Abstract
The present study describes the floristic composition and dominance pattern of sal forests in Ranchi, Jharkhand, eastern India. Vegetation was studied in 47 belt transects (50 × 100 m) that had 137 plant species (110 identified and 27 unidentified) belonging to 51 families. The family Fabaceae with 17 species (8 spp. belonging to subfamily Faboideae, 6 spp. to Caesalpinioideae and 3 spp. to Mimosoidieae) contributed the most to diversity, followed by Rubiaceae (8 spp.) and Euphorbiaceae (6 spp.). Tree density in sal was inversely related to species richness. Total tree density (≥ 10 cm GBH) in the studied forests was 397 individuals ha−1, with a basal cover of 262.50 m2 ha−1. Important plants of conservation concern are Pterocarpus marsupium Roxb, Andrographis paniculata (Burm.f.) Wall, Sterculiaurens Roxb., Tinospora cordifolia (Willd.) Miers, and Asparagus densiflorus (Kunth) Jessop. Phanerophytes had the highest percentage (71%, with percentage deviation from normal life form of + 25) followed by therophyte (15%, with percentage deviation + 2). Observed percentage deviation from normal life form was much lower (with + 2 percentage deviation) in both chamaephytes (8%) and therophytes (15%), suggesting that the studied sal forests are favorable for supporting various plants species. The forest management strategies should focus on the increasing demands for different timber and non-timber forest products to conserve the plant diversity of these natural forests.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sal (Shorea robusta C.F. Gaertn.) is a tropical tree species native to South Asia from Myanmar in the east to Bangladesh, Nepal and India in the west and covers more than 12 million hectares of forest area (Tiwari 1995). Although, the general distribution of sal is governed by climate, its local distribution is regulated largely by geological conditions and soil (Troup1921). On the basis of edaphic conditions and microclimate, the phenology of sal forests ranges from deciduous to evergreen and extends from tropical to subtropical. Sal has been described as deciduous (Cooke 1958; Kirtikar and Basu 1975; Tiwari 1995), semi-deciduous (Bor 1953), evergreen (Krishnaswamy and Mathauda 1954; Singh and Singh 1992; Borchert 2000) or deciduous or borderline between evergreen and deciduous (Joshi 1980). In addition to timber and fuel wood, sal produces fodder (Panday 1982; Gautam 1990; Pandey and Yadama 1990; Mathema 1991; Upadhyay 1992; Thacker and Gautam 1994; Fox 1995; Shakya and Bhattarai 1995; Edwards 1996; Gautam and Devkota 1999), leaves for plates (Rajan 1995; Gautam and Devkota 1999), seed for oil (Venkat and Sharma 1978; Sharma 1981), feed (Rai and Shukla 1977; Sinha and Nath1982), resin or latex from heartwood (FRIB 1947) and tannin and gum from bark (Narayanamurti and Das 1951; Karnik and Sharma 1968). Sal forests cover nearly 13.3% of the forested landscape (Satya and Nayaka 2005) including Uttar Pradesh, Uttarakhand and Shivalik Hills in Haryana, and the range also extends through the Eastern Ghats and to the eastern Vindhyan and Satpura ranges of central India.
Jharkhand is very rich in floristic diversity because of its past geological history. It is believed that in the geological past, this plateau formed a link between the Satpura Hill Ranges and the Eastern Himalaya that allowed species exchanges between these ranges (Hora 1949). The area is covered with a variety of different habitats, of which the sal forest is predominant. Champion and Seth (1968) classified the sal forests of Jharkhand mainly into two types: (1) Moist Peninsular Low Level Sal (3C/C2e) and (ii2 Dry Peninsular Sal Forest (5B/C1c). Sal is associated with various other species, and the companion species differ to some extent in different localities according to the local climate. The chief companions are Buchanania cochinchinensis (Lour.), Anogeis suslatifolia DC. Wallich ex Guill. and Perr, Diospyrus melanoxylon Roxb., Terminalia tomentosa Roxb. (exDC) Wight and Arn., Butea monosperma (Lam.) Taub., Adina cordifolia (Roxb.) Brandis, Lagerstroemia parviflora Roxb., Pterocarpus marsupium Roxb., Madhuca longifolia L.
