stadiometer. Weight was measured to the nearest 0.1 kg using the in-built BIA as a weight scale, and BMI was calculated as the weight in kilograms divided by the square of height in meters (kg/m2).
Analysis of Body Fat Mass
Tanita BC-420MA
During the last decade, new single-frequency (50 kHz) leg-to-leg BIA systems have been developed that combine a digital scale with stainless steel pressure-contact foot-pad electrodes for standing impedance and body weight measurements [12].
The Tanita BC-420MA leg-to-leg BIA device looks like a traditional bathroom scale and may be used for subjects up to a weight of 270 kg (http://www.tanita.co.uk/index.php?id=102&tx_f03showxtcommerceproducts_pi1[showUid]=602&cHash=b55c3a06d4, “last accessed 2 September 2010”). The electrodes on the metal foot plates provide a small alternating voltage of 90 μA (50 kHz). Body composition estimates are derived from total body water making use of proprietary equations not supplied by the manufacturer, based on segmental resistance index.
Dual-Energy X-ray Absorptiometry
Body composition was estimated by whole-body DXA scanning (Hologic Discovery W, software v12.6, 2; Hologic Bedford MA), as previously described [1]. For each pixel, the equipment calculates weight, bone mineral content, and FM, and it integrates the measurements for the whole body and different regions. Variables from DXA used in the analysis were FM in kilogram and in percentage of weight. We calculated FM in kilogram as the product of FM in percentage assessed by DXA and the weight in kg as measured by the weight scale.
Statistical Analysis
All data were reported as means ± SD. Analyses were performed separately by gender. Paired t tests and correlation coefficients were used for the comparison of body FM in the entire population, and FM loss in women undergoing RYGB assessed by BIA and DXA. Inter-method differences (bias) and level of agreement at individual level was calculated as the mean difference between methods ±1.96 SD by using the Bland and Altman analysis [13]. p Values <0.05 were considered significant. Statistical analysis was performed using Statview v4.0 (SAS Institute, Cary, N.C., USA).
Results
Total Population
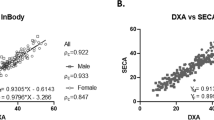
More than 80% of the patients in our study are defined as obese, with the mean BMI being 37.7 kg/m2. Table 1 shows body composition data by DXA and BIA, mean differences and correlation coefficients for body weight and FM (kg and %) between methods in the total population under study. As expected, body composition differed significantly by gender. BIA significantly overestimated FM (kg and %) in comparison with DXA both in women (1.3 ± 5.3 kg and 1.0 ± 5.2%) and men (0.4 ± 8.1 kg and 0.2 ± 6.7%). The regression correlation coefficient was high in both women (r 2 = 0.9 for FM in kilograms and 0.5 for FM in percentage) and men (r 2 = 0.8 for FM in kilograms and 0.5 for FM in percentage).
The Bland Altman plots are represented in Fig. 1. The BIA overestimated FM in kilograms with increasing mean FM ((DXA + BIA)/2) in women but not in men. Limits of agreement were wide (−14.2; +10.6), and higher in men (–17.3; +15.1) than in women (−12.9; +8.7).
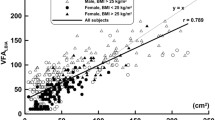
The mean difference in FM in kilograms between DXA and BIA was +0.5 (3.8) kg, −0.9 (4.8) kg, −2.5 (5.7), and −2.9 (6.9) kg in women with BMI < 30 (n = 723), BMI = 30–40 (n = 1,940), BMI = 40–50 (n = 1,216), and BMI > 50 kg/m2 (n = 407), respectively. The mean difference in FM in kilograms between the BIA and DXA was −0.5 (5) kg, −+0.1 (6.9) kg, +1.9 (10.5), and +0.2 (13.3) kg in men with BMI < 30 (n = 294), BMI = 30–40 (n = 742), BMI = 40–50 kg/m2 (n = 352), and BMI > 50 kg/m2 (n = 67), respectively (Fig. 2).
Women Undergoing RYGB
Seventy-two women were candidates for RYGB (BMI 47.2 ± 7.2 kg/m2 and 41.9 ± 11.2 years).
Table 2 shows the changes in weight, BMI, mean FM ((DXA + BIA)/2) in kilograms, and FM assessed by DXA and BIA at 3, 6, and 12 months after RYGB, and the mean differences and correlation coefficients for estimating FM and FM changes. BIA significantly overestimated FM both before and after weight loss. Despite a lower difference at 3 months, the overestimation of FM by BIA seemed to decrease with weight loss. Regression coefficients for FM increased both after surgery and weight loss.
