Abstract
Background
Roux-en-Y gastric bypass (RYGBP) has become a common surgical procedure to treat morbid obesity. Furthermore, it strongly reduces the incidence of type 2 diabetes and mortality. However, there is scant information on how magnesium status is affected by RYGBP surgery. Previous bariatric surgery methods, like jejunoileal bypass, are associated with hypomagnesemia.
Methods
Twenty-one non-diabetic morbidly obese patients who underwent RYGBP were evaluated before and 1 year after surgery and compared to a matched morbidly obese control group regarding serum magnesium. Groups were matched regarding weight, BMI, abdominal sagittal diameter and fasting glucose, blood pressure, and serum magnesium concentrations before surgery in the RYGBP group.
Results
The serum magnesium concentrations increased by 6% from 0.80 to 0.85 mmol/l (p = 0.019) in the RYGBP group while a decrease by 4% (p = 0.132) was observed in the control group. The increase in magnesium concentration at the 1-year follow-up in the RYGBP group was accompanied by a decreased abdominal sagittal diameter (r 2 = 0.32, p = 0.009), a lowered BMI (r 2 = 0.28, p = 0.0214), a lowered glucose concentration (r 2 = 0.28, p = 0.027) but not by a lowered insulin concentration (p = 0.242), a lowered systolic (p = 0.789) or a lowered diastolic (p = 0.785) blood pressure.
Conclusion
RYGBP surgery in morbidly obese subjects is characterized by reduced visceral adiposity, lowered plasma glucose, and increased circulating magnesium concentrations. The inverse association between lowered central obesity, lowered plasma glucose and increased magnesium concentrations, needs further detailed studies to identify underlying mechanisms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obesity has become a global epidemic, and more than 400 million individuals worldwide are obese [1]. Obesity is not only a burden per se, but also tightly connected to diabetes and cardiovascular diseases. Treatment for obesity includes a range of therapies, such as dietary advice and physical activity, behavior therapy, pharmacological therapy, and bariatric surgery.
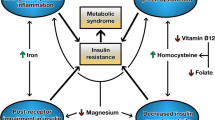
Bariatric surgery by the Roux-en-Y gastric bypass (RYGBP) is now a frequently used treatment for obesity and has been shown not only to induce rapid weight loss, followed by long-term weight stability, but also to reduce the risk of developing diabetes, cardiovascular diseases (CVD), and cancer [2–4]. Patients with insulin resistant conditions like type-2 diabetes and obesity have lower circulating magnesium concentrations than healthy subjects [5, 6]. Recently, a low serum magnesium concentration was shown to increase the risk of all-cause mortality when added to the conventional CVD risk factors and was significantly associated with all-cause mortality in type 2 diabetes patients [7]. A previous weight-reducing surgical method, the jejunoileal bypass (JIB) was associated with an increased risk of magnesium depletion [8]. Regarding RYGBP in morbidly obese patients, there is only scant information about changes, if any, in circulating magnesium concentrations [9].
The aim of the present study was to investigate possible alterations in magnesium status 1 year after RYGBP in morbidly obese patients as compared to a matched control group of morbidly obese patients.
Patients and Methods
Patients
Twenty-one patients (3 men, 18 women), all Caucasians, free from established diabetes, and not on pharmacological treatment for hypertension, were recruited from the Outpatient Clinic of Obesity Care, Uppsala University Hospital, Uppsala, Sweden. All the patients were recruited in a consecutive order. The patients were investigated before and 1 year after RYGBP. Data from the RYGBP group were compared to that of a control group, recruited from the waiting list for RYGBP, consisting of 21 obese patients, five men and 16 women, all free from established diabetes and not on pharmacological treatment for hypertension. The control group was recruited to match weight, body mass index (BMI), abdominal sagittal diameter, fasting glucose, blood pressure and serum magnesium in relation to the corresponding baseline values in the group of morbidly obese patients who underwent RYGBP. Baseline characteristics of the subjects are shown in Table 1. The study protocol was approved by the local ethics committee at the Faculty of Medicine at Uppsala University. All patients gave informed consent.