Phytosociological studies help in understanding the structure and the composition of plant communities (Braun-Blanquet et al. 1965). The species diversity, floristic composition and vegetation structure are important to judge the state of natural forests in any region and to formulate conservation and management strategies. Tropical and subtropical forests harbor the greatest diversity of plants (more than half of the world’s species) but cover only 7% of the Earth’s land surface (May and Stumpf 2000). Of 86% of tropical forests in India, 37% as moist deciduous forests (Kaul and Sharma 1971; Singh and Kushwaha 2005). Structurally and floristically, the tropical moist deciduous forests are more complex than dry deciduous forests. Tropical moist deciduous forests are distributed in the states of Jharkhand, Chhattisgarh, west Odisha and on the eastern slopes of Western Ghats, India. Because very limited work has been done in the forests of Ranchi, Jharkhand in terms of phytosociological analysis (Kumar and Abbas 2012; Lal and Singh 2012; Kumar et al. 2013; Rahul and Jain 2014; Divakara 2015). The present study evaluated the floristic diversity and dominance pattern of the sal forests in Ranchi, Jharkhand, eastern India to provide an overview of its present floristic composition to formulate appropriate management strategies for conserving the most economically and ecologically important species in the sal forests.
Materials and methods
Study area
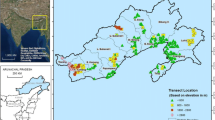
The present study was conducted in nine (Khelari, Burmu, Chano, Mandar, Ratu, Kanke, Ormanjhi, Angara and Namkum) of 18 blocks in the Ranchi district in the southern Chotanagpur administrative division (23.35°N, 85.33°E, 400–700 m above sea level; Fig. 1). There are two forests divisions in Ranchi district, the Ranchi forest division and Bundu forest division, which are further subdivided into 12 forest ranges. According to The Köppen Climate Classification, the climate in Ranchi is Cwa (Humid Subtropical), and mean annual rainfall is about 1430 mm. The topography is hilly with stony and gravelly, red and yellow, lateritic and alluvial soils and tropical moist deciduous and dry deciduous forests.
Sampling design and vegetation sampling
A total of 47 transects have been sampled in 43 grids (out of 88 grids of sal forests) of nine blocks of Ranchi district of Jharkhand during September 2016 to April 2017. In general, one transect is laid in each grid but in four grids two transects have been laid due to wrong identification of location at field. Details of sampled transects are mentioned in Table 1. For grid generation, all topographical sheets (scale 1:50,000) of Ranchi were collected from the Survey of India, and each sheet was further divided into 16 grids using the fishnet method in GIS software. Further, the grids were overlaid on the base map, and the number of grids with sal forests were identified. The entire district was thus covered by 153 grids, with sal forests in 88 grids. Details on the grid generation are shown in Fig. 2. Vegetation was sampled by the stratified random sampling approach using belt transect method. All strata, i.e., trees, shrubs and herbs, were sampled. The size of the sampling units for trees, shrubs and herbs is one 100 × 50 m transect for trees (≥ 10 cm GBH), one 5 × 5 m quadrat for shrubs and two 1 × 1 m quadrants for herbs at each sample site. The detail design of sampling transects showing the quadrat for shrubs and herbs are presented in Fig. 3. The girth at breast height (1.37 m above ground; GBH) using measuring tape and visual height of all the individual trees (≥ 10 cm GBH) was measured in each transect. Trees with GBH < 10 cm (saplings) were sampled as shrubs, and trees and shrubs with height ≤ 30 cm (seedlings) were sampled as herbs. Herbarium specimens of each of the plant species were prepared during the study, and specimens were photographed in the field. Plant species were identified on the basis of vernacular names, published field inventories, floras, consulting herbaria of the region as well as Botanical Survey of India. All tree species were grouped by diameter classes into seedlings (≤ 30 cm height), saplings (< 10 cm GBH and > 30 cm height) and trees (≥ 10 cm GBH) for analyses of population structure. The data were used to compute density (individuals ha−1) of seedlings, saplings and trees and basal area (cm2 ha−1) of trees in each transect.
Data analyses
Quantitative analysis of vegetation was done using the method of Misra (1968). Importance value index (IVI) was computed as the sum of relative density, relative frequency and relative basal area (i.e., relative dominance). Species richness was defined as the total number of species in the sampling sites. Margalef’s index was used as a simple measure of species richness (Margalef 1958) using the formula Margalef’s index of richness (D mg) = (S − 1)/ln N, where N = total number of species and N = total number of Individuals.
The Shannon–Wiener diversity index (Shannon and Wiener 1963) was calculated from the abundance values using the formula given by Magurran (1988): H′ = − Σ (pi ln pi), where pi is the proportion of the IVI of ith species and the IVI of all the species.
The concentration of dominance CD was assessed using Simpson’s index (Simpson 1949):
where pi is the same as used in the Shannon–Wiener information function.
For calculating the evenness of species E, Pielou’s evenness index (e) was used (Pielou 1996): E = H′/Hmax′, where H′ is the Shannon–Wiener diversity index and Hmax′ = ln S (where, S = total number of species). The species evenness measures the equality of species in a given sample and has a value between 0 and 1, where values closer to 0 indicate the abundance of the few species and 1 indicates that all species are equally abundant (Whittaker 1975). Species distribution pattern of the plant species was calculated as the ratio of abundance to frequency (A/F) as suggested by Whitford (1949) as a measure of contiguousness. The ratio indicates regular (< 0.025), random (0.025–0.05) and contiguous (> 0.05) distributions (Curtis and Cottam1956).
