Abstract
South American Canids are endemic and form a monophyletic clade supported by molecular and morphological data, with the exception of Urocyon cinereoargenteus, which is a typical North American form. South American canids occur in almost all environments in continent, and exhibit diet diversity and large size variation. Here we analyzed the skull ontogeny by applying 3D geometric morphometric techniques, in a well-represented sample of South American canids, with the aim of exploring variation in cranial size and shape as well as evolutionary patterns of such variation. The sample consisted of 1227 crania of the 11 living species of South American canids and 92 crania of Canis lupus and Vulpes vulpes, which were considered as out-groups. South American canids exhibited similar shape and diet (they are mostly omnivorous), which implies similar mechanical commitments, which are reflected in their almost uniform skulls. However, it is possible that cranial similarity is related to a shared evolutionary history with a short time of divergence. Speothos venaticus differed in its ontogenetic trajectory, with the difference being stronger between adults of this species and the remaining canids. The skull configuration is consistent with the hyper-carnivorous diet. In the case of Chrysocyon brachyurus, size plays an important role in the structure of a skull, giving rise to remarkable morphological differences from other species, although in terminal stages of its ontogeny.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Modern canids belong to the Family Canidae and to the only surviving subfamily, Caninae. Caninae originated in North America in the late Miocene (10 Ma) and were confined to this area before they spread over the world. Geological events such as the uplift of the Isthmus of Panama that connected North and South America (3 Ma) allowed Caninae to disperse to South America, radiate and reach the current diversity (Berta 1987; Wang et al. 2008; Prevosti 2010; Prevosti and Forasiepi 2018). The extant species in South America originated from several migrations, as suggested by different lineages (Wayne et al. 1997; Perini et al. 2010; Prevosti 2010; Prevosti and Soibelzon 2012; Prevosti and Forasiepi 2018). There are 11 living species: Atelocynus microtis, Cerdocyon thous, Chrysocyon brachyurus, Lycalopex culpaeus, L. fulvipes, L. griseus, L. gymnocercus, L. sechurae, L. vetulus, Speothos venaticus, and Urocyon cinereoargenteus (Wozencraft 2005; Sillero-Zubiri 2009). They are endemic to the region, are included in the tribe Canini, and form a monophyletic clade supported by molecular and morphological data (Tedford et al. 1995; Soibelzon and Prevosti 2007; Prevosti 2010; Prevosti and Soibelzon 2012; Prevosti and Forasiepi 2018) (Fig. 1). The exception is U. cinereoargenteus, which is a typical North American form (Tedford et al. 1995). South American canids occur in almost all environments in the continent (Sillero-Zubiri 2009). They exhibit diet diversity, ranging from omnivores, which consume mainly insects (e.g., L. vetulus), to hypercarnivores, which consume mainly vertebrate prey such as S. venaticus (Beisiegel and Zuercher 2005; Dalponte 2009). The dietary divergence seems to allow the coexistence of the species (Juarez and Marinho-Filho 2002). In addition, there is a large size variation, with larger species such as C. brachyurus (20.5–30 kg), and smaller ones such as L. fulvipes (1.9–3.95 kg) (Sillero-Zubiri 2009).
Cladogram of canid relationship based on Austin et al. 2013. This tree is pruned to indicate only species considered in this work
The skull is a complex structure that houses the brain and the sensory capsules, provides areas of attachment of muscles related to opening and closing of the mandible and chewing, and bears the teeth (Moore 1981; Emerson and Bramble 1993). The form (shape and size) of the cranium is different at different life stages, and natural selection acts particularly over some characters, varying with age (Zelditch and Carmichael 1989; Galatius et al. 2011). The adult skull can vary to develop resistance to external forces (e.g., tensile and compressive strains) generated by natural actions such as catching and subduing prey and processing a variety of food items included in the carnivorous diet, especially in canids, which are mostly omnivores (Thomason 1991; Damasceno et al. 2013). Besides, the size variation (allometry) can play an important role in the morphological diversity of cranial shape (Morales and Giannini 2010), and it could be related to mechanisms to reduce the overlap of niches and reduce the ecological pressure of competition (Werdelin 1983; Sicuro 2011; Sicuro and Oliveira 2011). Phylogeny, on the other hand, is a representation of evolutionary history, and different researchers have evaluated its effects as a factor that models the skull (e.g., Goswami 2006; Wroe and Milne 2007; Figueirido et al. 2011; Meloro and O′ Higgins 2011).
The skull morphology of South American canids has been previously approached from different perspectives such as phylogenetic relationship (Tedford et al. 1995; Perini et al. 2010; Prevosti 2010; Zrzavý et al. 2018); historical biogeography (Prevosti and Rincón 2007; Machado and Hingst-Zaher 2009); ecological variation (Martinez et al. 2013; Bubadué et al. 2016a, b; Zurano et al. 2017); sexual variation (Travaini et al. 2000); dental variation (Prevosti and Lamas 2006), and taxonomy (Zunino et al. 1995; Guzmán and Ortiz 2009; Prevosti et al. 2013). However, most of the studies in skull ontogeny (i.e. sequence of events of skull growth and development) in canids have been primarily descriptive or focused on dental eruption and age estimation (e.g. Canis familiaris: Kremenak 1969; Kremenak et al. 1969; Canis latrans: Linhart and Knowlton 1967; Bekoff and Jamieson 1975; Knowlton and Whittemore 2001; Canis lupus: Goodwin and Ballard 1985; Landon et al. 1998; Gipson et al. 2000; Canis mesomelas: Lombaard 1971; Bingham and Purchase 2003; L. culpaeus: Crespo and De Carlo 1963; Zapata et al. 1997; Segura 2013; L. gymnocercus: Crespo 1971; Prevosti and Lamas 2006; U. cinereoargenteus: Wood 1958; Root and Payne 1984; Vulpes lagopus: Prestrud and Nilssen 1995; Vulpes vulpes: Linhart 1968; Grue and Jensen 1973; Harris 1978). There are some exceptions, which have quantified the canid skull ontogeny: Wayne (1986) studied the ontogenetic trajectories in dog (C. familiaris), and found differences between them and other wild canids; Drake (2011) investigated heterochronic patterns in cranial morphology of C. familiaris, showing that adult cranial shape is not similar to any of the ontogenetic trajectories of wolves (C. lupus); Werneburg and Geiger (2017) studied pre- and postnatal patterns of cranial development in C. familiaris and compared them with other carnivorans; La Croix et al. (2011a, b) detected synchronous and asynchronous growth patterns in skulls of coyotes (C. latrans); Segura and Prevosti (2012) studied the ontogenetic trajectory in L. culpaeus and indicated that shape changes are related to functional improvements in terms of food catching and processing; and Forbes-Harper et al. (2017) analyzed the ontogeny of the diet of V. vulpes, in terms of cranial morphology and sexual dimorphism. However, the integral study of the skull ontogeny in South American canids (which share an evolutionary and biogeographic history), looking for answers in a morphological, functional and evolutionary context, is a very interesting, still unexplored topic that can be analyzed quantitatively.
Here, we analyzed the skull ontogeny by applying 3D geometric morphometric techniques in a well-represented sample of South American canids, exploring the variation in cranial size and shape as well as evolutionary patterns of such variation with the aim of to test hypotheses suggesting that cranial variation may be addressed by allometry (size), ecology (diet), and/or phylogeny.
Materials and methods
Background information
The biological information about diet, birth and adult weight, gestation period, litter size, weaning, sexual maturity, and lifespan of South American canids was compiled from published literature and is summarized in Table 1.
Sample
The sample consisted of 1227 crania of the 11 living species of South American canids and 92 crania of Canis lupus and Vulpes vulpes, which were considered as out-groups. The sample included cubs, juveniles and adults of different age classes estimated by dental eruption and tooth wear as in Segura et al. (2013), with modifications (Table 2). Age classes are present in the sample in different proportions (Table 3). The analyzed material belongs to the mammal collections of the American Museum of Natural History (AMNH), Colección Félix de Azara (CFA), Colección Mamíferos Lillo (CML), Field Museum of Natural History (FMNH), Colección del Grupo de Ecología comportamental de Mamíferos (GECM), Colección de Mamíferos del Laboratorio de Investigaciones en Evolución y Biodiversidad (LIEB), Museo Argentino de Ciencias Naturales Bernardino Rivadavia (MACN), Museo Florentino Ameghino de Zoología de vertebrados (MFA-ZV), Colección Mastozoológica del Museo de La Plata (MLP), Museu de zoología da Universidade de São Paulo (MZUSP), and National Museum of Natural History, Smithsonian Institution (NMNH) (Appendix 1).
Landmarks
To describe the postnatal cranial ontogeny in South American canids, 38 cranial landmarks (Fig. 2) types 1 and 2 (sensu Bookstein 1991) were digitized with a Microscribe MX6DOF System (GoMeasured3D, Amherst, VA, USA). Following Segura et al. (2017), only one half of the cranium was digitized and reflected in the symmetry plane using R-function AMP.r developed by Annat Haber (http://life.bio.sunysb.edu/morph/). This procedure improves visualization and avoids the putative Procrustes alignment artifacts.
Data analysis
To remove the spatial variation that does not correspond to form, landmark configurations were superimposed using Generalized Procrustes Analysis (GPA Goodall 1991; Rohlf 1999). This analysis minimizes the sum of squared distances between homologous landmarks by translating, rotating, and scaling them to unit (Dryden and Mardia 1998). Procrustes coordinates data, obtained from the Procrustes superposition, were used to calculate Procrustes distance (PD), an index of shape change (e.g. Tanner et al. 2009; Segura and Prevosti 2012), calculated as the square root of the sum of the squared distances between each landmark of one specimen and the mean configuration of the smallest age class. Centroid size (CS) was used as an estimate of skull size and was calculated as the square root of the sum of squared distances of each landmark from the centroid of the landmark configuration (Bookstein 1991; Zelditch et al. 2004). Both estimators were calculated using R 2.9.2 software (R Development Core Team 2018). Following Segura (2015), CS and PD of all age classes and for each species were included and used to know the age class at which the final adult cranial size and shape were reached. In addition, this information was combined with ontogenetic and reproductive data from the literature (Table 1) to obtain integrative results in our ontogeny comparisons. To identify the major components of variation, a principal component analysis (PCA) of all South American canids was performed across all age classes. This analysis allows us to visualize the ontogenetic shape changes along the components of interest (PCs). In addition, another PCA only taking into account the adult specimens was performed to explore the differences and similarities in the offset of all canid species of the sample.
To investigate how variation in shape is associated with size, a multivariate regression of the Procrustes coordinates against the log-transformed centroid size was performed for each species of South American canids, taking into account all age classes. Lycalopex fulvipes was not included because of its low sample size (N = 2). The significance of regressions was tested using a permutation test with 10,000 resamples (Bookstein 1991; Mitteroecker et al. 2004). To evaluate the relationship between the trajectories of the canid species, we pooled all the species together and performed the same analysis, taking into account all ontogenetic series. In addition, the significance of all analyses was assessed by 10,000 rounds of permutation test. In addition, another regression taking into account the mean of each age class for each species of South American canid was performed to explore if the elimination of static allometry produces differences in the results. All these morphometric analyses were performed with MorphoJ 1.05a software (Klingenberg 2011).
