Abstract
Soil fauna decompose litter, whereas land use changes may significantly alter the composition and structure of soil fauna assemblages. However, little is known of the effects of land-use on the contribution of soil fauna to litter decomposition. We studied the impacts of soil fauna on the decomposition of litter from poplar trees under three different land uses (i.e. poplar-crop integrated system, poplar plantation, and cropland), from December 2013 to December 2014, in a coastal area of Northern Jiangsu Province. We collected litter samples in litterbags with three mesh sizes (5, 1 and 0. 01 mm, respectively) to quantify the contribution of various soil fauna to the decomposition of poplar leaf litter. Litter decomposition rates differed significantly by land use and were highest in the cropland, intermediate in the poplar-crop integrated system, and lowest in the poplar plantation. Soil fauna in the poplar-crop integrated system was characterized by the highest numbers of taxa and individuals, and highest Margalef’s diversity, which suggested that agro-forestry ecosystems may support a greater quantity, distribution, and biodiversity of soil fauna than can single-species agriculture or plantation forestry. The individuals and groups of soil fauna in the macro-mesh litterbags were higher than in the meso-mesh litterbags under the same land use types. The average contribution rate of meso- and micro-fauna to litter decomposition was 18.46%, which was higher than the contribution rate of macro-fauna (3.31%). The percentage of remaining litter mass was inversely related to the density of the soil fauna (P < 0.05) in poplar plantations; however, was unrelated in the poplar-crop integrated system and cropland. This may have been the result of anthropogenic interference in poplar-crop integrated systems and croplands. Our study suggested that when land-use change alters vegetation types, it can affect species composition and the structure of soil fauna assemblages, which, in turn, affects litter decomposition.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Soil fauna decompose litter (Yin et al. 2002; Yang and Zou 2006; Li et al. 2009; Wood et al. 2012; Liu et al. 2013; Wu 2013; Wang et al. 2015), may influence litter decomposition rates, nutrient cycling, primary productivity, and subsequently impact the nutrient cycling of bulk soils and the health of the entire soil ecosystem (Seastedt 1984; Chauvel et al. 1999; Lavelle 2002; Xuluc-Tolosa et al. 2003; Moore et al. 2004). This occurs directly through the physical breakdown and digestion of litter, and indirectly, by altering the structure and function of microbe populations(Maraun and Scheu 1996; De Deyn et al. 2003; Lin et al. 2004; Ekschmitta et al. 2005; Wang et al. 2010; He et al. 2015b).
The composition and activity of soil fauna may be altered via changes in environmental factors (Read and Perez-Moreno 2003; Joo et al. 2006; Yang and Chen 2009; Yin et al. 2010; Wang et al. 2011; He et al. 2015a, b). The dynamics of soil fauna community diversity can change with elevation (Wang et al. 2009, 2010; Illig et al. 2010), climate (Wall et al. 2008), and vegetation type (Franklin et al. 2005). Further, soil fauna are influenced by land-use (Emmerling 1995; Wu et al. 2006, 2009). Soil fauna research under different land-uses has concentrated on urban ecosystems (Zhang et al. 2011; Wu et al. 2006; Liu et al. 2011), tropical rain forests (Deng et al. 2003), wetlands (Wang et al. 2005), and grasslands (Lin et al. 2012). To date, research has been limited on soil fauna under different land-uses in plantation forestry. Change in land-use often results in modifications of the physical and chemical properties of soils, such as changes in bulk density, fertility, or moisture. These changes in soil properties are the primary factors that influence soil fauna populations (Deng et al. 2003). The abundance and taxa representation of soil fauna will vary contingent on the land usage (Wu et al. 2006; Emmerling C 1995) as well as the degree of human disturbance (Wang et al. 2005). Land-use management may be conducive to the improvement of habitats for soil microorganisms (Geissen et al. 2009) and can enrich the diversity of soil fauna (Ammer et al. 2006).
Soil fauna of different body sizes contribute variably to litter decomposition (Yin et al. 2002; Lin et al. 2005; Li et al. 2011; Xia et al. 2012; Fan et al. 2014; Bao et al. 2015). For this reason, litterbags of different mesh sizes have been employed to study the effects of soil fauna body size on litter decomposition (Irmler 2000; Hunter et al. 2003; Wang et al. 2009, 2010; Bao et al. 2015).