On the basis of the number of individuals, species were grouped into very rare (1 individual), rare (2–10), common (11–25), dominant (26–50) and predominant (> 50) (Kadavul and Parthasarathy 1999). All plant species were grouped into one of five frequency classes based on the frequency distribution of the species according to Raunkier’s law of frequency (Raunkiaer 1934): A = 1–20%, B = 21–40%, C = 41–60%, D = 61–80% and E = 81–100%.
The life form of each species was classified according to Raunkiaer (1934) assignments as therophytes (Th), phanerophytes (Ph), chamaephytes (Ch), hemicryptophytes (H) and cryptophytes (C) as modified by Braun-Blanquet et al. (1965) has been followed. The percentage of each life form was calculated using the following formula:
Results and discussion
Floristic composition
Within the 47 studied transects, 12,812 individual vascular plants were recorded: 9328 trees (> 10 cm GBH) (73% of total), 1298 shrubs (including saplings of trees) (10% of total) and 2186 herbs (including shrub and tree seedlings) (17% of total). Among these plants, 137 plant species (110 identified and 27 unidentified) and the 110 identified species are belonging to 51 families, of which 20 species (18.18%) are invasive and 90 (81.82%) species are native to India. The richness of species is principally due to trees (5%) followed by herbs (25%) and shrubs (18%). In general, tropical forests have higher species richness as indicated by the presence of a relatively high number of species from various families. The reported species richness of the sal forest of Ranchi (137 spp.) is comparable with that of sal forests in other parts of India including the Gorakhpur division, Uttar Pradesh (Pandey and Shukla 2003), Eastern Himalayas (Shankar 2001), Central Himalayas (Pande 1999) and Corbett National Park, Uttarakhand (Singh et al. 1995; Singh and Singh 1987). The higher species diversity in tropical regions is mainly due to the diverse habitat and topography, productivity, prevailing climatic conditions, seasonality and dynamics such as immigration. In terms of species contribution, Fabaceae is the most dominant family with 17 species (8 spp. of Faboidae, 6 spp. of Caesalpinioidae, 3 spp. of Mimosoidae) followed by Rubiaceae with eight species and Euphorbiaceae with 6. Distribution of families according to number of species is presented in Fig. 4.
In forest ecosystems, the magnitude of changes is studied through phytosociological aspects, mainly by density and total basal cover, which are the principal bases of study for any vegetation type (Mishra et al. 2003). The total tree density of the studied transect was 397 individuals ha−1, and basal area of all the trees was 262.50 m2 ha−1. The observed tree density (397 individuals ha−1) is lower as compared to Piranmalai Forest, Eastern Ghats, Tamil Nadu, India (Siva et al. 2008) but within the range of tree densities reported in the tropics (276–905 stems ha−1; Murali et al. 1996; Sundarapandian and Swamy1997; Utkarsh et al. 1998; Reddy et al. 2007). Sal is one of the dominant trees in the forests of South Asia (Troup 1921). Shorea robusta was present at the highest density and basal cover (78.84% and 78.52% of the respective totals), followed by D. melanoxylon, B. cochinchinensis, M. longifolia, L. parviflora, Mangifera indica L., B. monosperma (Table 2). The most obvious variation in term of species composition and the proportion of dominant species in different forest stands was directly determined by altitude and rainfall distribution. The canopies of sal-dominated forests look similar across regions, but vary in floristic structure, patterns of species diversity, coverage of basal area, canopy height and species regeneration. Sal density in all 47 documented forests varied from 92 to 894 individuals ha−1 (mean 313 individuals ha−1 ± 28.30 SE), whereas species richness ranged from 1 to 25 (mean 11 ± 0.86 SE). In the present study, the density of sal was inversely related to total plant species richness. Forests with higher density of sal had low species richness and forests with low density sal had higher species richness (Fig. 5). Dominance of sal depends on the age, available resources, associated species, disturbance regime and successional changes as well as water accumulation and human interference for the timber and other uses. In the present study, interference caused by grazing, timber, fodder collection, fuel gathering (wood and leaves) and fresh leaves for use as plates, commercial extraction of leaves of D. melanoxylon used in the bidi industry are quite common in almost all the studied forests (Fig. 6). Floristic inventories in sal forests in different parts of India vary in terms of plot size, sampled area and measurement threshold of minimum stem diameter, thus limiting comparisons of species richness (Table 3). Analysis of IVI values showed that S. robusta was the dominant species (167.29), well within the limits of earlier studies (Rawat and Bhainsora 1999), followed by D. melanoxylon (13.04) and B. cochinchinensis (11.04). Tree species with higher IVI values indicate good regenerative capability, better adaptability and wide ecological amplitude.