We explored the change of cranial form (i.e. size and shape) and diet throughout the evolution of canids by optimizing diet and cranial form data with TNT 1.5 software (Goloboff and Catalano 2016). We followed the phylogenetic hypothesis proposed by Austin et al. (2013). Size data corresponded to the logarithm of centroid size, and our phylogenetic interpretation of the change was based on J1 (younger) and A3 classes (older) for each species, excluding species where these age classes are not represented. These values were optimized as a continuous character (Goloboff et al. 2006). Data of diet was compiled from Wilson and Mittermeier (2009) (Table 1) and coded as a non-additive discrete character: omnivore (state 0), mesocarnivore (1), and hypercarnivore (2). Shape data corresponded to the predicted shape (i.e. configurations) of J1 and A3 classes, obtained from regressions of shape coordinates vs logarithm of CS previously performed for each species. Cranial shape was optimized as landmark configurations (Catalano et al. 2010; Catalano and Goloboff 2012). Following Muñoz et al. (2017), visualization and graphics were made using the Morpho R package 2.5.1 (Schlager 2017).
Previous studies suggested sexual size dimorphism in external and cranial measurements of Lycalopex culpaeus (Crespo and De Carlo 1963; Travaini et al. 2000) and Lycalopex gymnocercus (Prevosti and Lamas 2006). For this reason, we tested the dimorphism in our sample. In the case of allometric variation, males and females exhibited the same ontogenetic trajectory, indicating that the observed allometric pattern is not biased by sexual dimorphism. We tested sexual size (CS) and shape (PD) dimorphism using the Mann-Whitney U-test (Zar 2009) with the software PAST (Hammer et al. 2001) (Table S1).
Results
The first principal component of the PCA (PC1) explained ~28.91% of the total skull variation (Fig. 3). Along PC1, information was related to ontogeny, with juveniles and adults located from the positive to the negative side. The three adult classes (A1, A2, and A3) exhibited superposition between them (for all species), whereas juveniles were progressively located in relation to this PC. Compared with the adult cranium morphology, the juvenile cranium was rounded and shorter, with shorter and wider nasals, rostrum, and palate; it had a wider frontal bone, larger orbits, shorter, wider and taller braincase, and narrower temporal fossa. The zygomatic process of the maxilla was taller; the basicranium was wider, with larger bullae, shallower glenoid fossa, lesser-expanded glenoid process, and the inion placed anterior to the level of foramen magnum.
The adult cranium exhibited relatively elongated and narrowed nasals, rostrum, and palate, with expanded incisive bone and nasal process of the nasals. The frontal bone and the braincase were narrower, the braincase was elongated and flatter, the temporal fossa was broader, and the orbits were relatively smaller. Zygomatic arches were laterally well developed; the basicranium was narrower, with smaller bullae, deeper glenoid fossa, expanded glenoid process, and the inion was placed posterior to foramen magnum. Speothos venaticus was exceptional, with juveniles and adults located on positive values of the PC1. PC2 explained ~8.33% of the total variation (Fig. 3). The adult specimens of S. venaticus were located to the negative score of this axis; the cranial configuration of this species was different from the remaining canids: the cranium was more massive and shorter, with shorter and wider palate, rostrum, and nasals. The orbits were smaller and more forward-oriented and the braincase was flatter and narrower, principally at the level of postorbital constriction. The basicranium was narrower; the bullae was smaller; the zygomatic arches were straight, well developed and laterally expanded; and the temporal fossa was broader. The occipital condyles and the foramen magnum were expanded posteriorly to the inion. Toward the positive values of the score, where the remaining canids were placed, the cranial configuration was more slender and elongated, with longer and narrower palate, rostrum, and nasals. The orbits were larger and more laterally oriented; the braincase was more rounded, taller and wider; the basicranium was wider and the bullae was larger. The zygomatic arches were curved; the temporal fossa was narrower; the occipital condyles and foramen magnum were at the same level of the inion. There was a cranial flexion because the premaxillary bone and the bullae were below the level of the tooth row.
The PCA including only adult specimens, showed the same pattern, with S. venaticus being located at a distance from the remaining species. In addition, Chrysocyon brachyurus exhibited a different position in the morphospace, although with some specimens overlapped with the rest of canids (Fig. S2).
In all species, CS values were gradually increased, reaching the asymptote in adult classes (Fig. S3): C. thous and L. sechurae in A1 class, L. culpaeus, L. griseus, L. gymnocercus, S. venaticus and U. cinereoargenteus in A2, and C. brachyurus in A3 class. The Procrustes distance values were increased, reaching the asymptote at different age classes (Fig. S3). The most precocial was L. gymnocercus, which arrived to the definitive shape at J4 class. C. thous, L. sechurae, S. venaticus, and U. cinereoargenteus reached the definitive shape at A1, and C. brachyurus, L. culpaeus, and L. griseus at A2 class. In four species, C. thous, L. culpaeus, L. griseus, and L. sechurae, growth and development of the cranium were synchronous (Fig. 4). Most species (62.5%) obtained the final size in A2 class, 25% in A1, and 12.5% in A3. Half of the species reached the final shape in A1 class, 37.5% in A2, and 12.5% in J4. In general, the final shape was obtained before final size, even in the juvenile class (J4). For all species, the final shape was reached before reaching the last class of adults. The final size was reached late, in adult classes, and in some cases, it was reached even in the last class (A3).
Timeline illustrating the age classes in relation to life-history events for South American canids. Symbols:  C. t., Cerdocyon thous;
C. t., Cerdocyon thous;  C. b., Chrysocyon brachyurus;
C. b., Chrysocyon brachyurus;  L. c., Lycalopex culpaeus;
L. c., Lycalopex culpaeus;  L. g., Lycalopex griseus;
L. g., Lycalopex griseus;  L. gy., Lycalopex gymnocercus;
L. gy., Lycalopex gymnocercus;  L. s., Lycalopex sechurae;
L. s., Lycalopex sechurae;  S. v., Speothos venaticus;
S. v., Speothos venaticus;  U. c., Urocyon cinereoargenteus.
U. c., Urocyon cinereoargenteus.  Represents cranial size;
Represents cranial size;  represents cranial shape. Dd, complete decidual dentition; Ad, complete adult dentition; L, lifespan; *information of lifespan in captivity
represents cranial shape. Dd, complete decidual dentition; Ad, complete adult dentition; L, lifespan; *information of lifespan in captivity
Size explained 6.03% of shape variation in Atelocynus microtis, 31.06% in Cerdocyon thous, 12.52% in C. brachyurus, 12.77% in Lycalopex culpaeus, 24.15% in L. griseus, 8.06% in L. gymnocercus, 12.87% in L. sechurae, 8.52% in L. vetulus, 25.46% in S. venaticus, and 28.84% in Urocyon cinereoargenteus (p < 0.0001). All species shared a similar pattern of allometry. In general, the larger forms presented a narrower and longer muzzle, expanded zygomatic arches, and a narrower and flatter braincase, whereas the smaller forms exhibited the opposite configuration (Fig. S4). In the general regression, including all species for all age classes plotted together (Fig. 5), size explained 16.79% of shape variation (p < 0.0001). This analysis clearly showed a different intercept for the regressions of S. venaticus and C. brachyurus. The regressions including the means of each age class for each species of South American canid showed the same pattern that the general regression including all individuals (Fig. S5).
General analysis of multivariate regression of the Procrustes coordinates against the log-transformed centroid size for all age classes. Symbols:  Atelocynus microtis,
Atelocynus microtis,  Cerdocyon thous,
Cerdocyon thous,  Chrysocyon brachyurus,
Chrysocyon brachyurus,  Lycalopex culpaeus,
Lycalopex culpaeus,  Lycalopex fulvipes,
Lycalopex fulvipes,  Lycalopex griseus,
Lycalopex griseus,  Lycalopex gymnocercus,
Lycalopex gymnocercus,  Lycalopex sechurae,
Lycalopex sechurae,  Lycalopex vetulus,
Lycalopex vetulus,  Speothos venaticus,
Speothos venaticus,  Urocyon cinereoargenteus
Urocyon cinereoargenteus
Regarding the diet (Fig. 6), optimization showed that omnivory (orange) seems to be the ancestral diet, whereas the mesocarnivorous (magenta) and hypercarnivorous (green) diet appeared in independent events. Regarding shape and size (Fig. 6), U. cinereoargenteus showed more changes in the phylogeny, exhibiting a juvenile cranium with a taller braincase than that of the ancestor, and a slightly broader adult cranium. In both juveniles and adults, size (i.e. CS) decreased in relation to the ancestor. The juvenile braincase of Canis lupus was slightly narrower and flatter, and the muzzle was longer than that of its ancestor. In turn, the adult braincase was narrower and taller, and the muzzle was longer than that of its ancestor. Size increased in juvenile and adult cranium of this species. The most important changes observed in S. venaticus occurred in the juvenile cranium, with a broader braincase and muzzle than those of their ancestor. In the adult cranium, the braincase and the muzzle were broader and taller. Only for the adult cranium, the size decreased in relation to that of the ancestor. Compared to ancestor braincase, the braincase of C. brachyurus juvenile was proportionally narrower and flatter, with developed zygomatic arches and more expanded incisive bone. The adult braincase was narrower, with more expanded supraorbital process and incisive bone than that of its ancestor. In both juveniles and adults, cranium size increased with respect to the ancestor. In L. griseus, only the juvenile cranium changed with respect to the ancestor cranium, showing a broader and taller braincase. The size of both juvenile and adult crania decreased. Conversely, in L. culpaeus, the juvenile cranium was narrower and flatter than the ancestor cranium, and the tip of the muzzle was more expanded. In the adult cranium, the difference with their ancestor was observed on the lateral expansion of the zygomatic arches. In juveniles and adults, the cranium size was increased in relation to their ancestor. In C. thous, the change in comparison to their ancestor was minor: the juvenile cranium had a taller braincase and a decreased cranium size. The adult cranium of L. sechurae was smaller than that of its ancestor (Fig. 6).
Discussion
Our main results suggest a striking uniformity in the cranial ontogenetic trajectories of South American canids. We found greater differences of the cranium in adults of Speothos venaticus (with short and broad muzzle, large bullae, and short, broad and tall braincase), and Chrysocyon brachyurus, with a similar pattern to that of the remaining canids (relatively elongated nasals, narrower and longer braincase, see Segura and Prevosti 2012), but larger size (see below). The processes responsible for the cranial phenotypic diversity (or its lack) could be related to the allometry (size), the ecology (diet), or the evolutionary history (phylogeny) of the species as the main factors that shape the carnivore cranium (Goswami 2006; Wroe and Milne 2007; Goswami et al. 2010; Figueirido et al. 2011). Other authors mentioned that the stabilizing selection and limited genetic variation are processes that could limit the phenotypic divergence (e.g. Wiens and Graham 2005; Zurano et al. 2017). However, understanding these processes is difficult because they are generally complex (Perez et al. 2011).
Growth and development of South American canids
Cubs and juveniles of South American canids are very similar in cranial shape (Figs. 3, 5, 6), showing more rounded cranium, with short and wide nasals, rostrum, palate and frontal bone; large orbits and bullae; short, wide and tall braincase; narrow temporal fossa; less developed zygomatic arches and glenoid process; wide basicranium; shallow glenoid fossa, and foramen magnum posterior to the inion. The adult crania show the opposite configuration (Figs. 3, 5, 6). These changes imply negative allometry of neurocranial components (e.g. braincase, orbits and auditory bullae) and positive allometry of splanchnocranium (e.g. rostrum, palate, zygomatic arches). This unequal growth produces notable differences in crania between juveniles and adults of canids, showing different commitments related to feeding (Radinsky 1981), environment perception, and size increase. The pattern seems to be plesiomorphic for mammals (Emerson and Bramble 1993), and was previously reported for canids and other carnivores (e.g. Biknevicius and Leigh 1997; Segura and Flores 2009; Slater and Van Valkenburgh 2009; Giannini et al. 2010; Segura and Prevosti 2012; Segura 2013; Segura et al. 2013; Segura 2015; Sánchez-Villagra et al. 2017; Flores et al. 2018). In eutherians, the central nervous system morphogenesis starts before somatic tissue differentiation. This process extends throughout the fetal and embryonic periods, when nutrition is relatively constant and there is little competition from other tissues. The central nervous system induces the membranous bone of the braincase, which also develops earlier (Smith 1997; Byron 2006; Rice 2008), determining the neurocranial form and the relative larger sensory capsules observed in juveniles.
