Abstract
Nectandra is one of the most representative genera of Lauraceae in the subtropical Atlantic Forest of Brazil. The objective of this work was to study the dendrochronological potential of Nectandra oppositifolia Nees and Mart. from two sites in Santa Catarina State in southern Brazil. A tree-ring chronology of 34 trees was developed. The time span ranged from 1843 to 2013. The oldest and youngest trees were 171 and 47 years-old and the average length of the series was 103 years. Average diameter and annual increment were 20.64 cm and 0.74 mm a−1; diameter and age were unrelated. The species has good dendrochronological potential with an intercorrelation of 0.61 between sites, indicating the existence of a synchronous pattern in the development of the trees. The climate response of the species could be seen by negative tree growth effects for previous hot and current rainy growth seasons.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Dendrochronology is a useful tool that provides important information about the development and ecology of species (Schweingruber 2007; Zuidema et al. 2013). Despite being a relatively recent science in the tropics, several studies have confirmed the existence of climatic seasonality and species that form seasonal growth rings, two essential elements that enable the use of dendrochronology in tropical environments (Worbes 1999, 2002; Schöngart et al. 2002).
Factors such as temperature, precipitation, flooding, droughts and/or photoperiod regulate the growth of species in tropical and subtropical regions (Callado et al. 2001; Schöngart et al. 2002; Oliveira et al. 2010). Dendroecology synthesizes this information in an accessible way that allows for the identification of climatic signals and the production of climate reconstructions (Worbes 1999; Brienen and Zuidema 2005; Oliveira et al. 2010).
In the Atlantic Forest, dendroecological studies have been conducted in different vegetation types that make up the biome (Lisi et al. 2008; Oliveira et al. 2010; Costa et al. 2015) such as rain forest (Callado et al. 2001; Callado and Guimarães 2010; Latorraca et al. 2015).
The abundance of arboreal species of Nectandra Rol. ex Roth (Lauraceae) in subtropical Atlantic Forests in southern Brazil has been noted in previous studies, and the dominance of Nectandra oppositifolia Nees and Mart. has also been recorded in dense rain forests in this region (Bechara et al. 2009; Lingner et al. 2013). Nectandra oppositifolia is economically and ecologically valuable because of the quality of its wood and because it occurs largely in secondary forests (Bechara et al. 2009). This makes understanding the development of this species in its native environment important, especially because there is a need for information about the lifetimes of subtropical forests and there is a lack of chronological studies of species from southern Brazil.
Alves and Angyolossy-Alfonso (2000) studied the anatomy of native species in Brazilian rain forests and confirmed the presence of growth rings in N. oppositifolia. Therefore, this study tested the hypothesis that N. oppositifolia from a dense rain forest in southern Brazil has distinct growth rings and cambial seasonality, which are favorable characteristics for the development of dendrochronological studies in this forest environment. The objectives of this study were the following: verify the presence and distinctiveness of tree rings in N. oppositifolia trees; identify their synchrony and periodicity; and, determine the dendroclimatic response of this species.
Materials and methods
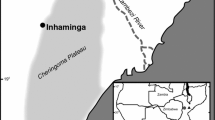
This study was conducted in two areas of dense rain forest in Santa Catarina State, southern Brazil (Fig. 1). The first site is a secondary forest fragment in early to mid regeneration stages located in Blumenau on Campus 5 of the Fundação Universidade Regional de Blumenau (FURB) (26°51′19″S, 49°03′02″W). The soil is an argisoil (EMBRAPA 2006). The second area is also a secondary forest fragment in Joinville (26°22′21″S, 48°52′10″W) with medium to advanced regeneration (Vibrans et al. 2013), near sampling unit # 997 of the Floristic Forest Inventory of Santa Catarina (FFISC). The site has a haplic organosoil (EMBRAPA 2006).
The climate in the two areas is humid subtropical (Ayoade 2007) with humid springs and summers, (the rainiest months are January, February and March), and lower precipitation and temperatures during the fall and winter (April, May, June, July and August) (Fig. 2). The regional climate has an average precipitation of 1512 mm, an average annual temperature of 20.4 °C, a maximum average temperature of 27.1 °C and a minimum average temperature of 16.2 °C (CEOPS/FURB 2015; INMET 2015). Previous studies have shown that the hotter and longer days of the spring and summer months trigger cambium activity and stem growth in subtropical environments (Dünisch 2005; Oliveira et al. 2010; Shimamoto et al. 2015).