The RYGBP Operation
RYGBP excluded the stomach and duodenum from the passage of food [11]. The flaccid part of the lesser omentum and the first gastric vessel on the lesser curvature was divided just below the fat pad, to create a small gastric pouch (2 × 3 cm). The pouch was then totally separated from the main stomach, which was left in the abdomen. The small bowel was divided 30 cm distal to the ligament of Treitz, and the aboral end was connected to the small gastric pouch. This jejunal limb, the so-called Roux limb, was made 70 cm long and placed behind the excluded stomach and transverse colon. The small bowel continuity was maintained by an entero-enterostomy between the Roux limb and the earlier divided proximal jejunum. This created the Y-shaped junction where the ingested food, via the Roux-limb, and the gastric acid and bile are mixed.
All patients treated by surgical intervention were given the same kind of dietary advice and were recommended to take a daily oral supplement containing vitamins and minerals but not magnesium.
Body Mass Index
BMI (kg/m2) was calculated as weight (kg) divided by height (m) squared. Abdominal sagittal diameter, a variable reflecting visceral adipose tissue, was recorded at the umbilical level as the height (cm) of the abdomen measured from the examination couch.
Blood Pressure and Pulse Rate
Systolic blood pressure (SBP) and diastolic blood pressure (DBP) were measured using a sphygmomanometer, with a cuff of appropriate size, twice in the supine position after a rest of 5 min. The mean of each of these sets of values was used in the analyses.
Routine Laboratory Tests
The following analyses were all carried out, using standard routine methods, at the Department of Clinical Chemistry, Uppsala University Hospital: plasma glucose, glycosylated hemoglobin (HbA1c), and insulin, sodium, potassium, albumin, and creatinine concentrations in serum. The serum magnesium was measured by spectrophotometric determination in serum with xylidyl blue (Architect, Abbott). The coefficient of variation is <2% for this method. The plasma aldosterone concentration was assayed by a commercial radioimmunoassay kit (Aldosterone-RIA, DPC Diagnostic Products, Los Angeles, CA, USA). Insulin sensitivity was assessed by calculating the HOMA-IR index [(fasting serum glucose × fasting serum insulin)/22.5] [10].
Statistics
All analyses were defined a priori. The results were given as arithmetic mean with SD and SEM. ANOVA was used for group comparisons. Adjusted analyses were made using ANCOVA. Baseline associations between continuous variables were analysed using Pearson product moment correlation coefficients. Tests were two-tailed and a p value <0.05 was considered significant. Statistical software JMP 3.0 for PC (SAS Corporation, Cary, TX, USA) was used for all statistical calculations.
Results
Baseline Data
At baseline, i.e., before patients underwent RYGBP surgery, there were no statistically significant differences between the group of patients directed for surgical treatment and the control group, regarding weight, BMI, abdominal sagittal diameter, fasting plasma glucose and serum insulin concentrations, systolic and diastolic blood pressure, serum aldosterone or serum magnesium concentrations; however, the control group was younger (Table 1). At baseline, an inverse correlation was observed between BMI and serum magnesium (r = −0.30, p = 0.03).The correlations between serum magnesium concentrations on the one hand, and abdominal sagittal diameter or glucose concentrations on the other, were not statistically significant (r = −0.27, p = 0.10; r = −0.24, p = 0.08, respectively). None of the patients in this study had any complications during the surgical performance or during the 1-year follow-up period.
Data at 1-year Follow-up
Serum magnesium concentration increased by 6% in the RYGBP-treated group, from 0.80 to 0.85 mmol/l (p = 0.009), while an opposite trend was observed during the corresponding period in the control group by 4%, from 0.80 to 0.77 mmol/l (p = 0.132). The intergroup difference in serum magnesium concentrations at the 1-year follow-up (10%, p = 0.014) was significant (Fig. 1).
In the RYGBP group, the mean BMI decreased from 42.3 kg/m2 at baseline to 29.7 kg/m2, i.e., by 30% (p < 0.001). In the control group, no change in mean BMI was observed between baseline and 1-year follow-up, BMI 44.3 kg/m2 and 44.2 kg/m2, respectively. Abdominal sagittal diameter decreased from 28.9 cm at baseline to 22.6 cm, i.e., by 22% in the RYGBP group (p < 0.001), while no change was observed in the control group (30.0 cm at baseline as well as at the 1-year follow-up). BMI and abdominal sagittal diameter were significantly different in the two groups at the 1-year follow-up (both p < 0.001).