The configuration of the juvenile palate (broad and short) is associated with the function as a platform where the tongue acts during lactation (German and Crompton 1996; Smith 1997). Thus, the tongue (the first muscle to differentiate in mammals) pushes the nipple against the broad palate, exerting a differential pressure that produces the flow of milk in the oral cavity (German et al. 1992; Wall and Smith 2001; Thexton et al. 2004). The reorganization of the splanchnocranium seems to emphasize the predatory ability of adults (Slater et al. 2009; Slater and Van Valkenburgh 2009; Black et al. 2010). The glenoid fossa is deeper and pre and post glenoid processes are well developed, reinforcing the temporomandibular joint, which undergoes strong mechanical stress in adults (e.g. Segura and Flores 2009). Additionally, the development of processes does not allow rostral-caudal and transversal movements, limiting the mobility of the mandible to a vertical plane (Biknevicius and Van Valkenburgh 1996; Segura and Flores 2009). The masticatory and cervical muscles increase in size in adults, which is related to the need to perform more complex movements that allow them to subdue their prey.
The growth of sagittal and nuchal crests (both structures are visible when the inion is placed posterior to foramen magnum), provides more surface to attach of larger masticatory and cervical musculature in adults (Wickland et al. 1991; Richmond et al. 1992; Evans 1993; Duckler 1998; Antón et al. 2004). The enlargement of the temporal fossa in adults has the same function as that of masticatory muscles (temporal and masseter). The temporal muscle is the most important in Carnivora because it pulls the jaw upwards and backwards and better resists the forces produced by prey during hunting, which act downwards and forwards (Cox 2007). The increase in volume of this muscle during growth implies a more powerful action (García-Perea 1996). The masseter is a smaller masticatory muscle in carnivores, although it is important because it keeps the stability of the jaw (Hildebrand 1995; Segura and Flores 2009). There is a tradeoff between temporal muscle and brain size: carnivores have a proportionally small braincase that gives room to a small brain and a large masticatory area, and producing high bite force. Conversely, in Homo (as an extreme example) and in a medium-sized herbivorous, such as Ozotoceros bezoarticus, have a large braincase and encephalization, and markedly reduced temporal muscle (Wroe and Milne 2007; Cassini et al. 2015).
The skull of Canids has a conservative plan that in general does not include reduction of teeth (Ewer 1973), keeping an elongated rostrum. This fact provides raw material upon which natural selection operates (Wayne 1986). For this reason, there is enough flexibility to have a relatively short rostrum in hyper-carnivore forms (such as Speothos) and long ones in omnivores (Machado et al. 2018; Wang et al. 2008). The lengthening of rostrum is related to a craniomandibular third-order lever, which implies faster jaw closure albeit at the expense of bite force (Westneat 2003; Cassini and Vizcaíno 2012). Selection tends to favor faster closing jaws in canids that specialize in small, fast moving prey, such as C. brachyurus (Aragona and Setz 2001; Cassini and Vizcaíno 2012). Conversely, a short and wide muzzle produces large bite forces probably required to subdue large vertebrate prey, as observed in S. venaticus (Slater et al. 2009). This morphology configures a craniomandibular lever system more suitable for force enhancement (Cassini and Vizcaíno 2012). The difference in cranial shape exhibited by S. venaticus in relation to the rest of canids is even stronger in adults. The skull has a reduced length of the tooth row due to a reduction in post carnassial molars; therefore, the muzzle is short. This combination of characters is expected for a hyper-carnivore, and in this sense, the skull of Speothos is more similar to a felid skull than a typical canid skull. In fact, the shortening of lever arms and the development of the areas to attach more massive masticatory muscles allow Speothos to have a larger bite force and a more powerful muscular action (Ewer 1973; Radinsky 1981; Christiansen and Adolfssen 2005; Wang et al. 2008).
Size
Size variation plays an important role in morphological diversity (Sicuro and Oliveira 2011), being a mechanism to reduce niche overlap and competitive pressure (Morales and Giannini 2010; Sicuro 2011). A larger body size enhances an animal’s ability to capture large prey and avoid predators (Van Valkenburgh et al. 2004). Small differences in genetically-regulated growth factors can alter size (Cardini and Polly 2013), and when size increases, shape tends to change (Wayne 1986; Slater and Van Valkenburgh 2009). These changes can define the ability of an organism to perform its activities (Emerson and Bramble 1993; Wainwright and Reilly 1994), such as those related to prey capture and feeding (Slater and Van Valkenburgh 2009). In our results, Chrysocyon brachyurus had a similar cranial pattern to that of other canids, but a different size, with adults being larger than the rest, and dragging part of the shape (Figs. 3-7). Chrysocyon brachyurus was born larger (350 g) and grew for a longer period than the remaining canids (Figs. 4, 7). In mammals, growth rate is maximal after birth and slows with age (Helm and German 1996). For this reason, skull is not expected to undergo changes in size and/or shape in an old age class, such as A3. However, this phenomenon occurred in C. brachyurus, and was previously reported for the hyaenid Crocuta crocuta (Tanner et al. 2009) and pantherines such as Panthera leo, P. onca, and P. tigris (Segura et al. 2017). There seems to be a correspondence between size and the time necessary to develop a skull able to resist the stress imposed by the diet. These carnivores have protracted growth and as result of their ontogenies, they reach reinforced and massive skulls to prey on larger animals. Previous works reported that among canids, size is associated with ecological differences, because larger canids tend to consume more meat and take larger prey than small canids do (Gittleman 1985; Van Valkenburgh 1989; Wayne et al. 1989). This does not apply to South American canids, because the largest species in this group, Chrysocyon brachyurus (20.5–30 kg.), has more flexible feeding habits than Speothos venaticus, a small-size hypercarnivorous canid (5–8 Kg). The South American canid pattern agrees with findings reported by Fuentes and Muñoz et al. (2017), who found a relationship of carnivore habits with socialization and not with the size of the canid species. Regarding cranial shape, the observed changes in the ontogeny of C. brachyurus are not so important (Figs. 3, 6), and despite the large size of this species, it feeds principally on small rodents and not on larger prey. Speothos venaticus is able to hunt prey that exceed its own body size because it is a gregarious pack hunter canid (Zuercher et al. 2004). The advantage of cooperative hunting could be the reduced development of morphological specializations to capture large prey (Van Valkenburgh and Koepfli 1993). However, in the case of Speothos venaticus, cranial morphology is typical of a hypercarnivore (i.e. wider and massive crania with shorter muzzle).
Diet
Juveniles undergo a transition from a diet based almost exclusively on milk to one involving active mastication, which can diverge into a more omnivorous or carnivorous diet. It is known that slight changes in feeding can produce great changes in skull morphology, and this shift produces two immediate impacts in carnivores: a change in the forces generated by capturing and chewing prey, and a difference in nutritional values (Helm and German 1996). The feeding morphotypes (or ecomorphs) that are clearly recognizable in the evolutionary history of Carnivora are limited (Van Valkenburgh 2007; Figueirido et al. 2011). The cranial morphology of hypercarnivores (with reduced dentition and short rostrum) can be differentiated from that of omnivores (with complete dentition and long rostrum) (Tseng and Flynn 2018). Our results showing an ancestor with an omnivorous diet (Fig. 6) are consistent with the hypothesis that canids evolved from a generalized to specialized forms (Wang and Tedford 2007; Fuentes-González and Muñoz-Durán 2017). Maintaining a generalist diet allowed canids to have more versatility to evolve to different diets, such as omnivorous and carnivorous (Wang and Tedford 2007; Fuentes-González and Muñoz-Durán 2017). It has been suggested increasing specialization leads to reduced morphological disparity (Holliday and Steppan 2004; Van Valkenburgh 2007), and, similarly, non-specialized species of South American canids are highly diverse because of their generalist feeding strategies (Berta 1987; Tchaicka et al. 2016; Zurano et al. 2017). Conversely, our results show that South American canids (except for S. venaticus) share a similar morphological pattern (Figs. 3, 6), suggesting an ancestral pattern in the diet (omnivorous, which is the general trend in the group), and in cranial morphology (narrow and long crania). This conservative pattern may be attributed to the biomechanically optimal morphology for this kind of generalist diet, and shows that under similar conditions, the skull form tends to converge (Van Valkenburgh 2007). Other authors found variation in the skull of canids in relation to diet based on the analysis of different tooth variables (e.g. Van Valkenburgh 1989, 1991; Wayne et al. 1997; 1989; Prevosti 2006; Wang and Tedford 2007; Fuentes-González and Muñoz-Durán 2017), but this suggests that difference among canids could be related to dentition, rather than to skull shape.
Cranial similarities and differences are related to the taxonomic scale of analysis. When the study included only the Lycalopex lineage or only South American species (excluding Chrysocyon brachyurus and Speothos venaticus), subtle cranial differences between these species were observed (e.g. Zurano et al. 2017). However, when the study included those species, which are morphologically extreme, they gathered the greatest amount of variation, blurring more subtle patterns that may appear in the remaining species.
Phylogeny
Speothos venaticus and Chrysocyon brachyurus are placed as sister group and shared an ancestor that diversified 3 Ma (Perini et al. 2010; Prevosti 2010; Austin et al. 2013; Zrzavý et al. 2018). This early separation from the remaining canids is reflected in their particular cranial morphologies, which are the most different (in shape and size) from that of the remaining South American canids (Figs. 3, 5). In fact, there are notable differences between those species, which have opposite cranial trends. The morphological conservatism of the ontogenetic trajectories of the remaining South American canids (Fig. 5) could be explained by their shared recent evolutionary history and the short time of divergence (e.g., ancestor of the Atelocynus + Cerdocyon + Lycalopex clade was inferred at 2.4–2.7 Ma, the diversification of the Lycalopex lineage at 1.3 Ma, and the age of the oldest fossil assigned to this genus is ca. 2.6 Ma [Perini et al. 2010; Prevosti and Soibelzon 2012; Prevosti and Forasiepi 2018]). In addition, the zoogeographical distribution of the Lycalopex lineage was restricted to South America (Eizirik 2012). For this reason, the similarity in their cranial ontogenetic patterns is not surprising (Blomberg et al. 2003; Zurano et al. 2017). Moreover, most changes were detected on terminals (autapomorphies), whereas fewer changes occur in internal nodes (synapomorphies) (Fig. 6), also denoting conservatism in the evolution of the ontogeny. This result agrees with the scenario of the adaptive radiation (i.e. the evolutionary divergence of members of phylogenetic lineage) that allowed specific differentiation over a short period (Losos and Miles 2002).
Our results show that the canid ancestor could be a medium-sized canid with an omnivorous diet (Fig. 6). In the Chrysocyon brachyurus-Speothos venaticus clade, these species exhibit an opposite trend. Adult Speothos venaticus has a rounded, short and wide cranium, which decreases in size in relation to the ancestor (Figs. 3, 6). These characteristics are similar to those of juveniles of the remaining species; for this reason, Biben (1983) described Speothos venaticus as paedomorphic. This type of heterochrony is probably produced by an earlier offset (progenesis or hypomorphosis), or by a reduced rate of growth (neoteny or deceleration) (Figs. 4, 6, 7, Reilly et al. 1997; Klingenberg 1998; McNamara 2012). By contrast, Chrysocyon brachyurus shows an elongated and narrow cranium, which increases in size in relation to the ancestor (Fig. 6). In addition, this species is considered peramorphic because it grows larger and for a longer period (Figs. 4, 7). This kind of heterochrony is probably produced by a delayed offset (hypermorphosis), or by an increased growth rate (acceleration) (Figs. 4, 6, 7, Reilly et al. 1997; Klingenberg 1998; McNamara 2012). However, this phenomenon must be tested in a more inclusive cladistic context.