Plantation foresters in China face challenges that are caused by single-species strategies, reduced tree resistance to disease, and instability (Chen et al. 2014; Liu et al. 2010). These challenges bring into question the sustainability of plantation forestry, and can degrade the integrity of ecological environments. Strategies for the transformation of land-use in plantation forests are essential toward the resolution of these issues. Dongtai Forest Farm is a protected coastal forest in Jiangsu Province, comprising one of several excellent poplar plantation distribution areas in China. Poplar plantations, poplar-crop integrated systems, and croplands are the primary land-use types in the coastal area of Northern Jiangsu Province, China. Changes in land-use types, land management methods, the use of pesticides and fertilizers, soil properties, and litter have led to soil habitat changes that may initiate structural changes in soil fauna communities. To quantify the effects of land-use and fauna on the decomposition of litter, we studied litter decomposition dynamics under three land-use models. We studied the role of decomposers (macro-, meso-, and micro-fauna) in the mass loss of litter. Our objective was to determine the effects of, and the interactions between, land-use and faunal decomposers. We addressed two questions: (1) How are the changes in soil fauna communities and litter decomposition associated with land-use? (2) What are the differences in the contribution rates of soil fauna of different sizes on decomposition under various land-uses?
Materials and methods
Study sites and experimental design
Three sampling sites under different land-uses, viz. poplar-crop integrated system, poplar plantation, and cropland, were established at a Dongtai Forest Farm in Northern Jiangsu Province, China (120°49′E, 32°52′N). The Dongtai Forest Farm is a key protected coastal forest in Jiangsu Province, which is located in a maritime transitional zone monsoon climate, with an annual average temperature of 14.6 °C, annual average relative humidity of 88.3%, a frost-free period of 220 d, with a yearly average rainfall of 1050 mm. The primary soil type in the Dongtai Forest Farm is desalting meadow soil, with the texture of sandy loam; the pH of the soil is alkaline, and the soil salinity is 1.1–2.1 gkg−1. The Dongtai Forest Farm is dominated by poplar trees (Populus deltoids), which cover an area of ~ 3000 ha; the coverage rate of the forests has attained 85%, timber reserves are ~ 5 × 104 m3, and it comprises one of several excellent poplar plantation distribution areas in China (Table 1).
(1) Poplar-crop integrated system (PC). For our experiment we selected a poplar plantation compound management pattern sample area of poplar forest (Populus deltoides CV. 35); corn, peanuts, and cotton were cyclically grown during the summer, while wheat was grown in winter, at the center of the poplar plantation. The average tree height was 23.45 m, while the average DBH was 21.21 cm. The shrub layers included Morus Alba, while the herb layers included Humulus scandens, Erigeron annuus, Carpesium abrotanoides, Achyranthes bidentata, Conyza Canadensis, and Trichosanthes kirilowii. (2) Poplar plantation (P). We selected a poplar plantation, which consisted of poplar trees (Populus deltoides CV. 35), with afforestation density of 5 m × 8 m, and a forest canopy cover of 0.7. The average tree height was 22.78 m and the average DBH was 24.63 cm. The shrub layers included Morus Alba, while the herb layers included Oplismenus undulatifolius, Cayratia japonica, Roegneria kamoji Ohwi, Erigeron annuus, Conyza Canadensis, Myosoton aquaticum, and Achyranthes bidentata. (3) Cropland (C). Corn, peanuts, and cotton were cyclically grown during the summer, while wheat was grown in the winter.
Experimental decomposition design
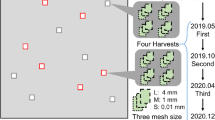
For this study, the pure poplar plantations, poplar-crop integrated systems, and croplands with similar site conditions were sampled and replicated in quadruplicate, with a spatial interspersion distance of approximately 600–800 m for the same land use. We established an experimental plot (20 × 30 m) within each land use sample, and a quadrat (3 m × 3 m) was set up within the central area of each plot. The different land uses in this region possessed the same basalt parent materials, and similar altitudes (less than 5 m altitude difference), respectively. The decomposition experiment was initiated in December, 2013, and continued until December, 2014. We collected recently senesced poplar leaves at the peak of fall in November, 2013, and placed approximately10 g of oven-dried litter in nylon litterbags (15 cm × 20 cm). The leaf litter samples were oven-dried at 60 °C to establish the relationship between air-dry and oven-dry mass. According to the classification standard of soil fauna body size (Swift et al. 1979); three types of litterbags were selected: (1) micro-mesh litterbags (0.01 mm, permitting the entry of microorganisms only), (2) meso-mesh litterbags (1 mm, permitting the entry of micro- and meso-fauna) and (3) macro-mesh litterbags (5 mm, permitting the entry of micro-, meso-, and macro-fauna). In order to minimize the loss of leaf litter, the 5 mm and 1 mm litterbags had 0.01 mm diameter mesh patches sewn to the bottom.