N. oppositifolia, commonly known as canela ferrugem, is an evergreen species (Backes and Irgang 2004) up to 30-m with a diameter at breast height (DBH) of up to 70 cm (Quinet 2006). This species is a native of the Atlantic Forest and rain forests from Central America to Brazil where it is widely distributed. In southern Brazil it occurs in dense rain forest (Zanon et al. 2009) and data from the FFISC reveals that it is one of the species of high importance values in lowland dense rain forests (Lingner et al. 2013). Nectandra oppositifolia is classified as an early to late secondary species (Gandolfi et al. 1995; Higuchi et al. 2006; Pinto-Sobrinho et al. 2009) and a species that can be used for urban forestry and for the recovery of degraded areas (Backes and Irgang 2004).
Samples were collected from 34 healthy trees and selected according to their natural occurence in the forest fragments, 12 in Blumenau and 22 in Joinville. The trees had straight trunks and free of imperfections. All trees were georeferenced and DBH measured (1.3 m) from 9 to 30 cm. Four wood samples 5.0 mm in diameter and diametrically opposite were extracted at breast height using a Pressler increment borer (Botoso et al. 2000).
Tree-ring analyses
After drying at room temperature, the wood samples or cores were glued to a wooden support and micro polished with abrasive paper to obtain a clear surface. Tree rings were marked, dated under a stereomicroscope and identified according to IAWA (1989). The samples were dated from the outermost ring which corresponded to the last growth year.
Digital core images (minimum resolution 1200–2400 dpi) were taken with a flatbed scanner and transferred to the program Image-Pro Plus program (version 4.5.0.29). The width of each ring was measured to 0.01 mm accuracy and the average annual width was calculated.
To cross-date the cores, out of 136 radiuses, 93 could be analyzed and their quality checked with the software COFECHA (Holmes 1983). Tree rings were cross dated within one tree and then between different trees, resulting in average intercorrelations for the sites. The validity of this procedure was checked using 30-year segments lagged by 15 years (critical value: 0.427). Two tree-ring chronologies, one from Blumenau and other from Joinville were calculated, representing the variations in mean radial increment of the stand over time, and since they showed a common climate signal, they were grouped in one regional chronology representing all the trees from both study areas (Blumenau + Joinville).
The final tree-ring width chronology was computed using the Arstan ARSTAN (V44h3) software package (Holmes 2001). The program removes trends (detrending) by curve fitting. A negative exponential function was used for detrending, dividing the ring width by the modelled curve and creating a tree-level index for each year. The ring width indices of each radius from all trees were averaged on a year by year basis to compute the mean index chronology (standard chronology) (Cook 1985). This removes portions of the variance not related to climatic factors by removing short-term variations related to forest dynamics (Cook and Peters 1981; Speer 2010). The ARSTAN also computed a prewhitened random mean index chronology using autoregressive modeling (residual chronology) (Cook 1985). For this study, the residual chronology used consisted of a series without autocorrelation (Speer 2010).
The residual chronology version was used for comparison between tree growth and climate. The inter-annual variability on ring width of the chronology was evaluated using mean sensitivity (MS), which quantifies the tree response to environmental factors. The quality of the chronology was evaluated by the value of EPS (expressed population signal), considering a threshold of 0.85 (Wigley et al. 1984). EPS values provide information that confirms the existence of common signals among trees, as well as the quality of the assessment of climate-growth responses (Mérian et al. 2013).
Climate data
Meteorological data (monthly precipitation and mean monthly temperature) from 1915 to 2013 were obtained from CEOPS/FURB (2015) and INMET (2015) and covered a period of 98 years.
Statistical analysis
DBH, age and mean tree-ring width were considered normal (p > 0.05; 95% significance level) when tested using the Shapiro–Wilk test (Shapiro and Wilk 1965). Mean and variance differences of the tree-ring widths between Blumenau and Joinville were investigated by t and F tests (Press et al. 1992).
The association and pattern of the relationship between DBH and age was tested using Pearson correlation and a non-linear regression model (fitted to a power function) with the software Past (V3.12) (Hammer et al. 2001).