Diversity indices provide important information about rarity and commonness of species in a community and is an important tool to understand community structure. The Shannon–Wiener diversity index (H′) for Indian forests has been reported to range from 0.83 to 4.10 (Jha and Singh 1990; Ayyappan and Parthasarathy 1999; Pandey 2000). In the present study, the value of the Shannon diversity index (H′) in the present study (2.25 for trees, 2.72 for shrubs and 2.98 for herbs) lies within the range reported for tropical forests but was much lower than the recorded value (3.59) for the Eastern Himalayan sal forest (Uma Shankar 2001) and (3.10) West Bengal moist sal forest of India (Kushwaha and Nandy 2012). Details of other community parameters including concentration of dominance, evenness index, and Margalef’s index of richness are presented in Table 4. The distribution pattern of species in a forest ecosystem is one of the important aspects of ecological studies. A contiguous distribution is characteristic of natural forests (Odum 1971). In the present study, all the plant species were distributed contiguously on the basis of A/F ratio. Contagious (i.e., clumped) distribution reflects the magnitude of interference (e.g., grazing and lopping) in natural forest stands. The general preponderance of contagiously distributed vegetation has been reported by various workers (Rao et al. 1990; Greig-Smith 1983; Singh and Yadava 1974; Kershaw 1973). Contagious distribution is based upon local habitats differences, daily and seasonal weather conditions, and reproductive processes of the species of that area. The dominance of contagious distribution may also be due to the fact that the majority of herb species reproduce vegetatively in addition to sexually. Based on the number of individuals present, the percentage of plants species in different rarity classes varied greatly, with very rare species accounting for 21.90%, rare species 32.12%, common species 16.06%, dominant species 10.22% and predominant species 19.71% (Fig. 7). Some example of very rare species were Sterculi aurens, Bauhinia variegata, Senna siamea, Ficus glomerata, Neonauclea purpurea (Roxb.) Merr.
Raunkiaer’s frequency analyses revealed that 77.63% of the plant species were rare, 14.47% were seldom present and 3.95% often present (Fig. 8). S. robusta was the most frequent species (100%), followed by D. melanoxylon (72.34%); Anacardium occidentale L., Elaeodendron glaucum (Rottb.) Per, Boswellia serrata, S. siamea (Lam.) H.S. Irwin and Barneby, Lagerstroemia speciosa, Carissa spinarum L., Grewia laevigata, Cassine glauca (Rottb.), B. variegata L., Tectona grandis L., and F. glomerata Roxb. were the least frequent. Most species (77.63%) in the sal forests of Ranchi had low frequency (0–20%), suggesting that most would be expected to be rare in typical species abundance distributions. Only a few species (1.32%) were present at a higher frequency (81–100%) in the studied forests and are constantly present. The normal species occurrence ratio given by Raunkiaer is A > B > C ≥ D < E, meaning that a particular plant community is homogeneous. But in the present study, the normal species occurrence ratio was A > B > C ≥ D > E, unlike the Raunkiaer normal species occurrence; thus, the sal forests of Ranchi are heterogeneous in nature, with a heterogeneous plants species distribution in the studied forests.
Raunkiaer’s life form and biological spectrum
The biological spectrum and maximum divergence from the normal spectrum as proposed by Raunkiaer (1934) for the 110 identified plant species in the present study, from highest to lowest, comprised 71% phanerophytes (78%, percentage deviation + 25), 15% therophytes (percentage deviation + 2), 7% chamaephytes (eight species, percentage deviation − 2), 5% hemicryptophytes (six species, with percentage deviation − 21), 2% cryptophytes (two species, percentage deviation − 4). The comparative biological spectrum of present study and Raunkiaer normal spectrum is presented in Fig. 9. A similar phytoclimatic association has also been reported by other workers from different parts of the world (Lakshmanan 1962; Misra et al. 1979; Saxena 1979; Saxena et al. 1982; Rajendraprasad et al. 1998; Khatri 2000; Thakur and Khare 2011). The much higher percentage of phanerophytes (71%) compared with Raunkiaer’s normal value (46%) is evidence that the climate of Ranchi is ideal for sustaining the tree diversities. A predominance of therophytes in natural forests is mainly due to grazing (Singh and Yadava 1974) and is also an indicator of biotic pressures (Bharucha and Dave 1944). Chamaephytes and therophytes, considered indicators of unfavorable environmental conditions, were found at almost similar proportions to the normal value of Raunkiaer. Observed percentage deviation from normal life form was much lower (+ 2 percentage deviation) for both chamaephytes (8%) and therophytes (15%), suggesting that the studied sal forests are favorable for supporting various plants species and maintaining biological diversity.