A total of 144 litterbags were placed at the three land use sites (PC, P, and C), where 48 litterbags were used for each land use site, and 12 litterbags were randomly placed into each quadrat (3 m × 3 m) in December, 2013. Every 3 months, 12 litterbags were collected at random from each land use site. The roots, soil, and weeds that had entered the litterbags were carefully removed in the laboratory. We initially transferred the macro-fauna by hand, after which a Tullgren funnel was employed to separate the soil fauna. All isolated soil fauna specimens were preserved in a 75% ethanol solution. Most of the soil fauna were identified by orders (Yin et al.1998), and imago and larva were classified in the same category. Following the separation of the soil fauna, all collected litterbags were cleaned, oven-dried at 60 °C and weighed to determine the remaining litter mass.
Data analyses
Diversity, Abundance, and Group Number were calculated for fauna communities at the three sites. The abundances of soil fauna were divided according to the percentage of the total number of all the species of soil fauna acquired, which accounted for more than 10% as the dominant groups, 1–10% for the common groups, whereas < 1% were rare taxa. The diversity of soil fauna is an excellent indicator as relates to richness and evenness, which not only reflects differences in composition, structure, function, and dynamics of the communities, but may also reveal relationships between different natural geographical conditions and soil fauna. The diversity of the soil fauna was analyzed using the Simpson dominance index, Shannon–Wiener index, Pielou's evenness index, and Margalef index. The Simpson dominance index (C, Simpson 1949), Shannon–Wiener index (H′, Shannon 1948), Pielou's evenness index (E, Pielou 1967) and Margalef index (D, Margalef 1958) were calculated as follows: Simpson dominance index (C)
Shannon–Wiener index (H’):
Pielou's evenness index (E):
Margalef index(D):
where S is the total number of species in the community, and P i is the proportion of individual species in the community.
The similarities of two communities were calculated using Jaccard’s similarity index (J′, Jaccard 1908):
where a and b are the number of species present in community 1 and community 2, respectively, and c is the total number of species present in both communities.
The relative mass loss contributed by the fauna was calculated as (Seastedt 1984):
where L fauna is the litter mass loss contributed by the fauna calculated by macro-mesh meso-mesh, and micro-mesh litterbag mass loss, L total is the macro-mesh litterbag mass loss contributed by most biological, microbiological, and abiotic factors of the soil fauna.
Repeated measures analysis of variance (ANOVA) was applied to determine the differences in litter decomposition among the different mesh sizes, and land uses. Pearson’s correlation coefficients were employed to express the relationships of the mass loss values with relative individual and group soil fauna density. Simpson dominance index, Shannon–Wiener index, Pielou’s evenness index, and Margalef index statistical analyses were performed using SPSS 20.0 software.
Results
Effects of mesh size on leaf litter decomposition under different land uses
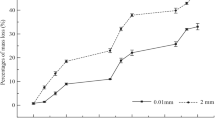
Mesh sizes significantly impacted the decomposition rate of the poplar litter (Fig. 1). The primary mass remaining rates were 0.01 mm (56.80%) > 1 mm (46.43%) > 5 mm (44.67%), and highly significant differences existed among the micro-mesh, macro-mesh, and meso-mesh litterbags (P < 0.01) under the same land use; however, the primary mass remaining rates were similar at the macro-mesh and meso-mesh litterbags(P = 0.053).
Under different land uses, the primary mass remaining rates demonstrated extremely significant differences (P < 0.01) in the macro-mesh litterbags. There were considerable differences in the meso-mesh litterbags among the poplar plantation, cropland (P < 0.01), and poplar-crop integrated system (P < 0.05). However, there was similarity between the cropland and poplar-crop integrated system (P = 0.058). In the micro-mesh litterbags, there were significant differences (P < 0.05) between the cropland and poplar plantation.