A Pearson correlation at a 95% significance level examined the relationship between residual chronology and climate data (Fritts 1976; Blasing et al. 1984). Mean monthly temperatures and monthly precipitation (1915–2013) and from September of the previous growing season to May at the end of the current growing season (21 months) were used. This was to determine how the interannual variation correlated to the current year’s growth. Residual chronology was used for the correlation analysis and dendroclimatic study.
Results
Tree-ring distinctiveness
Nectandra oppositifolia has distinct growth rings marked by thickening and radial flattening of the latewood fiber walls in contrast to thin-walled fibers in the earlywood (Fig. 3a). Anatomical irregularities like pith eccentricity (Fig. 3b) and growth rings with diffuse boundaries were verified in 31.6% of the wood samples, causing difficulties in cross-dating. However, these anomalies did not prevent dating 68.4% (93 increment cores) of the samples, and at least one core of each tree was used.
Tree size measurements
Older trees occur in Joinville with an average age of 124 years, while in Blumenau, the trees are younger with an average age of 90 years. Average diameters in the two sites was similar, varying by approximately 20.0 cm (Table 1). Maximum and minimum diameters were also similar, with younger trees having the smallest diameters on both sites. The same relationship was not observed with older trees (28.7 cm in Blumenau and 24.4 cm in Joinville), showing a weak relationship between DBH and age (Fig. 4). For both sites, the minimum and maximum annual ring- width values were 0.1 and 4.7 mm a−1, respectively, and the average value was 0.7 mm a−1 for all trees. There was no significant difference between average annual ring width for the two sites (Fig. 5).
Synchronizing and periodicity of tree-ring formation
The analysis of tree rings indicates a synchronized growth pattern of Nectandra oppositifolia trees in the study areas. At least one radius from each tree of the 34 sampled was used for cross-dating analysis. The average time series composed specific chronologies (Fig. 6a, b) and were included in the regional chronology (Blumenau + Joinville) (Fig. 6c).
Thirty time series were used from the Blumenau trees and 63 from the Joinville trees, giving a total of 93 series that could be satisfactorily viewed, marked and synchronized. The total length of the series in Blumenau had an interval of 127 years (1887–2013), while in Joinville the range was 171 years (1843–2013). The average length of the regional series was 103 years, allowing an average intercorrelation of 0.611, which is above the critical value of 0.422. Both average intercorrelations of the two study areas reached the critical value (0.523 for Blumenau and 0.636 for Joinville). Samples of both areas had an average sensitivity of 0.418. The expressed population signal (EPS) values were higher than the threshold standard (0.85) from 1900 onwards (calculated with ARSTAN) (Table 2).
Tree rings and climate correlations
The correlation profiles between the chronology and temperature and precipitation are shown in Fig. 7a, b, respectively. These show interactions between the species and the regional climate.
The regional chronology (Blumenau + Joinville) was correlated with average temperatures, showing significant negative correlations in the previous summer and early fall during February, March, April and May (r = − 0.23, − 0.22, − 0.25 and − 0.22) (Fig. 7a).
Nectandra oppositifolia growth showed a significant negative correlation with precipitation in April during the fall of the current growing season (r = − 0.24) (Fig. 7b).
Discussion
The anatomical observations indicate that Nectandra oppositifolia forms distinct annual growth rings, as confirmed by previous studies of this species and others from the same family on different tropical forest sites in Brazil (Alves and Angyolossy-Alfonso 2000; Barros et al. 2006). Anatomical irregularities were characterized by the presence of pith eccentricities which cause deformities in the tree rings leading to the loss of some increment cores.
There was no relationship between DBH and tree age for both areas (i.e., older trees did not have the highest DBH). This may be due to intrinsic characteristics of the species and environmental conditions. Nectandra oppositifolia is an early/late secondary or even a climax species (Gandolfi et al. 1995; Pinto Sobrinho et al. 2009), and the ecological group of this species may explain its behavioral and strategic adjustments. The growth trajectory may also be related to spacing among the sampled trees, given that they live in a natural forest free of management. In this regard, dendrochronological analyses have proven their importance in increasing what is known about the growth patterns of tropical species by revealing growth trajectories (Rozendal and Zuidema 2011; Costa et al. 2015).