Stratification
Forests in Ranchi have a complex canopy structure and are vertically divided into several layers, each with their own unique ecological significance, creating an integrated and biologically complex living space. The high diversity of tree species in tropical moist forests might permit the aggregation of trees into four or five strata, and the number of strata tends to decrease with increasing altitude (Greig-Smith 1964). Based on the average height of the plant species in the present study, there are five distinct strata in the sal forests of Ranchi: emergent (> 20 m) with 1685 individuals belonging to 31 tree species, canopy (> 10–20 m) with 7175 individuals belonging to 92 tree species, understorey (> 5–10 m) with 502 individuals belonging to 31 tree species, shrub (1–5 m including saplings of the tree species), and ground (< 1 m including seedlings of both tree and shrub species) (Fig. 10). From our data, the height of individual plants that attained 40 cm GBH exceeded 15 m and their crowns rise above the dense foliage layer (canopy strata). Canopy structure determines the distribution of light and precipitation received by subordinate trees and understorey plants (Anderson et al. 1969; Cannell and Grace 1993). The emergent layer was usually present in continuously moist soil on lower slopes and dominated by timber trees such as S. robusta, B. cochinchinensis (Lour.) Almeida, M. longifolia L., Syzygium cumini L., Semecarpus anacardium L.f., Schleichera oleosa, and similarly, the canopy stratum was also dominated by timber trees such as B. cochinchinensis, Cassia fistula L., B. monosperma, A. cordifolia, Dalbergia pseudo-sissoo Miq., M. longifolia, fruit plants such as Aeglemar melos (L.) Correa and had the largest cover (58%). The understory stratum contributed 3% of the total flora and was dominated by plants such as Psidium guajava, D. melanoxylon, Bombax ceiba L., etc. The shrub layer consisted of numerous shrub species and saplings of higher strata and contributed 12% of the total flora and was dominated by Croton caudatus Geiseler, Clerodendrum infortunatum L., C. spinarum L., Meyna laxiflora Robyns etc. and invasive species such as Lantana camara L. The ground stratum consisted of seedlings and mainly herbaceous weeds and grasses such as Elephantopus scaber L., Asparagus densiflorus (Kunth) Jessop, Panicum spp., Phyllanthus niruri L. Some herbs such as Smilax zeylanica L., Andrographis paniculata (Burm.f.) Wall, A. densiflorus are harvested by local tribal people for traditional healing practices.
Population structure and regeneration status
Total density of sal individuals in the studied forests was 19,930 individuals ha−1, of which 19,362 individuals ha−1 (97.15%) were seedlings, 255 individuals ha−1 (1.28%) were saplings and 313 individuals ha−1 (1.57%) were adults (≥ 10 cm GBH). On the basis of the density of seedling, sapling and adults of different girth classes, S. robusta had fair regeneration status (seedlings > trees > saplings) and wide adaptability in the different ecological conditions in the studied forests. Its percentage distribution from lower to higher girth classes according to density per hectare showed a decreasing trend and maximum density observed in the lower girth classes. Among adults, the highest percentage was contributed by the 30–60 cm girth class (44.76%), followed by 60–90 cm girth class (25.93%), then > 120 cm (15.19%). On the other hand, most of the individuals belonged to the higher height class, i.e., 15–20 m height class (50%), followed by 10–15 m height class (32%) of the 103 woody plant species (shrubs and trees) identified, only 18 species (17.48%) were regenerating (e.g., S. robusta, Alstonia scholaris (L.) R.Br., L. parviflora, Holarrhena pubescens (Buch.-Ham). Wall. ex G.Don, S. cumini). The highest percentage of trees were young (31–60 cm GBH; 44.48%), followed by older (61–90 cm GBH, 25.95%), then mature trees (> 120 cm GBH, 3.87%) (Fig. 11).
Conclusions
The sal forests of the chota Nagpur Palateu region in Ranchi have been subjected to centuries of anthropogenic pressure. The most serious threat has been the change in land-use patterns. Extensive forest tracts have been cleared for human settlements, agricultural practices, road network and industrial units. The remnant patches are not safe despite legal protection. Illicit timber and fuelwood cutting, collection of nontimber products, grazing, fire and hunting also pose a threat. Because timber extraction is illegal, quantifying the extent of the problem is not possible. However, this sal forest is an extremely important ecosystem by the virtue of high species richness and diversity (137 plant species). Important plants for conservation concern include P. marsupium, Sterculia urens, Tinospora cordifolia, A. densiflorus and A. paniculata. This study indicates that the climatic conditions of the region have favoured recruitment of sal and its associates in the climax formation over a long successional process. Forest management strategies should focus on meeting the increasing demands of different timber and non-timber forest products while conserving the plant diversity of these natural forests.