Fasting plasma glucose concentrations decreased by 15%, from 4.9 mmol/l before RYGBP surgery to 4.2 mmol/l at the 1-year follow-up (p < 0.001) and increased by 6% from 5.1 to 5.4 mmol/l, (ns) in the control group. The intergroup difference regarding fasting plasma glucose at the 1-year follow-up was statistically significant (p < 0.001).
The fasting plasma insulin concentrations decreased by 75%, from 83.4 pmol/l before RYGBP surgery to 24.6 pmol/l at the 1-year follow-up after RYGBP surgery (p < 0.001). In the control group, the corresponding figures were 103.2 pmol/l at baseline and 112.8 pmol/l at the 1-year follow-up (ns). The intergroup difference in insulin concentration, at the 1-year follow-up, was statistically significant (p < 0.001; Fig. 2).
The changes in percent from baseline to follow-up at 1 year after RYGBP in serum magnesium (s-Mg), body mass index (BMI), abdominal sagittal diameter (ASD), fasting plasma glucose (fP-glucose), fasting serum insulin (fS-insulin), Homeostatic model assessment (HOMA-IR), systolic (SBP) and diastolic (DBP) blood pressures. Statistical significance indicated by *p < 0.05, **p < 0.01, and ***p < 0.001
Insulin resistance assessed by HOMA-IR index decreased by 76%, from 3.1 to 0.75 in the GBP group (p < 0.001), while an opposite non-significant alteration from 4.1 to 4.9 was observed in the control group, implying a significant intergroup difference at the 1-year follow-up (p < 0.001; Fig. 2).
SBP decreased in the RYGBP group by 7%, from 123 to 114 mmHg (p = 0.05). No significant change was observed in the control group (121 mmHg at baseline and 123 mmHg at the 1-year follow-up, p = 0.20). DBP showed a similar pattern, decreasing in the RYGBP group by 7%, from 75 to 70 mmHg (p = 0.045). An opposite trend was observed in the control group (76 mmHg at baseline and 79 mmHg at the 1-year follow-up, p = 0.06). The intergroup differences regarding SBP and DBP at the 1-year follow-up were statistically significant (p = 0.019, p < 0.001, respectively; Fig. 2). Serum aldosterone did not change between baseline and 1-year follow-up in either group (data not shown).
Data at 1-year Follow-up: Associations Within the RYGBP Group
The increase in serum magnesium concentration at the 1-year follow-up in the RYGBP group was accompanied by a decreased abdominal sagittal diameter (r 2 = 0.32, p = 0.009), a lowered BMI (r 2 = 0.28, p = 0.021), a lowered glucose concentration (r 2 = 0.28, p = 0.027), and the lowered HOMA-IR index (r 2 = 0.24, p = 0.002), but not by a lowered insulin concentration (p = 0.242), a lowered SBP (p = 0.789) or a lowered DBP (p = 0.785).
Discussion
In the present study, we observed that the serum magnesium concentration increased significantly by 6% during the first year after RYGBP surgery. The increase in serum magnesium was associated with lowered central obesity and reduced fasting glucose concentrations. The previous information about changes in magnesium status after RYGBP in obesity is scarce. In a pilot study, Van Gaal et al. [12] reported a similar but non-significant increase in serum magnesium concentrations in eight obese patients who underwent a horizontal bypass similar to Alden’s technique using the Roux-en-Y anastomosis. Diniz et al. [13] who studied the impact of bariatric surgery on bone metabolism in 110 patients treated with RYGBP by the Capella technique observed elevated parathormone concentrations in one third of the patients; however, none of the patients showed hypomagnesemia. In a previous study on mineral status after bariatric surgery, Goode et al. [14] reported normal serum magnesium levels in a group of patients previously subjected to RYGBP surgery (with a range 3 to 5 years after surgery) as well as in a BMI-matched control group, which may imply that the magnesium increment might be associated to the change in BMI per se rather than the method of treatment. However, different methods of bariatric surgery might have different effects on the circulating magnesium status because hypomagnesemia frequently has been reported in patients treated with jejuno-ileal bypass although with a similar weight reduction as in the RYGBP-treated subjects in the present study. Different methodological aspects such as altered magnesium absorption or induced side effects like diarrhea might help to explain such differences between different surgical methods. The diet regime after RYGBP is characterized by a limited calorie intake but with a high content of nutrients; however, the diet is not supplemented with magnesium. Because no dietary registration was carried out in the present study, the possible influence of diet on magnesium concentrations remains open. The control group was significantly younger than the RYGBP group, and the possible influence of age on glucose metabolism, that is known to deteriorate with higher age, would have driven results towards the null hypothesis, thus possibly underestimating differences observed rather than overestimating them.