Conclusion
In summary, we observed that Speothos venaticus differed in its ontogenetic trajectory, with the difference being stronger between adults of this species and the remaining canids. The skull configuration is consistent with the hyper-carnivorous diet. In the case of Chrysocyon brachyurus, size plays an important role in the structure of the skull, giving rise to remarkable morphological differences from other species, although in terminal stages of its ontogeny. Excluding both species, the remaining South American canids exhibited similar shape and diet (they are mostly omnivorous). This result implies similar mechanical commitments, which are reflected in their almost uniform skulls. However, it is possible that cranial similarity is related to a shared evolutionary history with a short time of divergence.
We hypothesize that the evolution of the skull in South American canids responds to a mosaic of different factors related to size, diet, and phylogeny. Further studies will be aimed at including a greater taxonomic sample of living and fossil species to detect common ontogenetic patterns and variations in specialist groups that show divergences in ontogenetic changes. A strong stability of ontogenetic change in the Family Canidae is expected, although the divergences observed in hypercarnivores such as Speothos suggest different patterns that might be detected with a more inclusive taxonomic sample. A more speciose taxonomic sample also informs about the ontogenetic reconstruction of ancestors, giving a more robust approach of skull ontogenetic evolution in canids.
Availability of data and material
Data supporting this manuscript is available on digital repository of Universidad Nacional de La Plata http://sedici.unlp.edu.ar/handle/10915/41
References
Antón, M., Salesa, M. J., Pastor, J. F., Sánchez, I. M., Fraile, S., & Morales, J. (2004). Implications of the mastoid anatomy of larger extant felids for the evolution and predatory behaviour of sabretoothed cats (Mammalia, Carnivora, Felidae). Zoological Journal of the Linnean Society, 140, 207–221. https://doi.org/10.1111/j.1096-3642.2003.00093.x.
Aragona, M., & Setz, E. Z. F. (2001). Diet of the maned wolf, Chrysocyon brachyurus (Mammalia: Canidae), during wet and dry seasons at Ibitipoca State Park, Brazil. Journal of Zoology, 254, 131–136. https://doi.org/10.1017/S0952836901000620.
Austin, J. J., Soubrier, J., Prevosti, F. J., Prates, L., Trejo, V., Mena, F., & Cooper, A. (2013). The origins of the enigmatic Falkland Islands wolf. Nature Communications, 4, 1552. https://doi.org/10.1038/ncomms2570.
Beisiegel, B. M., & Zuercher, G. L. (2005). Speothos venaticus. Mammalian Species, 783, 1–6. https://doi.org/10.1644/783.1.
Bekoff, M., & Jamieson, R. (1975). Physical development in coyotes (Canis latrans), with a comparison to other canids. Journal of Mammalogy, 56, 685–692. https://doi.org/10.2307/1379485.
Berta, A. (1987). Origin, diversification, and zoogeography of the South American Canidae. Fieldiana Zoology, 39, 455–471.
Biben, M. (1982). Ontogeny of social behaviour related to feeding in the crab-eating fox (Cerdocyon thous) and the bush dog (Speothos venaticus). Journal of Zoology, 196, 207–216. https://doi.org/10.1111/j.1469-7998.1982.tb03501.x.
Biben, M. (1983). Comparative ontogeny of social behaviour in three South American canids, the maned wolf, crab-eating fox and bush dog: Implications for sociality. Animal Behaviour, 31, 814–826. https://doi.org/10.1016/S0003-3472(83)80238-3.
Biknevicius, A. R., & Leigh, S. R. (1997). Patterns of growth of the mandibular corpus in spotted hyenas (Crocuta crocuta) and cougars (Puma concolor). Zoological Journal of the Linnean Society, 120, 139–161. https://doi.org/10.1111/j.1096-3642.1997.tb01276.x.
Biknevicius, A. R., & Van Valkenburgh, B. (1996). Design for killing: Craniodental adaptations of predators. In J. L. Gittleman (Ed.), Carnivore behavior, ecology, and evolution (pp. 393–428). New York: Cornell University Press.
Bingham, J., & Purchase, G. K. (2003). Age determination in jackals (Canis adustus Sundevall, 1846, and Canis mesomelas Schreber, 1778; Carnivora: Canidae) with reference to the age structure and breeding patterns of jackal populations in Zimbabwe. African Zoology, 38, 153–160. https://doi.org/10.1080/15627020.2003.11657203.
Black, K. H., Archer, M., Hand, S. J., & Godthelp, H. (2010). First comprehensive analysis of cranial ontogeny in a fossil marsupial—From a 15-million-year-old cave deposit in northern Australia. Journal of Vertebrate Paleontology, 30, 993–1011. https://doi.org/10.1080/02724634.2010.483567.
Blomberg, S. P., Garland, T., & Ives, A. R. (2003). Testing for phylogenetic signal in comparative data: Behavioral traits are more labile. Evolution, 57, 717–745. https://doi.org/10.1111/j.0014-3820.2003.tb00285.x.
Bookstein, F. L. (1991). Morphometric tools for landmark data: Geometry and biology. Cambridge: Cambridge University Press.
Brady, C. A. (1978). Reproduction, growth and parental care in crab-eating foxes (Cerdocyon thous) at the National Zoological Park, Washington. International Zoo Yearbook, 18, 130–134. https://doi.org/10.1111/j.1748-1090.1978.tb00243.x.
Brady, C. A., & Ditton, M. K. (1979). Management and breeding of maned wolves. International Zoo Yearbook, 19, 171–176. https://doi.org/10.1111/j.1748-1090.1979.tb00557.x.
Bubadué, J., Cáceres, N., dos Santos Carvalho, R., & Meloro, C. (2016a). Ecogeographical variation in skull shape of South-American Canids: Abiotic or biotic processes? Evolutionary Biology, 43, 145–159. https://doi.org/10.1007/s11692-015-9362-3.
Bubadué, J., Cáceres, N., Carvalho, R., Sponchiado, J., Passaro, F., Saggese, F., Mondanaro, A., Raia, P., & Carotenuto, F. (2016b). Character displacement under influence of Bergmann’s rule in Cerdocyon thous (Mammalia: Canidae). Hystrix, 27, 83–90. https://doi.org/10.4404/hystrix-27.2-11433.
Byron, C. D. (2006). Role of the osteoclast in cranial suture waveform patterning. Anatomical Record, 288A, 552–563. https://doi.org/10.1002/ar.a.20322.
Cardini, A., & Polly, P. D. (2013). Larger mammals have longer faces because of size-related constraints on skull form. Nature Communications, 4, 2458. https://doi.org/10.1038/ncomms3458.
Cassini, G. H., & Vizcaíno, S. F. (2012). An approach to the biomechanics of the masticatory apparatus of early Miocene (Santacrucian age) south American ungulates (Astrapotheria, Litopterna, and Notoungulata): Moment arm estimation based on 3D landmarks. Journal of Mammalian Evolution, 19, 9–25. https://doi.org/10.1007/s10914-011-9179-5.
Cassini, G. H., Flores, D. A., & Vizcaíno, S. F. (2015). Postnatal ontogenetic scaling of pampas deer (Ozotoceros bezoarticus celer: Cervidae) cranial morphology. Mammalia, 79(1), 69–79. https://doi.org/10.1515/mammalia-2013-0051.
Catalano, S. A., & Goloboff, P. A. (2012). Simultaneously mapping and superimposing landmark configurations with parsimony as optimality criterion. Systematic Biology, 61, 392. https://doi.org/10.1093/sysbio/syr119.
Catalano, S. A., Goloboff, P. A., & Giannini, N. P. (2010). Phylogenetic morphometrics (I): The use of landmark data in a phylogenetic framework. Cladistics, 26, 539–549. https://doi.org/10.1111/j.1096-0031.2010.00302.x.
Christiansen, P., & Adolfssen, J. S. (2005). Bite forces, canine strength and skull allometry in carnivores (Mammalia, Carnivora). Journal of Zoology, 266, 133–151. https://doi.org/10.1017/S0952836905006643.
Courtenay, O., & Maffei, L. (2004). Crab-eating fox Cerdocyon thous. In C. Sillero-Zubiri, M. Hoffmann, & D. W. Macdonald (Eds.), Canids: Foxes, wolves, jackals and dogs. Status survey and conservation action plan (pp. 32–38). IUCN Species Programme: Gland.
Cox, P. G. (2007). A quantitative analysis of the eutherian orbit: Correlations with masticatory apparatus. Biological Reviews, 83(1), 35–69. https://doi.org/10.1111/j.1469-185X.2007.00031.x.
Crespo, J. A. (1971). Ecología del zorro gris Dusicyon gymnocercus antiquus (Ameghino) en la provincia de La Pampa. Revista del Museo Argentino de Ciencias Naturales “Bernardino Rivadavia”, 5, 1–205.
Crespo, J. A., & De Carlo, J. M. (1963). Estudio ecológico de una población de zorros colorados Dusicyon culpaeus. Revista del Museo Argentino de Ciencias Naturales “Bernardino Rivadavia”, 1, 1–55.
Dalponte, J. C. (2009). Lycalopex vetulus (Carnivora: Canidae). Mammalian Species, 847, 1–7. https://doi.org/10.1644/847.1.
Dalponte, J., & Courtenay, O. (2004). Hoary fox Pseudalopex vetulus. In C. Sillero-Zubiri, M. Hoffmann, & D. W. Macdonald (Eds.), Canids: Foxes, wolves, jackals and dogs. Status survey and conservation action plan (pp. 72–76). IUCN Species Programme: Gland.
Damasceno, E. M., Hingst-Zaher, E., & Astúa, D. (2013). Bite force and encephalization in the Canidae (Mammalia: Carnivora). Journal of Zoology, 290, 246–254. https://doi.org/10.1111/jzo.12030.
Drake, A. G. (2011). Dispelling dog dogma: An investigation of heterochrony in dogs using 3D geometric morphometric analysis of skull shape: Investigation of heterochrony in dogs. Evolution & Development, 13, 204–213. https://doi.org/10.1111/j.1525-142X.2011.00470.x.
Dryden, I. L., & Mardia, K. V. (1998). Statistical shape analysis. Chichester: Wiley.
Duckler, G. L. (1998). An unusual osteological formation in the posterior skulls of captive tigers (Panthera tigris). Zoo Biology, 17, 135–142. https://doi.org/10.1002/(SICI)1098-2361(1998)17:2<135::AID-ZOO8>3.0.CO;2-A.
Eizirik, E. (2012). A molecular view on the evolutionary history and biogeography of Neotropical carnivores (Mammalia, Carnivora). In B. D. Patterson & L. P. Costa (Eds.), Bones, clones, and biomes: The history and geography of recent Neotropical mammals (pp. 123–142). Chicago: The University of Chicago Press.
Emerson, S. B., & Bramble, D. M. (1993). Scaling, allometry and skull design. In J. Hanken & B. K. Hall (Eds.), The skull (pp. 384–416). Chicago: The University of Chicago Press.
Evans, H. E. (1993). Miller’s anatomy of the dog (3rd ed.). Philadelphia: W. B. Saunders Company.
Ewer, R. F. (1973). The carnivores. Ithaca: Cornell University Press.
Figueirido, B., MacLeod, N., Krieger, J., De Renzi, M., Pérez-Claros, J. A., & Palmqvist, P. (2011). Constraint and adaptation in the evolution of carnivoran skull shape. Paleobiology, 37, 490–518. https://doi.org/10.1666/09062.1.