References
Anderson RC, Loucks OL, Swain AM (1969) Herbaceous response to canopy cover light intensity, and through fall precipitation in coniferous forests. Ecology 50:255–263
Ayyappan N, Parthasarathy N (1999) Biodiversity inventory of trees in a large-scale permanent plot of tropical evergreen forest at Varagalaiar, Anamalais, Western Ghats, India. Biodivers Conserv 8(11):1533–1554
Bharucha F, Dave RN (1944) The biological spectrum of grassland association. J Univ Bombay 13:15–16
Bor NL (1953) Manual of Indian forest botany. Oxford University Press, Bombay
Borchert R (2000) Organismic and environmental controls of bud growth in tropical trees. Dormancy in plants: from whole plant behavior to cellular control. CAB Int 21:87–107
Braun-Blanquet J, Fuller GD, Conard HS (eds) (1965) Plant sociology: the study of plant communities. Hafner, London
Cannell MGR, Grace J (1993) Competition for light: detection, measurement, and quantification. Can J Forest Res 23:1969–1979
Champion HG, Seth SK (1968) A revised survey of forest types of India. Government of India, New Delhi
Cooke T (1958) Flora of Bombay, vol 1. Botanical Survey of India, Calcutta, p 375
Curtis JT, Cottam G (1956) Plant ecology work book: laboratory, field and reference manual. Burgess Publishing Company, Minneapolis
Divakara BN (2015) Floristic analysis, phytosociological studies and regeneration status of tree species in sal forests of Jharkhand, India. https://www.researchgate.net/publication/286443392. Accessed 23 Jan 2016
Edwards DM (1996) The trade in non-timber forest products from Nepal. Mt Res Dev 1:383–394
Fox J (1995) Society and non-timber forest products in tropical Asia. East-West Center Occasional Papers Environment Series No. (19)37–51
FRIB (1947) Experimental tapping of sal and blue pine. For Resour India Burma 6(1):88–90
Gautam KH (1990) Regeneration status of sal (Shorea robusta) forests in Nepal. Ministry of Forests and Soil Conservation, Kathmandu, p 11
Gautam KH, Devkota BP (1999) Sal (Shorea robusta) leaves can provide income to some community forestry user groups at Sindhupalchok district. Nepal J For 11:39–46
Greig-Smith P (1964) Quantitative plant ecology, 2nd edn. Butterworths, London, p 256
Greig-Smith P (1983) Quantitative plant ecology, 3rd edn. Blackwell, Oxford, p 359
Hora SL (1949) Satpura hypothesis of the distribution of the Malayan fauna and flora to Peninsular India. Proc Natl Inst Sci 15:309–314
Jha CS, Singh JS (1990) Composition and dynamics of dry tropical forest in relation to soil texture. J Veg Sci 1(5):609–614
Joshi HB (1980) The silviculture of Indian trees. Forest Research Institute (FRI), Dehradun, p 471
Kadavul K, Parthasarathy N (1999) Plant biodiversity and conservation of tropical semi-evergreen forest in the Shervarayan hills of Eastern Ghats, India. Biodivers Conserv 8:421–439
Karnik MG, Sharma OP (1968) Cellulose gums from sal (Shorea robusta) bark and Bamboo (Dendrocalamus strictus). Indian Pulp Pap 22:451–453
Kaul ON, Sharma DC (1971) Forest type statistics. Indian For 97:432–436
Kershaw KA (1973) Quantitative and dynamic plant-ecology, 3rd edn. ELBS and Edward Arnold Ltd, London, p 308
Khatri PK (2000) Study on biodiversity in tropical forest ecosystem of Satpura National Park, Madhya Pradesh (Doctoral dissertation, Ph.D. thesis. Forest Research Institute, Dehra Dun
Kirtikar KR, Basu BD (1975) Indian medicinal plants, vol 1. Jayyed Press, New Delhi
Krishnaswamy VS, Mathauda GS (1954) Phonological behavior of a few forest species at new forest, Dehradun. Indian For 80:124–153
Kumar K, Abbas SG (2012) Ethnomedicinal composition depends on floristic composition: a case studied in sal forests of Jharkhand. Int J Pharm Life Sci 3(5):1710–1719
Kumar A, Prasad S, Singh S (2013) Climbers and lianas distribution in Jharkhand Forests. Indian For 139(12):1121–1125
Kushwaha SP, Nandy S (2012) Species diversity and community structure in sal (Shorea robusta) forests of two different rainfall regimes in West Bengal, India. Biodivers Conserv 21(5):1215–1228
Lakshmanan NK (1962) The application of Raunkiaer’s life-forms. J Ind Bot Soc 41(4):585–589
Lal HS, Singh S (2012) Study of plant biodiversity of Hazaribag district Jharkhand India and its medicinal uses. J Biosci Discov 3(1):91–96