The values of FM change were highly correlated between BIA and DXA, especially at 12 months (r 2 = 0.9). The differences between BIA and DXA in FM change assessment represented 19.4%, 4%, and 6% of the mean FM change at 3, 6, and 12 months, respectively.
The Bland Altman plot corresponding to the assessment of FM changes during weight loss, both by DXA and BIA, revealed an overestimation of FM changes by BIA with increasing mean FM change whatever the period (data not shown). The limits of agreement for the evaluation of FM changes were wide (−12.9; +7.1), (−9.9; +8.1), (−9.7; +5.9) kg at 3, 6, and 12 months, respectively.
Discussion
BIA is potentially a useful and inexpensive tool for the follow-up of obese patients, including those undergoing RYGB. The purpose of our study was to determine if a simple leg-to-leg BIA is a good alternative assessment method to DXA in this group.
Our population was larger than in previous studies comparing the assessment of FM by BIA and DXA [14]. BIA assessment of FM was highly correlated with DXA, as indicated by the high correlation coefficients, in agreement with all previous studies (r 2 > 0.9) [14–23], especially in the most important study until now (n = 591 subjects, mean BMI = 26.3 kg/m2) [14]. Although BIA overestimated FM in comparison with DXA, we observed smaller bias using the leg-to-leg BIA device than in large healthy population where a multifrequency bioelectrical impedance was used (1.8 ± 4.1% for all subjects, 2.6 ± 4.5% for men and 1.7 ± 4.0% for women) [14]. The difference in the assessment of FM usually became larger as mean FM increased, and then we would have expected to have observed more bias in our study. Conversely, the leg-to-leg BIA used in our study provided a reasonable estimation of FM especially in the population of patients with a BMI under 40 kg/m2. However in extremely obese women (Table 2), leg-to-leg BIA provided higher bias than those reported by Das et al. using a four bioelectrical analyzer (with the Lukaski or Segal equations) in comparison with a four-compartmental method (n = 20 women, mean BMI = 47.8 kg/m2)[4].
Both over and underestimation of FM with BIA in comparison to DXA have been reported in previous studies, due to the confounding effects of multiples variables such as sex, age, ethnicity of the population, the BIA device, and its equation [14–24]. In the entire population, BIA overestimated FM in comparison with DXA in both sexes. However as mean FM increased, BIA underestimated FM in men with BMI over 30 kg/m2. Higher fat-free mass and increased hydratation of the legs in men may lead to underestimation of FM [25–28]. The equation used by the manufacturer has probably been developed to compensate for confounding factors in obesity according to sex, but the BIA still seems to be less accurate with increasing weight and FM.
Despite relatively small between-method differences in estimating FM wide limits of agreement were observed that may limit the interest of the BIA method at individual level. These limits were however quite comparable to those reported by Das et al. in healthy subjects. Despite this difference being statistically significant, it may not be clinically meaningful regarding the degree of obesity and fatness in our population.
This was the first study to examine the ability of BIA to assess the loss of FM in comparison with DXA at different intervals following RYGB, before and at 3, 6, and 12 months after surgery.
Close agreement between BIA and DXA in the assessment of FM changes after RYGB were observed and increased with weight reduction, as one would expect from previous studies on a non-obese population. In addition, the ability of BIA to measure FM and FM change was higher after weight loss, in agreement with better validity of BIA in lower weight populations (BMI = 30–40 kg/m2; Fig. 2) [2, 4, 6]. Savastano et al. recently reported the same observation in a group of obese women treated by laparoscopic adjustable gastric banding, undergoing DEXA and BIA before and at 6 and 12 months after surgery [6].
Hence, we did not find any significant bias in FM loss assessment between DXA and BIA at 6 months, when the patients have lower BMI (mean = 35.2 ± 6.2 kg/m2). Fourteen months after RYGB, Das et al. observed lower bias in the assessment of changes in FM using the Lukaski equation (−0.4 ± 1.1%) but not the Segal equation (−6.1 ± 1.1%), a demonstration of the role of the equation as used by the manufacturer in BIA validity.