Soil fauna community characteristics under different land uses
Composition of soil fauna communities under different land uses
Species composition, quantity distribution, and the biodiversity of soil fauna exhibited certain differences in poplar-crop integrated systems, poplar plantations, and croplands (Figs. 2, 3). A total of 13,272 individuals were collected, belonging to four phyla, 12 classes, and 32 groups (including suborders), including poplar-crop integrated systems (4859) > croplands (4517) > poplar plantations (3896). The number of groups was as follows: poplar-crop integrated systems (32) > poplar plantations (29) > croplands (25). Acarina and Collembola were the dominant genera in this area, and the combined population of the two groups comprised 82.51% of the total number of individuals, which constituted the body of the soil fauna community in a coastal area of Northern Jiangsu Province.
The common groups were as follows: Araneae, Coleoptera, Isopoda, Diptera, Hymenoptera, Nematode, Scolopendromorpha, and Lepidoptera, which accounted for 14.99% of the total number of individuals. The other groups were rare taxa, whose individual numbers accounted for only 2.50% of the total; however, the number of groups accounted for 62.50% of the total number of groups. The differences between common and rare taxa were significantly greater than the dominant genera under different land uses. The individuals and groups of soil fauna in the macro-mesh litterbags were higher than in the meso-mesh litterbags under the same land use types. Soil fauna individuals in macro-and meso-mesh litterbags were significantly different, whereas poplar-crop integrated system (P < 0.01), poplar plantation and cropland (P < 0.05), and soil fauna groups were significantly different (P < 0.01) under various land uses.
However, under different land use, soil fauna resident individuals in macro-mesh litterbags exhibited significant differences only between the poplar plantation and cropland (P < 0.05), whereas the others were similar between poplar-crop integrated system and poplar plantation(P = 0.404),and cropland (P = 0.060).In the meso-mesh litterbags there were significant differences between the poplar plantation and cropland (P < 0.05), and poplar-crop integrated system (P < 0.01); however, the cropland and poplar-crop integrated system were similar (P = 0.177). The soil fauna groups in macro-mesh litterbags were significantly different between the poplar-crop integrated system, poplar plantation, and cropland (P < 0.01); however, in the meso-mesh litterbags, there were no significant differences (P > 0.05). This indicated no significant differences in the meso- and micro- fauna groups under different land usages.
Index of soil fauna communities under different land uses
Using the Simpson dominance index, Shannon–Wiener index, Pielou’s evenness index, and Margalef index analyses of the diversity of soil fauna, the results indicated that community diversity and the complexity of soil fauna were different among the poplar-crop integrated system, poplar plantation, and cropland (Fig. 4). The results revealed that the Simpson dominance index was highest in the cropland; the Pielou evenness index was the highest in the poplar plantation; the Margalef abundance index, Shannon diversity index were the highest in the poplar-crop integrated system. Soil fauna in the poplar-crop integrated system was characterized by the highest numbers of taxa and individuals, and highest Margalef’s diversity, which suggested that agro-forestry ecosystems may support a greater quantity, distribution of soil fauna than single-species agriculture or plantation forestry.
Under different land uses, the Simpson dominance index in the macro-mesh litterbags had extremely significant differences (P < 0.01) between the cropland and poplar-crop integrated system, and poplar plantation, but no significant difference between the poplar-crop integrated system and cropland(P = 0.885). The Shannon–Wiener index showed extremely significant differences among the three sites (P < 0.01); Pielou’s evenness index had significant differences between only the poplar plantation and cropland (P < 0.01). The Margalef index had extremely significant differences (P < 0.01) between the poplar-crop integrated system and cropland, and poplar plantation, but no significant difference between the poplar plantation and cropland(P = 0.231). However, in the meso-mesh litterbags, the Simpson dominance index, Shannon–Wiener index, Pielou’s evenness index and Margalef index showed no significant differences among the three sites.