Flores, D. A., Giannini, N., & Abdala, F. (2018). Evolution of post-weaning skull ontogeny in New World opossums (Didelphidae). Organisms Diversity & Evolution, 18, 367–382. https://doi.org/10.1007/s13127-018-0369-3.
Forbes-Harper, J. L., Crawford, H. M., Dundas, S. J., Warburton, N. M., Adams, P. J., Bateman, P. W., et al. (2017). Diet and bite force in red foxes: Ontogenetic and sex differences in an invasive carnivore. Journal of Zoology, 303, 54–63. https://doi.org/10.1111/jzo.12463.
Fuentes-González, J. A., & Muñoz-Durán, J. (2017). Comportamiento social y preferencias alimentarias en la evolución de los cánidos actuales. Revista Mexicana de Biodiversidad, 88, 192–206. https://doi.org/10.1016/j.rmb.2017.01.022.
Fuller, T. K., & Cypher, B. L. (2004). Gray fox Urocyon cinereoargenteus. In C. Sillero-Zubiri, M. Hoffmann, & D. W. Macdonald (Eds.), Canids: Foxes, wolves, jackals and dogs. Status survey and conservation action plan (pp. 92–97). IUCN Species Programme: Gland.
Galatius, A., Berta, A., Frandsen, M. S., & Goodall, R. N. P. (2011). Interspecific variation of ontogeny and skull shape among porpoises (Phocoenidae). Journal of Morphology, 272, 136–148. https://doi.org/10.1002/jmor.10900.
García-Perea, R. (1996). Patterns of postnatal development in skull of lynxes, Genus Lynx (Mammalia: Carnivora). Journal of Morphology, 229, 241–254. https://doi.org/10.1002/(SICI)1097-4687(199609)229:3<241::AID-JMOR1>3.0.CO;2-1.
German, R. Z., & Crompton, A. W. (1996). Ontogeny of suckling mechanisms in opossums (Didelphis virginiana). Brain, Behavior and Evolution, 48, 157–164. https://doi.org/10.1159/000113194.
German, R. Z., Crompton, A. W., Levitch, L. C., & Thexton, A. J. (1992). The mechanism of suckling in two species of infant mammal: Miniature pigs and long-tailed macaques. Journal of Experimental Zoology, 261, 322–330. https://doi.org/10.1002/jez.1402610311.
Giannini, N. P., Segura, V., Giannini, M. I., & Flores, D. (2010). A quantitative approach to the cranial ontogeny of the puma. Mammalian Biology, 75, 547–554. https://doi.org/10.1016/j.mambio.2009.08.001.
Gipson, P. S., Ballard, W. B., Nowak, R. M., & Mech, L. D. (2000). Accuracy and precision of estimating age of gray wolves by tooth wear. Journal of Wildlife Management, 64, 752–758 http://digitalcommons.unl.edu/usgsnpwrc/400.
Gittleman, J. L. (1985). Carnivore body size: Ecological and taxonomic correlates. Oecologia, 67, 540–554. https://doi.org/10.1007/BF00790026.
Goloboff, P. A., & Catalano, S. A. (2016). TNT version 1.5, including a full implementation of phylogenetic morphometrics. Cladistics, 32, 221–238. https://doi.org/10.1111/cla.12160.
Goloboff, P. A., Mattoni, C. I., & Quinteros, A. S. (2006). Continuous characters analyzed as such. Cladistics, 22, 589–601. https://doi.org/10.1111/j.1096-0031.2006.00122.x.
González del Solar, R., & Rau, J. (2004). Chilla Pseudalopex griseus. In C. Sillero-Zubiri, M. Hoffmann, & D. W. Macdonald (Eds.), Canids: Foxes, wolves, jackals and dogs. Status survey and conservation action plan (pp. 56–63). IUCN Species Programme: Gland.
Goodall, C. (1991). Procrustes methods in the statistical analysis of shape. Journal of the Royal Statistical Society: Series B (Methodological), 53, 285–321. https://doi.org/10.1111/j.2517-6161.1991.tb01825.x.
Goodwin, E. A., & Ballard, W. B. (1985). Use of tooth cementum for age determination of gray wolves. The Journal of Wildlife Management, 49, 313. https://doi.org/10.2307/3801522.
Goswami, A. (2006). Morphological integration in the carnivoran skull. Evolution, 60, 169–183. https://doi.org/10.1111/j.0014-3820.2006.tb01091.x.
Goswami, A., Milne, N., & Wroe, S. (2010). Biting through constraints: cranial morphology, disparity and convergence across living and fossil carnivorous mammals. Proceedings of the Royal Society B: Biological Sciences, 278, 1831–1839. https://doi.org/10.1098/rspb.2010.2031.
Grue, H., & Jensen, B. (1973). Annular structures in canine tooth cementum in red foxes. Vulpes vulpes. Danish Review of Game Biology, 8, 1–16.
Guzmán, J. A., & Ortiz, J. C. (2009). Variación geográfica del zorro Lycalopex culpaeus (Mammalia, Canidae) en Chile: implicaciones taxonómicas. Revista de Biología Tropical, 57, 421–432.
Hammer, Ø., Harper, D. A. T., & Ryan, P. D. (2001). PAST: Paleontological statistics software package for education and data analysis. Palaeontologia Electronica, 4, 1–9 http://palaeo-electronica.org/2001_1/past/past.pdf.
Harris, S. (1978). Age determination in the red fox (Vulpes vulpes)-an evaluation of technique efficiency as applied to a sample of suburban foxes. Journal of Zoology, 184, 91–117. https://doi.org/10.1111/j.1469-7998.1978.tb03268.x.
Helm, J. W., & German, R. Z. (1996). The epigenetic impact of weaning on craniofacial morphology during growth. The Journal of Experimental Zoology, 276, 243–253. https://doi.org/10.1002/(SICI)1097-010X(19961101)276:4<243::AID-JEZ1>3.0.CO;2-O.
Hildebrand, M. (1995). Analysis of vertebrate structure. Hoboken: Wiley.
Holliday, J. A., & Steppan, S. J. (2004). Evolution of hypercarnivory: The effect of specialization on morphological and taxonomic diversity. Paleobiology, 30, 108–128. https://doi.org/10.1666/0094-8373(2004)030<0108:EOHTEO>2.0.CO;2.
Jantschke, F. (1973). On the breeding and rearing of bush dogs Speothos venaticus at Frankfurt Zoo. International Zoo Yearbook, 13, 141–143. https://doi.org/10.1111/j.1748-1090.1973.tb02130.x.
Jiménez, J. E., & McMahon, E. (2004). Darwin’s fox Pseudalopex fulvipes. In C. Sillero-Zubiri, M. Hoffmann, & D. W. Macdonald (Eds.), Canids: Foxes, wolves, jackals and dogs. Status survey and conservation action plan (pp. 50–55). IUCN Species Programme: Gland.
Jiménez, J. E., & Novaro, A. J. (2004). Culpeo Pseudalopex culpaeus. In C. Sillero-Zubiri, M. Hoffmann, & D. W. Macdonald (Eds.), Canids: Foxes, wolves, jackals and dogs. Status survey and conservation action plan (pp. 44–49). IUCN Species Programme: Gland.
Juarez, K. M., & Marinho-Filho, J. (2002). Diet, habitat use, and home ranges of sympatric canids in Central Brazil. Journal of Mammalogy, 83, 925–933. https://doi.org/10.1644/1545-1542(2002)083<0925:DHUAHR>2.0.CO;2.
Klingenberg, C. P. (1998). Heterochrony and allometry: The analysis of evolutionary change in ontogeny. Biological Reviews, 73, 79–123. https://doi.org/10.1017/S000632319800512X.
Klingenberg, C. P. (2011). MorphoJ: An integrated software package for geometric morphometrics. Molecular Ecology Resources, 11, 353–357. https://doi.org/10.1111/j.1755-0998.2010.02924.x.
Knowlton, F. F., & Whittemore, S. L. (2001). Pulp cavity-tooth width ratios from known-age and wild-caught coyotes determined by radiography. Wildlife Society Bulletin, 239, 244 www.jstor.org/stable/3784003.
Kremenak, C. R. (1969). Dental eruption chronology in dogs: Deciduous tooth gingival emergence. Journal of Dental Research, 48, 1177–1184. https://doi.org/10.1177/00220345690480061501.
Kremenak, C. R., Russell, L. S., & Christensen, R. D. (1969). Tooth-eruption ages in suckling dogs as affected by local heating. Journal of Dental Research, 48, 427–430. https://doi.org/10.1177/00220345690480061501.
La Croix, S., Holekamp, K. E., Shivik, J. A., Lundrigan, B. L., & Zelditch, M. L. (2011a). Ontogenetic relationships between cranium and mandible in coyotes and hyenas. Journal of Morphology, 272, 662–674. https://doi.org/10.1002/jmor.10934.
La Croix, S., Zelditch, M. L., Shivik, J. A., Lundrigan, B. L., & Holekamp, K. E. (2011b). Ontogeny of feeding performance and biomechanics in coyotes. Journal of Zoology, 285, 301–315. https://doi.org/10.1111/j.1469-7998.2011.00847.x.
Landon, D. B., Waite, C. A., Peterson, R. O., & Mech, L. D. (1998). Evaluation of age determination techniques for gray wolves. The Journal of Wildlife Management, 62, 674. https://doi.org/10.2307/3802343.
Leite Pitman, M. R. P., & Williams, R. S. R. (2004). Short-eared dog Atelocynus microtis. In C. Sillero-Zubiri, M. Hoffmann, & D. W. Macdonald (Eds.), Canids: Foxes, wolves, jackals and dogs. Status survey and conservation action plan (pp. 26–31). IUCN Species Programme: Gland.
Linhart, S. B. (1968). Dentition and pelage in the juvenile red fox (Vulpes vulpes). Journal of Mammalogy, 49, 526. https://doi.org/10.2307/1378217.
Linhart, S. B., & Knowlton, F. F. (1967). Determining age of coyotes by tooth cementum layers. The Journal of Wildlife Management, 31, 362. https://doi.org/10.2307/3798334.
Lombaard, L. J. (1971). Age determination and growth curves in the black-backed jackal, Canis mesomelas Schreber, 1775 (Carnivora: Canidae). Annals of the Transvaal Museum, 27, 135–169.
Losos, J. B., & Miles, D. B. (2002). Testing the hypothesis that a clade has adaptively radiated: Iguanid lizard clades as a case study. The American Naturalist, 160, 147–157. https://doi.org/10.1086/341557.
Lucherini, M., Pessino, M., & Farias, A. A. (2004). Pampas fox Pseudalopex gymnocercus. In C. Sillero-Zubiri, M. Hoffmann, & D. W. Macdonald (Eds.), Canids: Foxes, wolves, jackals and dogs. Status survey and conservation action plan (pp. 63–68). IUCN Species Programme: Gland.
Machado, F. D. A., & Hingst-Zaher, E. (2009). Investigating south American biogeographic history using patterns of skull shape variation on Cerdocyon thous (Mammalia: Canidae). Biological Journal of the Linnean Society, 98, 77–84. https://doi.org/10.1111/j.1095-8312.2009.01274.x.
Machado, F. A., Zahn, T. M. G., & Marroig, G. (2018). Evolution of morphological integration in the skull of Carnivora (Mammalia): Changes in Canidae lead to increased evolutionary potential of facial traits. Evolution, 72, 1399–1419. https://doi.org/10.1111/evo.13495.
Martinez, P. A., Marti, D. A., Molina, W. F., & Bidau, C. J. (2013). Bergmann’s rule across the equator: A case study in Cerdocyon thous (Canidae). Journal of Animal Ecology, 82, 997–1008. https://doi.org/10.1111/1365-2656.12076.