However, despite lower bias and a better agreement after weight loss, this body of data is not sufficient to prove the equivalence of the two methods for assessment of FM changes. The analysis of the Bland Altman plots and the large limits of agreement demonstrate the presence of systematic errors, whatever the period following weight loss in an obese population, even though it tend to reduce after weight loss (Fig. 1) [2, 4, 6, 14–24]. Das et al. reported similar limits of agreement 14 months after RYGB, although they used different equations and a three compartment model as their reference method [4]. Then BIA and DXA have a limited concordance, and BIA may be used as a surrogate for the latter only in an epidemiological setting, and not in clinical practice.
The use of DXA as a criterion method may be open to criticism, as variations in fat-free mass hydration are suspected by other authors as being responsible for error [7, 29–32]. In addition, beam hardening errors for DXA systems may be considerable in obesity [33]. However, DXA represents to date the only reasonable method to assess the validity of a novel body composition assessment method in large populations, especially in an obese population. In addition, LaForgia et al. did not find any bias in the percentage of FM when assessed by DXA (Lunar prodigy scanner) in comparison to a 4 C model, in a population of obese patients. They proposed that DXA may indeed be used to provide reasonable FM percentage estimates in obese adult cohorts (n = 14; BMI, 33.7 ± 3.5 kg/m2) [7].
In summary, our results suggest that the commercially available leg-to-leg Tanita BC420MA represents a simple non-invasive method for the assessment of FM in a population of patients referred for the evaluation of body composition, especially in obese patients, and in the assessment of the loss of FM in women undergoing RYGB. According to the wide limits of agreement, BIA may be more appropriate for epidemiological rather than individual use.
Abbreviations
- BIA:
-
Bioelectrical impedance analysis
- DXA:
-
Dual-energy X-ray absorptiometry
- RYGB:
-
Roux-en-Y gastric bypass
- BMI:
-
Body mass index
- FFM:
-
Fat-free mass
- FM:
-
Fat mass
- kg:
-
Kilogram
- g:
-
Gram
- TBW:
-
Total body water
References
Ciangura C, Bouillot JL, Lloret-Linares C, et al. Dynamics of change in total and regional body composition after gastric bypass in obese patients. Obesity (Silver Spring). 2009;18(4):760–5.
Cox-Reijven PL, van Kreel B, Soeters PB. Accuracy of bioelectrical impedance spectroscopy in measuring changes in body composition during severe weight loss. JPEN J Parenter Enteral Nutr. 2002;26:120–7.
Das SK. Body composition measurement in severe obesity. Curr Opin Clin Nutr Metab Care. 2005;8:602–6.
Das SK, Roberts SB, Kehayias JJ, et al. Body composition assessment in extreme obesity and after massive weight loss induced by gastric bypass surgery. Am J Physiol Endocrinol Metab. 2003;284:E1080–8.
Lee SY, Gallagher D. Assessment methods in human body composition. Curr Opin Clin Nutr Metab Care. 2008;11:566–72.
Savastano S, Belfiore A, Di Somma C, et al. Validity of bioelectrical impedance analysis to estimate body composition changes after bariatric surgery in premenopausal morbidly women. Obes Surg. 2009;20:332–9.
LaForgia J, Dollman J, Dale MJ, et al. Validation of DXA body composition estimates in obese men and women. Obesity (Silver Spring). 2009;17:821–6.
Kyle UG, Bosaeus I, De Lorenzo AD, et al. Bioelectrical impedance analysis-part II: utilization in clinical practice. Clin Nutr. 2004;23:1430–53.
Kyle UG, Bosaeus I, De Lorenzo AD, et al. Bioelectrical impedance analysis—part I: review of principles and methods. Clin Nutr. 2004;23:1226–43.
Fried M, Hainer V, Basdevant A, et al. Inter-disciplinary European guidelines on surgery of severe obesity. Int J Obes (Lond). 2007;31:569–77.
Suter M, Giusti V, Heraief E, et al. Laparoscopic Roux-en-Y gastric bypass: initial 2-year experience. Surg Endosc. 2003;17:603–9.
Nunez C, Gallagher D, Visser M, et al. Bioimpedance analysis: evaluation of leg-to-leg system based on pressure contact footpad electrodes. Med Sci Sports Exerc. 1997;29:524–31.
Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet. 1986;1:307–10.
Sun G, French CR, Martin GR, et al. Comparison of multifrequency bioelectrical impedance analysis with dual-energy X-ray absorptiometry for assessment of percentage body fat in a large, healthy population. Am J Clin Nutr. 2005;81:74–8.