For the same sites, only the Margalef index showed significant differences between the macro-mesh and meso-mesh litterbags (P < 0.01) in the poplar-crop integrated system, poplar plantation, and cropland, respectively; Simpson dominance index showed differences between the macro-mesh and meso-mesh litterbags (P < 0.05) in the poplar-crop integrated system; Shannon–Wiener index showed significant differences between the macro-mesh and meso-mesh litterbags in the poplar-crop integrated system (P < 0.01), and poplar plantation (P < 0.05); and Pielou’s evenness index showed no major differences between macro-mesh and meso-mesh litterbags in three sites.
The similarity of two communities was calculated using Jaccard’s similarity index. The highest similarity index was 0.9063, between the poplar-crop integrated system and the poplar plantation, followed by the poplar-crop integrated system and the cropland 0.7813, with the lowest being 0.6875, between the poplar plantation and cropland.
The contribution of soil fauna to decomposition
Following 1 year of incubation in the field, the contribution of macro-fauna to litter decomposition was evident in the poplar-crop integrated system (2.72%) < , cropland (3.00%) < , and poplar plantation (4.20%), while the contribution of meso-and micro- fauna to litter decomposition was the poplar-crop integrated system (19.06%) > , poplar plantation (18.59%) > , and cropland (17.72%) (Fig. 5). In general, the average contribution rate of soil fauna to litter decomposition was 21.76%. The average contribution rate of meso-and micro- fauna to litter decomposition was 18.46%, which was much higher than the contribution rate of macro-fauna to litter decomposition (3.31%), which had extremely significant differences (P < 0.01). This indicated that the role of meso- and micro- fauna on litter decomposition was more significant than the macro-fauna in the poplar-crop integrated system, cropland, and poplar plantation.
Correlation between mass remaining rate and soil fauna
There were negative relationships of the percentage of litter mass remaining with the individual densities of soil fauna (P < 0.05) in macro-mesh and meso-mesh litterbags in the poplar plantation. However, there were negative relationships of the mass remaining rate with the individual density of soil fauna (P < 0.01) in the macro-mesh litterbags in September and December, and in the meso-mesh litterbags in June and December, in the poplar-crop integrated system. Further, there was no significant correlation with the soil fauna in the macro-mesh litterbags, or in the meso-mesh litterbags, except for December, where there was a significant negative correlation (P < 0.05) in cropland. The soil fauna groups, Simpson dominance index, Shannon–Wiener index, Pielou’s evenness index, and Margalef index had no obvious correlations with the litter mass remaining rate (Tables 2, 3).
These results demonstrated that there were negative relationships between the mass remaining rate and the individual density of the soil fauna in the poplar plantation. However, no significant correlations between the litter mass remaining and the individual density of soil fauna were found in the poplar-crop integrated system, and cropland. This may have been the result of human anthropogenic interference in both the poplar-crop integrated system, and cropland.
Discussion
Land use effects
In this study, the species composition, quantity distribution, and biodiversity of soil fauna revealed certain differences between the poplar-crop integrated system, poplar plantation, and cropland. Due to their robust migration and activity, soil fauna were more vulnerable to the influences of environmental factors (Xu et al.2012). Different land uses altered the quantity and quality of the litter input (Ruan et al. 2005; Ge et al. 2012; David et al. 2012; Galizzi et al. 2012), the physical and chemical properties of the soil (Deng et al. 2003), and subsequently affected the species composition and quantity distribution of the soil fauna (Koehler and Born 1989). The groups, individuals, and Margalef index of soil fauna in the poplar-crop integrated system were the highest, which indicated that agro-forestry ecosystems may improve the quantity distribution and biodiversity of soil fauna to some extent. The cropland altered some of the physical and chemical properties of the soil, as well as a proportion of important soil organic matter (Mando et al. 2005; Wei et al. 2006; Hati et al. 2007), which thus impacted the community characteristics of the soil fauna (Cutz-Pool et al. 2007; Liang et al. 2009), and initiated decreases in the soil fauna diversity and the higher Simpson dominance index.
For this study, Acarina, and Collembola were the dominant genera, and the combined population of the two groups attained 82.51% of the total number of individuals. This constituted the bulk of the soil fauna community in a coastal area of Northern Jiangsu Province; however, the Acarina composition was not consistent. The Oribatida population in the poplar plantation was higher than in the poplar-crop integrated system and cropland, which may have been associated with the environmental sensitivity of Oribatida. Farmland comprised the most seriously anthropogenically impacted ecosystem; cultivation, crop rotation, and the use of pesticides eliminated some of the more sensitive species, such as Oribatida (Cao 2007).