Magurran AE (1988) Why diversity. Ecological diversity and its measurement. Springer, Netherlands, pp 1–5
Majumdar K, Shankar U, Datta BK (2014) Trends in tree diversity and stand structure during restoration: a case study in fragmented moist deciduous forest ecosystems of Northeast India. J Ecosystems 2014:1–10
Margalef R (1958) Temporal succession and spatial heterogeneity in phytoplankton. In: Buzzati-Traverso A (ed) Perspectives in marine biology. University of California Press, Berkeley, pp 323–347
Mathema P (1991) Sal regeneration management. Nepal J For 6(2):1121–1124
May RM, Stumpf MPH (2000) Species-area relations in tropical forests. Science 290:2084–2086
Mishra BP, Tripathi RS, Tripathi OP, Pandey HN (2003) Effect of disturbance on the regeneration of four dominant and economically important woody species in a broad-leaved subtropical humid forest of Meghalaya, northeast India. Curr Sci 84(10):1449–1453
Misra R (1968) Ecology workbook. Oxford & IBH Publishing Company, Calcutta, p 244
Misra MP, Misra PN, Pandey BN (1979) An ecological account of the vegetation of Brahmyoni Hills (Gaya). Proc Indian Sci Congr 3:108–109
Murali KS, Uma S, Shanker U, Ganeshaiah KN, Bawa KS (1996) Extraction of forest products in the forests of Biligirirangan Hills, India and impact of NTFP extraction on regeneration, population structure, and species composition. Ecol Bot 50:252–269
Narayanamurti D, Das NR (1951) A preliminary note on adhesives, building boards and moulding powders from tree barks. Indian For 77(11):706–708
Odum EP (1971) Fundamentals of ecology. W.B. Saunders Co., Philadelphia, p 574
Panday K (1982) Fodder trees and tree fodder in Nepal. Fodder trees and tree fodder in Nepal (Swiss Development Cooperation, Berne)
Pande PK (1999) Comparative vegetation analysis and sal (Shorea robusta) regeneration in relation to their disturbance magnitude in some sal forests. Trop Ecol 40(1):51–61
Pandey SK (2000) Population status and regeneration strategy of some perennial legumes in plantation forests of North-Eastern Uttar Pradesh, Ph.D. thesis, DDU Gorakhpur University, Gorakhpur, India
Pandey SK, Shukla RP (2003) Plant diversity in managed sal (Shorea robusta Gaertn.) forests of Gorakhpur, India: species composition, regeneration and conservation. Biodivers Conserv 12(11):2295–2319
Pandey S, Yadama GN (1990) Conditions for local level community forestry action a theoretical explanation. Mt Res Dev 10(1):88–95
Pielou EC (1996) The measurement of diversity in different types of biological collections. J Theor Biol 13:131–144
Rahul J, Jain MK (2014) Documentation of floristic inventory along the national highway: a case study of Dhanbad district, Jharkhand, India. J Biodivers Environ Sci 5(6):241–247
Rai SN, Shukla PC (1977) Influence of feeding deoiledsal seed meal (DSSM) with urea and molasses on digestibility and balances of nitrogen, phosphorus and calcium in lactating cows. Indian J Anim Sci 47:111–115
Rajan RP (1995) Sal leaf plate processing and marketing in West Bengal. In: Fox J (ed) Society and non-timber forest products in tropical Asia. East-West Center, Honolulu, pp 27–36
Rajendraprasad M, Krishnan PN, Pushpangadan P (1998) The life form spectrum of sacred groves—a functional tool to analyse the vegetation. Trop Ecol 39(2):211–217
Rao P, Barik SK, Pandey HN, Tripathi RS (1990) Community composition and tree population structure in a sub-tropical broad-leaved forest along a disturbance gradient. Vegetatio 88(2):151–162
Raunkiaer C (1934) The life forms of plant and statistical plant geography. Oxford University Press, Oxford, p 632
Rawat GS, Bhainsora NS (1999) Woody vegetation of Shivaliks and outer Himalaya in north western India. Trop Ecol 40(1):119–128
Reddy CS, Pattanaik C, Mohapatra A, Biswal AK (2007) Phytosociological observations on tree diversity of tropical forest of Similipal Biosphere Reserve, Orissa, India. Taiwania 52:352–359
Satya DK, Nayaka S (2005) Shorea robusta—an excellent host tree for lichen growth in India. Curr Sci 89:594–595
Saxena AK (1979) Ecology of vegetation complex of north-western catchment of river Gola (Doctoral dissertation, Ph.D. thesis, Kumaun University, Nainital, India)
Saxena AK, Pandey P, Singh JS (1982) Biological spectrum and other structural functional attributes of the vegetation of Kumaun Himalaya. Plant Ecol 49(2):111–119