Andreoli A, Melchiorri G, De Lorenzo A, et al. Bioelectrical impedance measures in different position and vs dual-energy X-ray absorptiometry (DXA). J Sports Med Phys Fitness. 2002;42:186–9.
Evans EM, Saunders MJ, Spano MA, et al. Body-composition changes with diet and exercise in obese women: a comparison of estimates from clinical methods and a 4-component model. Am J Clin Nutr. 1999;70:5–12.
Frisard MI, Greenway FL, Delany JP. Comparison of methods to assess body composition changes during a period of weight loss. Obes Res. 2005;13:845–54.
Kim HJ, Gallagher D, Song MY. Comparison of body composition methods during weight loss in obese women using herbal formula. Am J Chin Med. 2005;33:851–8.
Neovius M, Hemmingsson E, Freyschuss B, et al. Bioelectrical impedance underestimates total and truncal fatness in abdominally obese women. Obesity (Silver Spring). 2006;14:1731–8.
Pateyjohns IR, Brinkworth GD, Buckley JD, et al. Comparison of three bioelectrical impedance methods with DXA in overweight and obese men. Obesity (Silver Spring). 2006;14:2064–70.
Thomson R, Brinkworth GD, Buckley JD, et al. Good agreement >between bioelectrical impedance and dual-energy X-ray absorptiometry for estimating changes in body composition during weight loss in overweight young women. Clin Nutr. 2007;26:771–7.
Utter AC, Nieman DC, Ward AN, et al. Use of the leg-to-leg bioelectrical impedance method in assessing body-composition change in obese women. Am J Clin Nutr. 1999;69:603–7.
Varady KA, Santosa S, Jones PJ. Validation of hand-held bioelectrical impedance analysis with magnetic resonance imaging for the assessment of body composition in overweight women. Am J Hum Biol. 2007;19:429–33.
Newton Jr RL, Alfonso A, York-Crowe E, et al. Comparison of body composition methods in obese African-American women. Obesity (Silver Spring). 2006;14:415–22.
Bray GA, DeLany JP, Harsha DW, et al. Evaluation of body fat in fatter and leaner 10-y-old African American and white children: the Baton Rouge Children’s Study. Am J Clin Nutr. 2001;73:687–702.
Coppini LZ, Waitzberg DL, Campos AC. Limitations and validation of bioelectrical impedance analysis in morbidly obese patients. Curr Opin Clin Nutr Metab Care. 2005;8:329–32.
Deurenberg P, Weststrate JA, Hautvast JG. Changes in fat-free mass during weight loss measured by bioelectrical impedance and by densitometry. Am J Clin Nutr. 1989;49:33–6.
Withers RT, LaForgia J, Pillans RK, et al. Comparisons of two-, three-, and four-compartment models of body composition analysis in men and women. J Appl Physiol. 1998;85:238–45.
Friedl KE, DeLuca JP, Marchitelli LJ, et al. Reliability of body-fat estimations from a four-compartment model by using density, body water, and bone mineral measurements. Am J Clin Nutr. 1992;55:764–70.
Fuller NJ, Jebb SA, Laskey MA, et al. Four-component model for the assessment of body composition in humans: comparison with alternative methods, and evaluation of the density and hydration of fat-free mass. Clin Sci (Lond). 1992;82:687–93.
Pietrobelli A, Wang Z, Formica C, et al. Dual-energy X-ray absorptiometry: fat estimation errors due to variation in soft tissue hydration. Am J Physiol. 1998;274:E808–16.
Deurenberg-Yap M, Schmidt G, van Staveren WA, et al. Body fat measurement among Singaporean Chinese, Malays and Indians: a comparative study using a four-compartment model and different two-compartment models. Br J Nutr. 2001;85:491–8.
Goodsitt MM. Evaluation of a new set of calibration standards for the measurement of fat content via DPA and DXA. Med Phys. 1992;19:35–44.
Acknowledgements
Thanks are expressed to Sophie Festis and Marianne Merlet, both at the Department of Nutrition, Pitié Salpêtrière Hospital, Paris, France, for expert technical assistance with DXA measurements. The authors are grateful to Dr. Andrew Green, general practitioner in Cheltenham (UK), for reviewing the English.
Conflict of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lloret Linares, C., Ciangura, C., Bouillot, JL. et al. Validity of Leg-to-Leg Bioelectrical Impedance Analysis to Estimate Body Fat in Obesity. OBES SURG 21, 917–923 (2011). https://doi.org/10.1007/s11695-010-0296-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-010-0296-7