The average age of 125 years in Joinville may be due to the colonization process that occurred 162 years ago in 1851 (Mafra 2008). Knowing that N. oppositifolia shows the characteristics of a secondary species (Gandolfi et al. 1995; Pinto Sobrinho et al. 2009), and that the oldest individual is 170 years old, suggests that the arrival of immigrants and the construction of the Dona Francisca Road, which is less than 30 km from the study area, may be factors responsible for the establishment and development of this species in the region.
The average annual increment of N. oppositifolia trees is low compared to studies of other species of the same family and tropical environments. Spathelf et al. (2010) that found Ocotea pulchella (Lauraceae) and N. megapotamica trees with similar DBHs but younger than the ones in this present study. However, considering the high average age found for N. oppositifolia trees, the stabilizing growth trend related to the age of the trees can interfere with annual growth rates, which become smaller over the lifetime of a tree (Cook 1985), and justify the low values found in this study. Besides this, differences between the study areas (e.g., the phytoecological region, suppressing growth conditions, and climate and geomorphological variables) may have influenced the results, given that the soils in the study areas are different from those of the Spathelf et al. (2010) study.
Few dendrochronological studies have been performed in dense rain forest due to difficulties encountered by climate fluctuations, by a competition for light, and by differences in ages between sampled trees (Andreacci et al. 2013). However, in this study, most of the N. oppositifolia trees were older and appeared to have colonized the areas at the same time. The presence of anatomically distinct tree rings (enabling cross-dating among the trees), an average intercorrelation above the critical value of COFECHA (0.423, 99% of confidence level), and responses to climatic factors confirm the dendrochronological potential of N. oppositifolia (Stahle 1999).
The climate responses found in N. oppositifolia may be seen by the average sensitivity of the species, with an index of 0.42. Fritts (1976) states that indexes equal or greater than 0.400 indicate the presence of sensitivity to environmental variations, allowing dendrochronological studies to be conducted. The present work confirms this finding. In response to meteorological variables, the N. oppositifolia trees showed a significant correlation with average temperature and precipitation.
An increase in the average temperature during the previous growing season was harmful to the trees. The responses to temperature increase may be linked to climate changes in the area. Molion (2010) says that climate changes associated with the Pacific temperature oscillation may influence air temperatures and precipitation levels causing serious consequences to the environment. Trenberth (1997) estimated that since 1950 more than half of the years experienced events of the El Niño Southern Oscillation (ENSO) effect. Blumenau showed an average temperature increase of about 3 °C during the autumn, winter and spring months over the years, and an increase of about 2 °C in minimum temperatures (CEOPS/FURB 2015; INMET 2015); the summers became warmer and the winters milder.
Therefore, warmer summers have proved harmful to the growth of the species due to the response to water stress caused by an increase in evapotranspiration during warmer months, according to the findings of previous studies in subtropical forests (Oliveira et al. 2010). The average high temperatures in the summer and fall months are harmful to the growth of N. oppositifolia, corroborating the findings of Galbraith et al. (2010) that associated reduced carbon assimilation rates affect photosynthesis with an increase in autotrophic respiration (Clark 2007; Feeley et al. 2007). The increase in minimum temperatures throughout the year interferes with thermal amplitude, causing hotter summers and milder winters. This may interfere with behavior and species growth responses.
Phenological studies have identified an association between the phenophases of N. oppositifolia and variations in temperature. Spathelf et al. (2010) emphasize that phenophases and the presence of seasonal growth rings can be linked to the conservative character and genetic predispositions of species, considering that species of Lauraceae migrated from Cerrado environments where there are dry periods from May to September.
Precipitation influenced the growth of N. oppositifolia in a negative way during the early spring (current April). Excessive rain proved harmful to growth. Maia (2013) also found negative correlations between precipitation and the growth of Cedrela fissilis Vell. evaluated in nearby sites during the current January, which corroborates the results obtained for N. oppositifolia. This may be due to soil drenching as soil composition leads to erosive environments prone to flooding. Negative correlations between growth and precipitation may also occur due to increased clouds during rainy periods which reduce solar radiation and slow the development of trees (Clark and Clark 1994; Lovejoy and Schertzer 2006; Gamboa et al. 2011). Therefore, an increase in rainfall may be harmful to N. oppositifolia growth.