McNamara, K. J. (2012). Heterochrony: The evolution of development. Evolution: Education and Outreach, 5, 203–218. https://doi.org/10.1007/s12052-012-0420-3.
Meloro, C., & O’Higgins, P. (2011). Ecological adaptations of mandibular form in fissiped Carnivora. Journal of Mammalian Evolution, 18, 185–200. https://doi.org/10.1007/s10914-011-9156-z.
Mitteroecker, P., Gunz, P., Bernhard, M., Schaefer, K., & Bookstein, F. L. (2004). Comparison of cranial ontogenetic trajectories among great apes and humans. Journal of Human Evolution, 46, 679–698. https://doi.org/10.1016/j.jhevol.2004.03.006.
Moore, W. J. (1981). The mammalian skull. Cambridge: Cambridge University Press.
Morales, M. M., & Giannini, N. P. (2010). Morphofunctional patterns in Neotropical felids: Species co-existence and historical assembly. Biological Journal of the Linnean Society, 100, 711–724. https://doi.org/10.1111/j.1095-8312.2010.01461.x.
Muñoz, N. A., Cassini, G. H., Candela, A. M., & Vizcaíno, S. F. (2017). Ulnar articular surface 3-D landmarks and ecomorphology of small mammals: A case study of two early Miocene typotheres (Notoungulata) from Patagonia. Earth and Environmental Science Transactions of the Royal Society of Edinburgh, 106, 315–323. https://doi.org/10.1017/S1755691016000141.
Nowak, R. M. (2005). Walker’s carnivores of the world. Baltimore: Johns Hopkins University Press.
Perez, S. I., Klaczko, J., Rocatti, G., & Dos Reis, S. F. (2011). Patterns of cranial shape diversification during the phylogenetic branching process of New World monkeys (Primates: Platyrrhini): Cranial shape diversification of Platyrrhini. Journal of Evolutionary Biology, 24, 1826–1835. https://doi.org/10.1111/j.1420-9101.2011.02309.x.
Perini, F. A., Russo, C. A. M., & Schrago, C. G. (2010). The evolution of south American endemic canids: A history of rapid diversification and morphological parallelism. Journal of Evolutionary Biology, 23, 311–322. https://doi.org/10.1111/j.1420-9101.2009.01901.x.
Prestrud, P., & Nilssen, K. (1995). Growth, size, and sexual dimorphism in Arctic foxes. Journal of Mammalogy, 76, 522–530. https://doi.org/10.2307/1382360.
Prevosti, F. J. (2006). Grandes cánidos (Carnivora, Canidae) del Cuaternario de la República Argentina: Sistemática, Filogenia, Bioestratigrafía y Paleoecología. Doctoral thesis, Universidad Nacional de La Plata, Facultad de Ciencias Naturales y Museo.
Prevosti, F. J. (2010). Phylogeny of the large extinct South American Canids (Mammalia, Carnivora, Canidae) using a “total evidence” approach. Cladistics, 26, 456–481. https://doi.org/10.1111/j.1096-0031.2009.00298.x.
Prevosti, F. J., & Forasiepi, A. M. (2018). Evolution of South American mammalian predators during the Cenozoic: Paleobiogeographic and paleoenvironmental contingencies. Cham: Springer Nature.
Prevosti, F. J., & Lamas, L. (2006). Variation of cranial and dental measurements and dental correlations in the pampean fox (Dusicyon gymnocercus). Journal of Zoology, 270, 636–649. https://doi.org/10.1111/j.1469-7998.2006.00187.x.
Prevosti, F. J., & Rincón, A. D. (2007). A new fossil canid assemblage from the late Pleistocene of Northern South America: The canids of the Inciarte asphalt pit (Zulia, Venezuela), fossil record and biogeography. Journal of Paleontology, 81, 1053–1065. https://doi.org/10.1666/pleo05-143.1.
Prevosti, F., & Soibelzon, L. H. (2012). Evolution of the south American carnivores (Mammalia, Carnivora): A paleontological perspective. In B. D. Patterson & L. P. Costa (Eds.), Bones, clones, and biomes: The history and geography of recent Neotropical mammals (pp. 102–122). Chicago: Oxford University Press.
Prevosti, F. J., Segura, V., Cassini, G. H., & Martin, G. M. (2013). Revision of the systematic status of Patagonian and Pampean gray foxes (Canidae: Lycalopex griseus and Lycalopex gymnocercus) using 3 D Geometric Morphometrics. Mastozoología Neotropical, 20, 289–300.
R Development Core Team (2018). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. Available in: https://www.r-project.org/
Radinsky, L. B. (1981). Evolution of skull shape in carnivores: 1. Representative modern carnivores. Biological Journal of the Linnean Society, 15, 369–388. https://doi.org/10.1111/j.1095-8312.1981.tb00770.x.
Reilly, S. M., Wiley, E. O., & Meinhardt, D. J. (1997). An integrative approach to heterochrony: The distinction between interspecific and intraspecific phenomena. Biological Journal of the Linnean Society, 60, 119–143. https://doi.org/10.1111/j.1095-8312.1997.tb01487.x.
Rice, D. P. (2008). Craniofacial sutures development, disease and treatment (Frontiers of Oral Biology Vol. 12). Basel: Karger.
Richmond, F. J. R., Thomson, D. B., & Loeb, G. E. (1992). Electromyographic studies of neck muscles in the intact cat: I. Patterns of recruitment underlying posture and movement during natural behaviors. Experimental Brain Research, 88, 41–58. https://doi.org/10.1007/BF02259127.
Rodden, M., Rodrigues, F., & Bestelmeyer, S. (2004). Maned wolf Chrysocyon brachyurus. In C. Sillero-Zubiri, M. Hoffmann, & D. W. Macdonald (Eds.), Canids: Foxes, wolves, jackals and dogs. Status survey and conservation action plan (pp. 38–43). IUCN Species Programme: Gland.
Rohlf, F. J. (1999). Shape statistics: Procrustes method for the optimal superimposition of landmarks. Systematic Zoology, 39, 40–59. https://doi.org/10.1007/s003579900054.
Root, D. A., & Payne, N. F. (1984). Evaluation of techniques for aging gray fox. The Journal of Wildlife Management, 48, 926–933. https://doi.org/10.2307/3801440.
Sánchez-Villagra, M. R., Segura, V., Geiger, M., Heck, L., Veitschegger, K., & Flores, D. (2017). On the lack of a universal pattern associated with mammalian domestication: Differences in skull growth trajectories across phylogeny. Royal Society Open Science, 4, 170876. https://doi.org/10.1098/rsos.170876.
Schlager, S. (2017). Morpho and Rvcg – Shape analysis in R: R-packages for geometric Morphometrics, shape analysis and surface manipulations. In G. Zheng, S. Li, & G. Székely (Eds.), Statistical shape and deformation analysis (pp. 217–256). Elsevier: Academic Press.
Segura, V. (2013). Skull ontogeny of Lycalopex culpaeus (Carnivora: Canidae): Description of cranial traits and craniofacial sutures. Mammalia, 77(2), 205–214. https://doi.org/10.1515/mammalia-2012-0028.
Segura, V. (2015). A three-dimensional skull ontogeny in the bobcat (Lynx rufus) (Carnivora: Felidae): A comparison with other carnivores. Canadian Journal of Zoology, 93, 225–237. https://doi.org/10.1139/cjz-2014-0148.
Segura, V., & Flores, D. (2009). Aproximación cualitativa y aspectos funcionales en la ontogenia craneana de Puma concolor (Felidae). Mastozoología Neotropical, 16, 169–182.
Segura, V., & Prevosti, F. (2012). A quantitative approach to the cranial ontogeny of Lycalopex culpaeus (Carnivora: Canidae). Zoomorphology, 131, 79–92. https://doi.org/10.1007/s00435-012-0145-4.
Segura, V., Prevosti, F., & Cassini, G. (2013). Cranial ontogeny in the Puma lineage, Puma concolor, Herpailurus yagouaroundi, and Acinonyx jubatus (Carnivora: Felidae): A three-dimensional geometric morphometric approach. Zoological Journal of the Linnean Society, 169, 235–250. https://doi.org/10.1111/zoj.12047.
Segura, V., Cassini, G. H., & Prevosti, F. J. (2017). Three-dimensional cranial ontogeny in pantherines (Panthera leo, P. onca, P. pardus, P. tigris, Carnivora: Felidae). Biological Journal of the Linnean Society, 120, 210–227. https://doi.org/10.1111/bij.12888.
Sicuro, F. L. (2011). Evolutionary trends on extant cat skull morphology (Carnivora: Felidae): A three-dimensional geometrical approach. Biological Journal of the Linnean Society, 103, 176–190. https://doi.org/10.1111/j.1095-8312.2011.01636.x.
Sicuro, F. L., & Oliveira, L. F. B. (2011). Skull morphology and functionality of extant Felidae (Mammalia: Carnivora): A phylogenetic and evolutionary perspective. Zoological Journal of the Linnean Society, 161, 414–462. https://doi.org/10.1111/j.1096-3642.2010.00636.x.
Sillero-Zubiri, C. (2009). Family Canidae. In D. E. Wilson & R. A. Mittermeier (Eds.), Handbook of the mammals of the world (pp. 352–447). Barcelona: Lynx Editions.
Slater, G. J., & Van Valkenburgh, B. (2009). Allometry and performance: The evolution of skull form and function in felids. Journal of Evolutionary Biology, 22, 2278–2287. https://doi.org/10.1111/j.1420-9101.2009.01845.x.
Slater, G. J., Dumont, E. R., & Van Valkenburgh, B. (2009). Implications of predatory specialization for cranial form and function in canids. Journal of Zoology, 278, 181–188. https://doi.org/10.1111/j.1469-7998.2009.00567.x.
Smith, K. K. (1997). Comparative patterns of craniofacial development in eutherian and metatherian mammals. Evolution, 51, 1663–1678. https://doi.org/10.1111/j.1558-5646.1997.tb01489.x.
Soibelzon, L. H., & Prevosti, F. J. (2007). Los carnívoros (Carnivora, Mammalia) terrestres del Cuaternario de América del Sur. In G. X. Pons & D. Vicens (Eds.), Geomorfología litoral i Quaternari. Homenatge a Joan Cuerda Barceló (pp. 49–68). Societat d’Història Natural de les Balears: Palmas de Mallorca.
Tanner, J. B., Zelditch, M. L., Lundrigan, B. L., & Holekamp, K. E. (2009). Ontogenetic change in skull morphology and mechanical advantage in the spotted hyena (Crocuta crocuta). Journal of Morphology, 271(3), 353–365. https://doi.org/10.1002/jmor.10802.
Tchaicka, L., Freitas, T. R. O., Bager, A., Vidal, S. L., Lucherini, M., Iriarte, A., & Eizirik, E. (2016). Molecular assessment of the phylogeny and biogeography of a recently diversified endemic group of South American canids (Mammalia: Carnivora: Canidae). Genetics and Molecular Biology, 39, 442–451. https://doi.org/10.1590/1678-4685-GMB-2015-0189.
Tedford, R. H., Taylor, B. E., & Wang, X. (1995). Phylogeny of the Caninae (Carnivora, Canidae): The living taxa. American Museum Novitates, 3146.
Thexton, A. J., Crompton, A. W., Owerkowicz, T., & German, R. Z. (2004). Correlation between intraoral pressures and tongue movements in the suckling pig. Archives of Oral Biology, 49, 567–575. https://doi.org/10.1016/j.archoralbio.2004.02.002.