The abdominal obesity in the present study assessed by the sagittal diameter declined, the circulating glucose and insulin concentrations decreased, and the blood pressure was lowered in the RYGBP group. The reduced degree of central obesity and lowered glucose concentrations were related to the increased serum magnesium concentrations at follow-up, whereas reduced insulin concentrations and blood pressure levels were not. These findings are congruent with previous data on the association between magnesium status and obesity and different body-weight-related glucometabolic variables, indicating that the increased serum magnesium concentration parallel to weight loss after RYGBP surgery might be explained by the improved glucometabolic status in these patients. The latter is also supported by the HOMA-IR which declines along with increased serum magnesium; although no statistical association was indicated between serum magnesium and insulin concentrations at follow-up, implying that lowering of glucose concentrations mainly underlie the association between HOMA-IR and magnesium.
The serum magnesium concentrations have been shown to be inversely related to circulating glucose concentrations and to insulin resistance in diabetic patients as well as in non-diabetic patients [15–17]. Huerta et al. recently reported lower serum magnesium concentrations in obese non-diabetic children and furthermore that lowered circulating magnesium concentrations were associated with increased insulin concentrations and impaired insulin sensitivity [18]. The association between circulating magnesium concentrations and glycemia has been investigated in studies by Djurhuus et al. who managed to lower plasma glucose concentrations by 20% in type 2 diabetic patients by intensified insulin treatment [19]. Similar findings have been reported by Schnack et al. [20]. As insulin may increase the renal magnesium excretion, the pronounced decrease in circulating insulin concentrations observed after RYGBP surgery in the present study, might contribute to the increased serum magnesium concentrations; however, renal magnesium excretion was not measured in this study.
It may also be speculated whether increased serum magnesium concentration might contribute to the improvements observed in several of the metabolic variables because supplementation with magnesium has been reported to improve insulin sensitivity [21]; however, such findings have not been reported consistently in other trials [22, 23].
Regarding blood pressure, magnesium has been suggested to have calcium antagonistic effects, reducing blood pressure. Widman et al. [24] reported a dose-dependent reduction in blood pressure by administration of magnesium. The serum magnesium increment in that study was similar to what was observed in the RYGBP-treated group in the present study; however, statistically, we could not observe an association between blood pressure and serum magnesium concentrations.
In summary, morbidly obese patients who underwent RYGBP surgery are characterized by reduced abdominal sagittal diameter representing reduced visceral adiposity, lowered plasma glucose, and increased circulating magnesium concentrations. These findings, reflecting the inverse association between lowered central obesity and improved magnesium status, need further more detailed studies to identify the exact underlying mechanisms.
References
World Health Organization. Obesity and overweight: fact sheet no 3011. Geneva; 2006.
Sjostrom L, Lindroos AK, Peltonen M, et al. Lifestyle, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med. 2004;351:2683–93.
Sjostrom L, Narbro K, Sjostrom CD, et al. Effects of bariatric surgery on mortality in Swedish obese subjects. N Engl J Med. 2007;357(8):741–52.
Adams TD, Gress RE, Smith SC, et al. Long-term mortality after gastric bypass surgery. N Engl J Med. 2007;357(8):753–61.
Sharma A, Dabla S, Aqrawal RP, et al. Serum magnesium: an early predictor of course and complications of diabetes mellitus. J Indian Med Assoc. 2007;105(1):16, 18, 20.