Conclusion
This work confirms the presence and distinctiveness of tree rings in Nectandra oppositifolia from dense rain forest in southern of Brazil. Growth was negatively influenced by hot temperatures and heavy precipitation. These results clarify environmental aspects related to the population age, average annual increment, climate trends, and adaptive responses in the face of environmental changes, generating valuable information that may be used to manage and conserve ecosystems. The interpretation of chronologies may help predict future scenarios, providing the basis for the development of new studies about this subject.
References
Alves ES, Angyolossy-Alfonso V (2000) Ecological trends in the wood anatomy of some Brazilian species, 1. Growth rings and vessels. IAWA J 21:3–30
Andreacci F, Botosso PC, Galvão F (2013) Sinais climáticos em anéis de crescimento de Cedrela fissilis em diferentes tipologias de florestas ombrófilas do sul do Brasil. Floresta 44(2):323–332
Ayoade JO (2007) Introdução a climatologia para os trópicos, 12th edn. Bertrand Brasil, Rio de Janeiro, p 332
Backes P, Irgang B (2004) Mata Atlântica. As árvores e a paisagem. Paisagem do Sul, Porto Alegre, p 396
Barros CF, Ferreira-Marcon ML, Callado CH, Lima HRP, Cunha M, Marquete O, Costa CG (2006) Tendências ecológicas na anatomia da madeira de espécie da comunidade arbórea da Reserva Biológica de Poço das Antas, Rio de Janeiro, Brasil. Rodriguésia 57(3):443–460
Bechara FC, Tiepo EN, Reis A (2009) Contribuição ao manejo sustentável do complex ferruginoso Nectandra na Floresta Nacional de Ibirama, SC. Rev Árvore 33:125–132
Blasing TJ, Solomon AM, Duvick DN (1984) Response function revisited. Tree Ring Bull 44:1–15
Botoso PC, Vetter RE, Tomazelo-Filho M (2000) Periodicidade e taxa de crescimento de árvores de cedro (Cedrela odorata L., Meliaceae), jacareúba (Callophyllum angulare A.C. Smith, Clusiaceae) e muirapiranga (Eperua bijuga Mart. ex Benth, Leg., Caesalpinoideae) de floresta de Terra Firme, em Manaus-AM. In: Roig FA (ed) Dendrocronologia en América Latina. Ediunc, Mendoza, pp 357–380
Brienen R, Zuidema P (2005) Relating tree growth to rainfall in Bolivian rainforests: a test for six species using tree ring analysis. Oecologia 146:1–12
Callado C, Guimarães RC (2010) Estudo dos anéis de crescimento de Schizolobium parahyba (Leguminosae: Caesalpinioideae) após episódio de mortalidade em Ilha Grande, Rio de Janeiro. Rev Bras Bot 33:85–91
Callado C, Neto SS, Scarano F, Costa C (2001) Periodicity of growth rings in some flood-prone trees of the Atlantic Rain Forest in Rio de Janeiro, Brazil. Trees 15:492–497
CEOPS/FURB (2015) Centro de Operação do Sistema de Alerta 392 da Bacia Hidrográfica do Rio Itajaí/Universidade Regional de Blumenau. http://ceops.furb.br. Accessed 03 July 2015
Clark DA (2007) Detecting tropical forests responses to global climatic and atmospheric change: current challenges and a way forward. Biotropica 39:4–19
Clark DA, Clark DB (1994) Climate-induced annual variation in canopy tree growth in a Costa Rican tropical rain forest. Tree Ring Bull 82:865–872
Cook ER (1985) A time series analysis approach to tree-ring standardization. University of Arizona, Tucson, Tucson, p 171
Cook ER, Peters K (1981) The smoothingspline: a new approach to standardizing forest interior tree-ring width series for dendroclimatic studies. Tree Ring Bull 41:45–53
Costa MS, Ferreira KEB, Botosso PC, Callado CH (2015) Growth analysis of five Leguminosae native tree species from a seasonal semidecidual lowland forest in Brazil. Dendrochronologia 36:23–32
Dünisch O (2005) Influence of the El-niño southern oscillation on cambial growth of Cedrela fissilis Vell. in tropical and subtropical Brazil. J Appl Bot Food Qual 79:5–11