Shakya CM, Bhattarai DR (1995) Market survey of non-wood forest products in Bara and Rautahat districts for operational forest management plans. Forest Management and Utilization Development Protect, Department of Forest, Kathmandu, p 30
Shankar UA (2001) Case of high tree diversity in a sal (Shorea robusta)-dominated lowland forest of Eastern Himalaya: floristic composition, regeneration and conservation. Curr Sci 81(7):776–786
Shannon CE, Wiener W (1963) The mathematical theory of communication. University of Illinois Press, Urbana
Sharma BK (1981) Further studies on seed production in sal (Shorea robusta Gaertn.) crops in Dehra Dun District (UP). Indian For 107(8):505–509
Simpson EH (1949) Measurement of diversity. Nature 163:688
Singh KP, Kushwaha CP (2005) Emerging paradigms of tree phenology in dry tropics. Curr Sci 89(6):964–975
Singh JS, Singh SP (1987) Forest vegetation of the Himalaya. Bot Rev 53:80–92
Singh JS, Singh SP (1992) Forests of Himalaya. Gyanodaya Prakashan, Nainita, p 294
Singh JS, Yadava PS (1974) Seasonal variation in composition, plant biomass, and net primary productivity of a tropical grassland at Kurukshetra, India. Ecol Monogr 44(3):351–376
Singh A, Reddy VS, Singh JS (1995) Analysis of woody vegetation of Corbett National Park, India. Vegetatio 120(1):69–79
Sinha RP, Nath K (1982) Effect of urea supplementation on nutritive value of deoiledsal-meal in cattle. Indian J Anim Sci 52:1165–1169
Siva N, Pitchairamu C, Muthuchelian K (2008) Floristic inventory and quantitative vegetation analysis of tropical dry deciduous forest in Piranmalai Forest, Eastern Ghats, Tamil Nadu, India. Ethnobot Leafl 2(1):25
Sundarapandian SM, Swamy PS (1997) Plant biodiversity at low-elevation evergreen and moist deciduous forests at Kodayar (Western Ghats, India). Int J Ecol Environ Sci 23:363–379
Thacker P, Gautam KH (1994) A socio-economic study of participatory issues in forest management in the Terai. HMGN/FINNIDA
Thakur AS, Khare PK (2011) Study of life-forms and biological spectrum of forest of Sagar district, Madhya Pradesh. In: Vyas D, Paliwal GS, Khare PK, Gupta RK (eds) Microbial biotechnology and ecology. Daya Publishing House, New Delhi, pp 729–740
Tiwari DN (1995) A monograph on sal (Shorea robusta). International Book Distributors, Dehra Dun
Troup RS (1921) The silviculture of Indian trees. Clarendon Press, London
Upadhyay LR (1992) Use of tree fodder in Jhapa and Sunsari Districts in the eastern Terai. Banko Janakari 3(3):17–18
Utkarsh G, Joshi NV, Gadgil M (1998) On the patterns of tree diversity in the Western. Curr Sci 75(6):594–603
Venkat VP, Sharma BK (1978) Studies on production and collection of sal (Shorea robusta Gaer). seeds. Indian For 104(6):414–420
Whitford PB (1949) Distribution of woodland plants in relation to succession and clonal growth. Ecology 30:199–288
Whittaker RH (1975) Communities and ecosystems. MacMillan, New York
Acknowledgements
The authors sincerely thank the Science and Engineering Research Board (SERB) for financial assistance for the project “Status, Distribution and Composition of Sal Forests of Ranchi, Jharkhand, Eastern India in relation to Microclimatic as well as Edaphic Conditions” (Ref. No. YSS/2015/000479 dated 12 January 2016). The help and support received from the State Forest Department, Ranchi and local people involved in the survey and sampling are duly acknowledged.
Author information
Authors and Affiliations
Contributions
RK participated in data collection, analysis and interpretation and also drafted the manuscript. PS conceived the study, participated in its design, coordinated during data collection and finalized the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Project funding: This work was supported by Science and Engineering Research Board (SERB) under Young Scientist Scheme (Ref. No. YSS/2015/000479 dated 12th January 2016).
The online version is available at http://www.springerlink.com
Corresponding editor: Chai Ruihai.
Rights and permissions
About this article
Cite this article
Kumar, R., Saikia, P. Floristic analysis and dominance pattern of sal (Shorea robusta) forests in Ranchi, Jharkhand, eastern India. J. For. Res. 31, 415–427 (2020). https://doi.org/10.1007/s11676-018-0829-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-018-0829-9