Thomason, J. J. (1991). Cranial strength in relation to estimated biting forces in some mammals. Canadian Journal of Zoology, 69, 2326–2333. https://doi.org/10.1139/z91-327.
Travaini, A., Juste, J., Novaro, A. J., & Capurro, A. F. (2000). Sexual dimorphism and sex identification in the South American culpeo fox, Pseudalopex culpaeus (Carnivora : Canidae). Wildlife Research, 27, 669. https://doi.org/10.1071/WR99064.
Tseng, Z. J., & Flynn, J. J. (2018). Structure-function covariation with nonfeeding ecological variables influences evolution of feeding specialization in Carnivora. Science Advances, 4, eaao5441. https://doi.org/10.1126/sciadv.aao5441.
Van Valkenburgh, B. (1989). Carnivore dental adaptations and diet: A study of trophic diversity within guilds. In J. L. Gittleman (Ed.), Carnivore behavior, ecology, and evolution (pp. 410–436). Boston: Springer.
Van Valkenburgh, B. (1991). Iterative evolution of hypercarnivory in canids (Mammalia: Carnivora): Evolutionary interactions among sympatric predators. Paleobiology, 17, 340–362. https://doi.org/10.1017/S0094837300010691.
Van Valkenburgh, B. (2007). Deja vu: The evolution of feeding morphologies in the Carnivora. Integrative and Comparative Biology, 47, 147–163. https://doi.org/10.1093/icb/icm016.
Van Valkenburgh, B., & Koepfli, K. P. (1993). Cranial and dental adaptations to predation in canids. In N. Dunstone & M. L. Gorman (Eds.), Mammals as predators (pp. 15–37). London: Symposia of the Zoological Society of London, Oxford University Press.
Van Valkenburgh, B., Theodor, J., Friscia, A., Pollack, A., & Rowe, T. (2004). Respiratory turbinates of canids and felids: A quantitative comparison. Journal of Zoology, 264, 281–293. https://doi.org/10.1017/S0952836904005771.
Wainwright, P. C., & Reilly, S. M. (1994). Ecological morphology. Chicago: University of Chicago Press.
Wall, C. E., & Smith, K. K. (2001). Ingestion in Mammals. In J. Wiley & Sons (Ed.), Encyclopedia of life sciences (p. a0001837). Chichester: Wiley.
Wang, X., & Tedford, R. H. (2007). Evolutionary history of canids. In P. Jensen (Ed.), The behavioural biology of dogs (pp. 3–20). London: CAB International.
Wang, X., Tedford, R. H., & Antón, M. (2008). Dogs: Their fossil relatives and evolutionary history. New York: Columbia University Press.
Wayne, R. K. (1986). Cranial morphology of domestic and wild canids: The influence of development on morphological change. Evolution, 40, 243–261. https://doi.org/10.1111/j.1558-5646.1986.tb00467.x.
Wayne, R. K., Van Valkenburgh, B., Kat, P. W., Fuller, T. K., Johnson, W. E., & O’Brien, S. J. (1989). Genetic and morphological divergence among sympatric canids. Journal of Heredity, 80, 447–454. https://doi.org/10.1093/oxfordjournals.jhered.a110896.
Wayne, R. K., Geffen, E., Girman, D. J., Koepfli, K. P., Lau, L. M., & Marshall, C. R. (1997). Molecular systematics of the Canidae. Systematic Biology, 46, 622–653. https://doi.org/10.1093/sysbio/46.4.622.
Werdelin, L. (1983). Morphological patterns in the skulls of cats. Biological Journal of the Linnean Society, 19, 375–391. https://doi.org/10.1111/j.1095-8312.1983.tb00793.x.
Werneburg, I., & Geiger, M. (2017). Ontogeny of domestic dogs and the developmental foundations of carnivoran domestication. Journal of Mammalian Evolution, 24, 323–343. https://doi.org/10.1007/s10914-016-9346-9.
Westneat, M. W. (2003). A biomechanical model for analysis of muscle force, power output and lower jaw motion in fishes. Journal of Theoretical Biology, 223, 269–281. https://doi.org/10.1016/S0022-5193(03)00058-4.
Wickland, C. R., Baker, J. F., & Paterson, B. W. (1991). Torque vectors of neck muscles in the cat. Experimental Brain Research, 84, 64–659. https://doi.org/10.1007/BF00230978.
Wiens, J. J., & Graham, C. H. (2005). Niche conservatism: Integrating evolution, ecology, and conservation biology. Annual Review of Ecology, Evolution, and Systematics, 36, 519–539. https://doi.org/10.1146/annurev.ecolsys.36.102803.095431.
Wilson, D. E., & Mittermeier, R. A. (2009). Handbook of the mammals of the world (Vol. 1. Carnivores). Barcelona: Lynx Editions.
Wood, J. E. (1958). Age structure and productivity of a gray fox population. Journal of Mammalogy, 39, 74–86. https://doi.org/10.2307/1376612.
Wozencraft, W. C. (2005). Order Carnivora. In D. E. Wilson & D. M. Reeder (Eds.), Mammal species of the world. A taxonomic and geographic reference (pp. 532–628). Baltimore: The Johns Hopkins University Press.
Wroe, S., & Milne, N. (2007). Convergence and remarkably consistent constraint in the evolution of carnivore skull shape: Convergence in carnivore skull shape. Evolution, 61, 1251–1260. https://doi.org/10.1111/j.1558-5646.2007.00101.x.
Zapata, S. C., Funes, M., & Novaro, A. (1997). Estimación de la edad en el zorro colorado patagónico (Pseudalopex culpaeus). Mastozoología Neotropical, 4, 145–150.
Zar, J. H. (2009). Biostatistical analysis. New Jersey: Prentice Hall.
Zelditch, M. L., & Carmichael, A. C. (1989). Growth and intensity of integration through postnatal growth in the skull of Sigmodon fulviventer. Journal of Mammalogy, 70, 477–484. https://doi.org/10.2307/1381419.
Zelditch, M. L., Lundrigan, B. L., & Garland, T. (2004). Developmental regulation of skull morphology. I. Ontogenetic dynamics of variance. Evolution and Development, 6, 194–206. https://doi.org/10.1111/j.1525-142X.2004.04025.x.
Zrzavý, J., Duda, P., Robovský, J., Okřinová, I., & Řičánková, V. P. (2018). Phylogeny of the Caninae (Carnivora): Combining morphology, behaviour, genes and fossils. Zoologica Scripta, 47, 373–389. https://doi.org/10.1111/zsc.12293.
Zuercher, G. L., Swarner, M., Silveira, L., & Carrillo, O. (2004). Bush dog Speothos venaticus. In C. Sillero-Zubiri, M. Hoffmann, & D. W. Macdonald (Eds.), Canids: Foxes, wolves, jackals and dogs. Status survey and conservation action plan (pp. 76–80). IUCN Species Programme: Gland.
Zunino, G. E., Vaccaro, O. B., Canevari, M., & Gardner, A. L. (1995). Taxonomy of the genus Lycalopex (Carnivora: Canidae) in Argentina. Proceedings of the Biological Society of Washington, 108, 729–747.
Zurano, J. P., Martinez, P. A., Canto-Hernandez, J., Montoya-Burgos, J. I., & Costa, G. C. (2017). Morphological and ecological divergence in south American canids. Journal of Biogeography, 44, 821–833. https://doi.org/10.1111/jbi.12984.
Acknowledgments
For access to collections under their care, we thank all the curators. This is a contribution to PICT 2015-2389, 2015-0966 and 2016-3151. We thank the anonymous reviewer who provided precise comments that considerably improved this article.
Funding
Préstamo BID PICT 2015-0966, and 2016-3151.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors report no potential conflict of interest.
Consent to participate and for publication
All authors have approved this submission and given consent for publication.
Ethical approval
No ethical approval was required.
Supplementary information
Below is the link to the electronic supplementary material.
Appendix 1 List of specimens used in this study
Appendix 1 List of specimens used in this study
Atelocynus microtis (N=23). AMNH: 76031; 76579; 95284; 95285; 98639; 100095. FMNH: 52491; 57836; 60674; 60675; 60676; 93955; 98080; 98081; 110949; 121286. MZUSP: 4320; 19750; 19751; 19752; 19753; 19754. NMNH: 361013.
Cerdocyon thous (N=163). AMNH: 14628; 14850; 17555; 21349; 23572; 61797; 63799; 132787; 133931; 135462; 205796; 205797; 205798; 205800; 205803; 205813; 205814; 205826. CFA: 3556; 3763; 3875; 3913; 4265; 4402; 4419; 4496; 4511; 4512; 4661; 4663; 5048; 5197; 5198; 5263; 5278; 5282; 5284; 5285; 5313; 5414; 6000; 6071; 6346. CML: 588; 3715; 3716; 3717; 3718; 3719; 3756; 3827; 4083; 4083; 4692; 5964; 5966; 5967; 5968; 6213; 6214; 6339; 6340; 6341. GECM: 154. MACN: 4.213; 17.122; 20.32; 20.34; 23.45; 24.127; 25.119; 25.159; 29.839; 30.344; 30.345; 32.261; 32.262; 32.75; 32.93; 33.6; 34.676; 36.191; 36.481; 43.26; 44.11; 45.34; 45.40; 47.116; 47.189; 47.190; 47.191; 47.192; 47.193; 47.402; 48.3; 48.5; 48.6; 48.7; 48.10; 49.367; 50.40; 50.43; 50.45; 50.57; 50.59; 50.60; 50.61; 50.62; 50.63; 50.64; 50.65; 50.66; 52.54; 52.63; 52.64; 13051; 14322; 14681; 15741; 16189; 19071; 20316; 20454; 20455; 20456; 20815; 20816; 20817; 21228; 23180; 23669; 23670; 23726; 23727; 24045; 24202; 24264; 24267. MFA-ZV: 228; 1204; 1205. MLP: 16.X.01.7; 20.IX.49.13; 1322. MZUSP: 19755; 2922; 32617; 3486; 4289; 856; 9687. NMNH: 123342; 137512; 143813; 238670; 239965; 241404; 253824; 270367; 281496; 372840; 372844; 372847.
Chrysocyon brachyurus (N=85). AMNH: 120999; 133940; 133941; 135274. CFA: 12826; 12827. CML: 1376; 6352; 6353. FMNH: 28311; 28312; 28313; 44534; 46003; 96003; 99213; 101848; 104088; 125401; 127434; 134483; 137425; 150739. MACN: 3.71; 3.73; 4.32; 4.303; 4.385; 24.4; 30.198; 30.29; 53.49; 13466; 19146; 20646; 23456; 23983; 23984; 24043; 24201. MFA-ZV: 185; 273; 517; 524; 553; 568; 581; 651; 652; 919; 1166. MLP: 2.IV.02.4; 5.X.99.1; 6; 92; 564; 695; 1684; 1686. MZUSP: 525; 3025; 3090; 3338; 3700; 9420; 19733; 19736; 19745; 28870; 31981; 32039; 32042; 32043; 32056; 32199; 32505; 32629. NMNH: 196975; 261022; 261023; 270371; 271567; 314863; 534807; 588223.