EMBRAPA-Centro Nacional de Pesquisa de Solos (2006) Sistema Brasileiro de Classificação de Solos, 2nd edn. Emprapa-Centro Nacional de Pesquisa de Solos, Rio de Janeiro, p 306
Feeley KJ, Joseph Wright S, Nur Supardi MN, Kassim AR, Davies SJ (2007) Decelerating growth in tropical forest trees. Ecol Lett 10:461–469
Fritts HC (1976) Tree rings and climate. Academic Press, London, p 567
Galbraith D, Levy PE, Sitch S, Huntingford C, Cox P, Williams M, Meir P (2010) Multiple mechanisms of Amazonian forest biomass losses in three dynamic global vegetation models under climate change. New Phytol 187:647–665
Gamboa CCS, Rozendaal DMA, Ceccantini G, Angyalossy V, Van Der Borg K, Zuidema PA (2011) Evaluating the annual nature of juvenile rings in Bolivian tropical rainforest trees. Trees 25:17–27
Gandolfi S, Leitão-Filho HF, Bezerra CLF (1995) Levantamento florístico e caráter sucessional das espécies arbustivo-arbóreas de uma floresta mesófila semidecídua no município de Guarulhos, SP. Rev Bras Bot 55:753–767
Hammer Ø, Harper D, Ryan P (2001) Past: paleontological statistics software package for education and data analysis. Palaeontol Electron 4:01–09
Higuchi P, Reis MGF, Reis GG, Pinheiro AL, Silva CT, Oliveira CHR (2006) Composição florística da regeneração natural de espécies arbóreas ao longo de oito anos em um fragmento de Floresta Estacional Semidecidual, em Viçosa, MG. Rev Árvore 30(6):893–904
Holmes RL (1983) Computer-assisted quality control in tree-ring dating and measurement. Tree Ring Bull 43:69–78
Holmes RL (2001) Dendrochronology program library. Available from the Laboratory of Tree-Ring Research, University of Arizona, Tucson
IAWA Committee (1989) IAWA list of microscopic features for hardwood identification. IAWA Bull 10:219–232
INMET (2015) Instituto Nacional de Meteorologia. Séries meteorológicas 460 históricas para o Brasil. http://www.inmet.gov.br/portal/. Accessed 05 Oct 2015
Latorraca JVF, Souza MT, Silveira LD, Silva AB, Ramos LMA (2015) Dendrocronologia de árvores de Schizolobium parahyba (Vell.) S.F. Blake de ocorrência na Rebio de Tinguá-RJ. Rev Árvore 39:385–394
Lingner DV, Schorn LA, Vibrans AC, Meyer L, Sevegnani L, Gasper AL, Sobral MG, Kruger A, Klemz G, Schmidt R, Junior CA (2013) Fitossociologia do componente arbóreo/arbustivo da floresta ombrófila densa em Santa Catarina. In: Vibrans AC, Sevegnani L, Gasper AL, Lingner DV (eds) Inventário Florístico Florestal de Santa Catarina. Floresta Ombrófila Densa. Edifurb, Blumenau, pp 159–200
Lisi CS, Tomazello-Filho M, Botosso PC, Roig FA, Maria VRB, Ferreira-Fedele L, Voigt ARA (2008) Tree-ring formation, radial increment periodicity, and phenology of tree species from a seasonal semi-deciduous forest in southeast Brazil. IAWA J 29:189–207
Lovejoy S, Schertzer D (2006) Multifractals, cloud radiances and rais. J Hydrol 322:59–88
Mafra AD (2008) Aconteceu nos ervais: A disputa territorial entre Paraná e Santa Catarina pela exploração da erva mate - região sul do Vale do Rio Negro. Universidade do Contestado, Canoinhas, Brazil, p 150
Maia TM (2013) Estimativa da idade, avaliação do incremento e análise dendrocronológica de Cedrela fissilis Vell. (Meliaceae) em Santa Catarina. Universidade Regional de Blumenau, Blumenau, Brazil, p 73
Mérian P, Pierrat JC, Lebourgeois F (2013) Effect of sampling effort on the regional chronology statistics and climate–growth relationships estimation. Dendrochronologia 31:58–67
Molion LCB (2010) Variabilidade e alterações climáticas. In: Figueiredo T, Ribeiro LF, Ribeiro AC, Fernandes LF (eds) Clima e recursos naturais. Instituto Politécnico de Bragança, Bragança, pp 17–41