Guerrero-Romero F, Rodriguez-Moran M. Low serum magnesium levels and metabolic syndrome. Acta Diabetol. 2002;39:209–13.
Haglin L, Tornkvist B, Backman L. Prediction of all-cause mortality in a patient population with hypertension and type 2 DM by using traditional risk factors and serum-phosphate, -calcium and -magnesium. Acta Diabetol. 2007;44(3):138–43.
Jorgensen S, Olesen M, Gudman-Hoyer E. A review of 20 years of jejunoileal bypass. Scand J Gastroenterol. 1997;32(4):334–9.
Bloomberg RD, Fleisman A, Nalle JE, et al. Nutritional deficiencies following bariatric surgery: what have we learned? Review article. Obes Surg. 2005;15:145–54.
Correa FH, Nogueira VG, Bevilacqua, et al. Insulin resistance and secretion assessment across a range of glucose tolerance from normal individuals through diabetes. Arg Bras Endocrinol Metabol. 2007;51(9):1498–505.
Sundbom M, Gustavsson S. Bariatric surgery. Clin Dermatol. 2004;22:325–31.
Van Gaal L, Delvigne C, Vandewoude M, et al. Evaluation of magnesium before and after jejuno-ileal versus gastric bypass surgery for morbid obesity. J Am Coll Nutr. 1987;5:397–400.
Diniz Mde F, Diniz MT, Sanches SR, et al. Elevated serum parathormone after Roux-en-Y gastric bypass. Obes Surg. 2004;14(9):1222–6.
Goode LR, Brolin RE, Chowdhury HA, et al. Bone and gastric bypass surgery: effects of dietary calcium and vitamin D. Obesity Res. 2004;12(1):40–7.
Yajnik CS, Smith RF, Hockaday TD, et al. Fasting plasma magnesium concentrations and glucose disposal in diabetes. Br Med J (Clin Res Ed). 1984;288(6423):1032–4.
Rosolová H, Mayer O Jr, Reaven GM. Insulin-mediated glucose disposal is decreased in normal subjects with relatively low plasma magnesium concentrations. Metabolism. 2000;49(3):418–20.
Lima Mde L, Pousada J, Barbosa C, et al. Magnesium deficiency and insulin resistance in patients with type 2 diabetes mellitus. Arg Bras Endocrinol Metabol. 2005;49(6):959–63.
Huerta MG, Roemmich JN, Kington ML, et al. Magnesium deficiency is associated with insulin resistance in obese children. Diabetes Care. 2005;28(5):1175–81.
Djurhuus MS, Henriksen JE, Klitgaard NA, et al. Effect of moderate improvement in metabolic control on magnesium and lipid concentrations in patients with type 1 diabetes. Diabetes Care. 1999;22(4):546–54.
Schnack C, Bauer I, Preqant P, et al. Hypomagnesaemia in type 2 (non-insulin-dependent) diabetes mellitus is not corrected by improvement of long-term metabolic control. Diabetologia. 1992;35(1):77–9.
Paolisso G, Sgambato S, Gambardella A, et al. Daily magnesium supplements improve glucose handling in elderly subjects. Am J Clin Nutr. 1992;55(6):1161–7.
de Valk H, Verkaaik R, van Rijn H, et al. Oral magnesium supplementation in insulin-requiring type 2 diabetic patients. Diabet Med. 1998;15:1161–7.
Eibl N, Kopp H, Nowak H, et al. Hypomagnesemia in type II diabetes: effect of a 3 month replacement therapy. Diabetes Care. 1995;18:188–92.
Widman L, Wester PO, Stegmayer BK, et al. The dose-dependent reduction in blood pressure through administration of magnesium. A double blind placebo controlled cross-over study. Am J Hypertens. 1993;6(1):41–5.
Acknowledgements
Funding was by research grants from The Family Ernfors Fund for Diabetes Research, the Swedish Diabetes Association, the Erik, Karin and Gösta Selander fund, Uppsala University, and The Thuring family foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Johansson, HE., Zethelius, B., Öhrvall, M. et al. Serum Magnesium Status After Gastric Bypass Surgery in Obesity. OBES SURG 19, 1250–1255 (2009). https://doi.org/10.1007/s11695-008-9536-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-008-9536-5