Lycalopex culpaeus (N=143). CFA: 4229; 6451. CML: 5067; 5068; 5069; 5070; 5071; 5970; 5974; 6343; 6344. LIEB: 791; 792. MACN: 3.68; 4.41; 4.211; 7.42; 25.128; 27.131; 30.69; 31.58; 31.59; 33.67; 33.68; 36.231; 38.39; 41.55; 15022; 15024; 15025; 15028; 15033; 15037; 15040; 15044; 15045; 15049; 15049; 15050; 15055; 15062; 15063; 15064; 15073; 15078; 15081; 15082; 15083; 15089; 15093; 15096; 15101; 15106; 15112; 15119; 15121; 15122; 15123; 15124; 15127; 15129; 15130; 15131; 15132; 15133; 15138; 15140; 15149; 15151; 15154; 15158; 15163; 15168; 15172; 15173; 15177; 15181; 15182; 15190; 15194; 15196; 15197; 15199; 15200; 15201; 15202; 15203; 15208; 15212; 15220; 15223; 15224; 15226; 15227; 15228; 15229; 15232; 15233; 15240; 15243; 15246; 15248; 15258; 15259; 15260; 15261; 15266; 15267; 15268; 19221; 19222; 20813; 21899; 23072; 23076; 23077; 23093; 23095; 23098; 23099; 23100; 23101; 23102; 23103; 23104; 23108; 23119; 23123; 23125; 23143; 23148; 23720; 23721; 23841; 23915; 23916; 24.119; 24047; 24210; 24270; 24271. MLP: 15.V.96.2; 1264; 1266.
Lycalopex fulvipes (N=2). FMNH: 23814; 23815.
Lycalopex griseus (N=137). AMNH: 14081; 17440a; 17440b; 17441a. CFA: 2175; 2197; 4197; 5291; 5649; 5777; 5782; 10243. CML: 837; 838; 1177; 1178; 1489; 3714; 4967; 6189; 6190; 6191. FMNH: 154639; 154640. LIEB: 794; 809. MACN: 2.38; 2.39; 3.63; 4.253; 7.40; 7.41; 23.19; 23.20; 24.50; 24.51; 24.52; 24.53; 24.54; 24.55; 24.63; 24.66; 24.76; 24.78; 24.79; 24.80; 24.81; 32.263; 50.419; 50.420; 50.432; 50.490; 51.170; 53.42; 223; 224; 225; 226; 319; 13781; 14540; 14902; 15020; 15185; 15186; 15189; 15262; 15263; 15264; 15265; 15269; 16321; 16322; 16325; 20205; 20206; 20207; 20208; 20276; 20277; 20278; 20814; 20829; 23150; 23662; 23663; 23664; 23668; 23718; 23728; 23729; 23730; 23919; 24206; 24251; 24256; 24257; 24258; 24262; 24263. MLP: 2.IV.60.1; 4.VIII.98.4; 5.III.36.27; 27.XII.01.27; 240; 441; 559; 696; 701; 712. NMNH: 482163; 482164; 92139; 92140; 92141; 92142; 92143; 92144; 92145; 92146; 92147; 92149; 92150; 92151; 92152; 92158; 92159; 92160; 92161; 92162; 92169; 92170; 92173; 92175; 92176; 92177; 92178; 92179.
Lycalopex gymnocercus (N=374). AMNH: 41502; 41503; 41504; 41505; 41506; 41507; 41508; 41509; 41510. CFA: 3255; 3698; 3962; 4256; 4416; 4417; 4659; 5219; 5280; 8588; 8589; 11062. CML: 192; 236; 495; 545; 645; 834; 836; 895; 908; 909; 959; 1179; 1526; 1526; 1529; 1558; 3072; 4081; 4082; 5143; 5473; 5474; 5479; 5480; 5772; 6342. GECM: 24; 34; 40; 57; 65; 67; 75; 76; 85; 100; 108; 112; 119; 121; 129; 139; 149; 152; 153; 179; 217Bis; 227Bis; 220Bis. MACN: 2.32; 3.10; 4.5; 4.54; 4.271; 4.290; 4.299; 20.33; 20.35; 23.33; 23.37; 23.38; 24.47; 24.48; 24.49; 24.133; 24.134; 24.140; 24.141; 24.143; 24.144; 24.148; 24.156; 24.162; 24.169; 24.171; 25.27; 26.6; 26.28; 26.87; 26.129; 26.162; 26.163; 27.53; 28.182; 29.35; 30.144; 30.150; 30.210; 30.211; 32.252; 32.263; 33.177; 33.266; 33.268; 34.317; 35.241; 35.407; 36.479; 36.480; 37.82; 38.243; 39.191; 39.194; 41.221; 44.17; 48.266; 49.134; 49.139; 49.148; 49.149; 49.159; 49.160; 49.167; 50.56; 50.443; 50.491; 50.492; 50.494; 50.495; 50.497; 50.498; 50.500; 50.501; 50.502; 50.503; 50.504; 50.505; 53.2; 53.6; 54.133; 246; 285; 293; 13299; 13313; 13327; 13331; 13337; 14319; 14323; 14386; 14409; 15363; 15364; 15387; 15388; 15389; 15390; 15601; 15692; 15742; 15748; 15749; 15750; 15751; 15752; 15754; 15757; 15758; 15760; 15761; 15762; 15764; 15765; 15766; 15769; 15771; 15783; 15784; 15785; 15787; 15788; 15789; 15791; 15792; 15794; 15795; 15796; 15797; 15800; 15818; 15820; 15831; 15833; 15834; 15838; 15854; 15856; 15858; 15859; 15860; 15861; 15862; 15863; 15864; 15865; 15866; 15867; 15868; 15869; 15870; 15871; 15873; 15875; 15879; 15882; 15888; 15892; 15894; 15895; 15896; 15898; 15901; 15902; 15906; 15908; 15909; 15917; 15932; 15933; 15934; 15938; 15941; 15954; 15958; 15963; 15964; 15966; 15970; 15973; 15979; 15981; 15982; 15986; 15987; 15992; 15998; 15999; 16000; 16001; 16002; 16003; 16004; 16005; 16006; 16007; 16009; 16010; 16013; 16014; 16015; 16024; 16025; 16026; 16027; 16030; 16031; 16032; 16035; 16036; 16037; 16038; 16039; 16040; 16041; 16046; 16047; 16048; 16049; 16050; 16053; 16055; 16059; 16062; 16063; 16066; 16068; 16074; 16077; 16079; 16080; 16083; 16085; 16088; 16094; 16096; 16097; 16099; 16100; 16101; 16102; 16103; 16104; 16105; 16106; 16107; 16108; 16110; 16111; 16115; 16117; 16118; 16120; 16122; 16123; 16130; 16131; 16139; 16143; 16145; 16149; 16151; 22936; 23153; 23154; 23155; 23156; 23157; 23158; 23290; 23910; 23913; 23920; 24203; 24204; 24205; 24208; 24209; 24259; 24265; 24266; 24282. MLP: 16.III.99.16; 20.IV.95.3; 30.III.99.13; 13.IV.99.3; 13.IV.99.13; 13.IV.99.14; 13.IV.99.19; 13.IV.99.20; 13.IV.99.31; 13.IV.99.35; 13.IV.99.36; 13.IV.99.44; 26.V.95.9; 4.VIII.98.9; 30.XII.02.65; 442; 710. NMNH: 172789; 172790; 236362; 236366; 331065.
Lycalopex sechurae (N=39). AMNH: 46525; 46527; 46528; 46529; 46530; 46531; 46532; 46533; 63709. FMNH: 19541; 19971; 19972; 21129; 41237; 53911; 53912; 53913; 80953; 80954; 80955; 80956; 80957; 80958; 80959; 80960; 80961; 80962; 80963; 80964; 80965; 80966; 80967; 80968; 80969. NMNH: 121171; 121172; 181150; 545109; 545110.
Lycalopex vetulus (N=34). AMNH: 349; 391; 70091; 100091; 100100; 133926; 133927; 133928; 133929; 133937; 245699. FMNH: 20747. MACN: 3.17. MLP: 1258. MZUSP: 825; 1011; 1012; 1014; 1015; 1016; 1018; 1075; 1076; 1084; 2170; 2479; 3046; 3047; 3047; 3048; 3049; 3050; 12040; 13611.
Speothos venaticus (N=43). AMNH: 37472; 76035; 76805; 76806; 98558; 98559; 98560; 98640; 136284; 136285; 167846; 175306; 184688; 202839. FMNH: 60290; 60579; 87861; 121544; 125402. MACN 33.154; 50.67; 16510. MZUSP: 19743; 19744. NMNH: 179047; 179048; 253504; 269135; 270165; 270171; 270368; 270369; 270370; 307650; 314048; 395841; 398030; 521045; 538307; 544414; 546298; 582465; 583255.
Urocyon cinereoargenteus (N=92). AMNH: 6405; 8197; 19207; 26012; 26013; 100301; 120989; 121664; 127561; 127573; 127575; 127576; 183939; 183942; 183943; 183953; 183954; 183956; 183960; 183973; 183979; 183991; 183995; 184002; 184007; 184009; 184012; 184013; 184014; 184064; 184065; 184068; 184077; 184080; 184083; 184087; 184091; 184092; 184094; 184097; 184098; 184105; 184117; 184121; 184122; 184126; 184132; 185512; 203066; 243095; 243449; 254470; 255645; 255646; 255648; 255649. NMNH: 14552; 14553; 21391; 29287; 34860; 35531; 54991; 55501; 61207; 68193; 73466; 76424; 91173; 91174; 98305; 107209; 117637; 117640; 130065; 133191; 151224; 151237; 180262; 180740; 188094; 203121; 215376; 215377; 215378; 265932; 265933; 271091; 287505; 507119; 529675; 569080.
Canis lupus (N=11). AMNH: 16848; 18215; 18218; 18238; 19124; 34468. MACN: 3.76; 23.12; 23.15; 35.210. MLP: 1031.
Vulpes vulpes (N=81). AMNH: 3796; 6412; 6413; 6414; 6415; 6416; 19024; 19841; 59311; 60131; 60132; 70165; 80061; 85032; 85033; 90799; 146724; 147569; 150299; 150300; 242479. FMNH: 74469; 74472; 74987; 74988; 74989; 75644; 75645; 75646; 77130; 77136; 78650; 78651; 80827; 80829; 80836; 80837; 80839; 80840; 84697; 84698; 85216; 85217; 85218; 86820; 89369; 89370; 89371; 89372; 89587; 89595; 89710; 89712; 89963; 90361; 90473; 90474; 91605; 91725; 91726; 91731; 91741; 92264; 92265; 92267; 92636; 92727; 95863; 95865; 95867; 95870; 95872; 95873; 98733; 98734; 98735; 98736; 106726; 107271; 140172; 140176.
Rights and permissions
About this article
Cite this article
Segura, V., Cassini, G.H. & Prevosti, F.J. Evolution of Cranial Ontogeny in South American Canids (Carnivora: Canidae). Evol Biol 48, 170–189 (2021). https://doi.org/10.1007/s11692-020-09529-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11692-020-09529-3



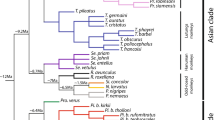



 Atelocynus microtis,
Atelocynus microtis,  Cerdocyon thous,
Cerdocyon thous,  Chrysocyon brachyurus,
Chrysocyon brachyurus,  Lycalopex culpaeus,
Lycalopex culpaeus,  Lycalopex fulvipes,
Lycalopex fulvipes,  Lycalopex griseus,
Lycalopex griseus,  Lycalopex gymnocercus,
Lycalopex gymnocercus,  Lycalopex sechurae,
Lycalopex sechurae,  Lycalopex vetulus,
Lycalopex vetulus,  Speothos venaticus,
Speothos venaticus,  Urocyon cinereoargenteus
Urocyon cinereoargenteus


 Size shift in J1,
Size shift in J1,  Shape shift in J1,
Shape shift in J1, Size shift in A3,
Size shift in A3,  Shape shift in A3
Shape shift in A3
 , Chrysocyon brachyurus;
, Chrysocyon brachyurus; , Speothos venaticus;
, Speothos venaticus; , Cerdocyon thous;
, Cerdocyon thous; , Urocyon cinereoargenteus taken from literature: Brady
, Urocyon cinereoargenteus taken from literature: Brady