Oliveira JM, Roig FA, Pillar VD (2010) Climatic signals in tree-rings of Araucaria angustifolia in the southern Brazilian highlands. Aust Ecol 35:134–147
Pinto Sobrinho F de A, Christo AG, Guedes-Bruni RR, Silva AF (2009) Composição florística e estrutura de um fragmento de floresta estacional semidecidual Aluvial em Viçosa (MG). Rev Floresta 39:793–805
Press WH, Teukolsky SA, Vetterling WT, Flannery BP (1992) Numerical recipes in C, 2nd edn. Cambridge University Press, Cambridge
Quinet A (2006) Lauraceae na Reserva Biológica de Poço das Antas, Silva Jardim, Rio de Janeiro, Brasil. Rodriguésia 57(3):543–568
Rozendal DMA, Zuidema PA (2011) Dendroecology in the tropics: a review. Trees 25(1):3–16
Schöngart J, Piedade MTF, Ludwigshausen S, Hornas V, Worbes M (2002) Phenology and stem-growth periodicity of tree species in Amazonian floodplain forests. J Trop Ecol 18:581–597
Schweingruber FH (2007) Wood structure and environment. Springer, Berlin, p 279
Shapiro SS, Wilk MB (1965) An analysis of variance test for normality (complete samples). Biometrika 52:591–611
Shimamoto CY, Botosso PC, Amano E, Marques MCM (2015) Stem growth rhythms in trees of a tropical rainforest in Southern Brazil. Trees 30:99–111
Spathelf P, Tomazello-Filho M, Tonini H (2010) Dendroecological analysis of Ocotea pulchella and Nectandra megapotamica on two sites near Santa Maria, Rio Grande do Sul (Brazil). Floresta 40:777–788
Speer JH (2010) Fundamentals of tree-ring research. The University of Arizona Press, Arizona, p 509
Stahle DW (1999) Useful strategies for development of tropical tree-ring chronologies. IAWA J 20:249–253
Trenberth KE (1997) The definition of El Niño. Bull Am Meteor Soc 78:2771–2777
Vibrans AC, Sevegnani L, Gasper AL, Lingner DV (2013) Inventário Florístico Florestal de Santa Catarina. Floresta Ombrófila Densa. Edifurb, Blumenau, p 576
Wigley TM, Briffa KR, Jones PD (1984) On the average value of correlated time series, with applications in dendroclimatology and hydrometeorology. J Climate Appl Meteorol 23:201–213
Worbes M (1999) Annual growth rings, rainfall-dependent growth and long-term growth patterns of tropical trees from the Caparo Forest Reserve in Vezenuela. J Ecol 87:391–403
Worbes M (2002) One hundred years of tree-ring research in the tropics—a brief history and an outlook to future challenges. Dendrochronologia 20:217–231
Zanon MMF, Goldenberg R, Moraes PLR (2009) O gênero Nectandra Rol. ex Rottb. (Lauraceae) no Estado do Paraná, Brasil. Act Bot Bras 23:22–35
Zuidema PA, Baker PJ, Groenendijk P, Schippers P, Van der Sleen P, Vlam M, Sterck F (2013) Tropical forests and global change: filling knowledge gaps. Trends Plant Sci 18:413–419
Acknowledgements
The authors thank the Universidade Regional de Blumenau (FURB) and CAPES for granting the master’s scholarship to the first author, researcher Joanna Rebello de Oliveira for helping with fieldwork, researcher Adilson Nicolleti for assistance with fieldwork and making the maps, and the journal reviewers for their useful comments. This paper was developed from research conducted by the first author while completing a master’s degree at the wood anatomy and dendrochronology laboratory at FURB.
Author information
Authors and Affiliations
Corresponding author
Additional information
The online version is available at http://www.springerlink.com
Corresponding editor: Zhu Hong.
Rights and permissions
About this article
Cite this article
Granato-Souza, D., Adenesky-Filho, E. & Esemann-Quadros, K. Dendrochronology and climatic signals in the wood of Nectandra oppositifolia from a dense rain forest in southern Brazil. J. For. Res. 30, 545–553 (2019). https://doi.org/10.1007/s11676-018-0687-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-018-0687-5