Mesh size effects
Different litterbag mesh-sizes may limit the entry of specific soil fauna, segregate different sized soil fauna, and have small differences in the microenvironment (Bokhorst and Wardle 2013); hence, the use of different litterbag mesh sizes to quantify the soil fauna involved with litter decomposition is quite reliable(Swift et al. 1979). In this study, the mass loss rates of leaf litter appeared consistent with the taxa and individual relative density of the soil fauna in litterbags with different mesh sizes. The primary mass remaining rates were 0.01 mm (56.80%) > 1 mm (46.43%) > 5 mm (44.67%), which indicated soil fauna and microbes > meso- and micro- fauna and microbes > only microbes. This result was consistent with the conclusions of previous research (Yin et al. 2002; Yang and Zou 2006; Wu et al. 2010; Xia et al. 2012; Fan et al. 2014; Yu et al.2015). The reason was that litterbags with different mesh sizes limited the entry of soil fauna, and influenced the composition and structure of the decomposition food webs. Macro-meshed litterbags were conducive to the ingress and egress of macro-fauna, thus it promoted litter decomposition (Bradford et al. 2002), whereas smaller meshed litterbags limited the soil fauna ingress and egress, and thereby slowed the litter decomposition rate. Litter was observed to decompose slowly in the micro-mesh litterbags, due to the lack of soil fauna. These results demonstrated that soil fauna played a critical role in litter decomposition in the coastal areas of North Jiangsu Province.
Soil fauna promoted litter decomposition; however, the contribution rate of soil fauna to litter decomposition was different from that of other fauna. In this study, the average contribution rate of meso- and micro- fauna to litter decomposition was much higher than the contribution rate of macro-fauna. This result was consistent with the conclusions of previous research (Wang et al. 2011; Fan et al. 2014; Bao et al. 2015), but inconsistent with Xia et al. (Xia et al. 2012).However, due to the leakage of litter, the contribution rate of soil fauna to litter decomposition may have been somewhat overestimated (Yang and Zou 2006). Also, under naturally ambient conditions, fresh litter would constantly be added to the process of litter decomposition, and there would be no limit to the soil fauna; hence, changes in soil fauna and its effects on litter decomposition were more complex.
In summary, our experiment demonstrated that soil fauna under different land uses played a critical role in litter decomposition; the contributions of soil fauna of different sizes were different under varied land use in a coastal area of Northern Jiangsu Province, China. However, under practical conditions, in addition to anthropogenic interference, there are likely further potential factors that influence litter decomposition via microorganisms, climate (temperature, lighting, rainfall), site conditions (soil fertility, elevation, slope aspect), and litter properties. Therefore, these factors should be combined with environmental gradient data for further analysis.
References
Ammer S, Weber K, Abs C, Ammer C, Prietzel J (2006) Factors influencing the distribution and abundance of earthworm communities in pure and converted Scots pine stands. Appl Soil Ecol 33:10–21
Bao J, Yin X, Li X (2015) Study on the contribution of soil fauna to litter decomposition of Rhododendron chrysanthum in the Changbai Mountains. Acta Ecol Sin 35:1–12
Bokhorst S, Wardle DA (2013) Microclimate within litter bags of different mesh size: implications for the ‘arthropod effect’ on litter decomposition. Soil Biol Biochem 58:147–152
Bradford MA, Tordoff GT, Jones TH, Newington JE (2002) Microbiota, fauna, and mesh size interactions in litter decomposition. Oikos 99:317–323
Cao Z (2007) Soil ecology. Chemical Industry Press, Beijing
Chauvel A, Grimaldi M, Barros E, Blanchart E, Desjardins T, Sarrazin M, Lavelle P (1999) Pasture damage by an Amazonian earthworm. Nature 398:32–33
Chen XL, Ju Q, Lin KL (2014) Development status, issues and countermeasures of China's plantation. World For Res 27:54–59
Cutzpool LQ, Palaciosvargas JG, CastañOmeneses G, GarcíAcalderón NE (2007) Edaphic Collembola from two agroecosystems with contrasting irrigation type in Hidalgo State, Mexico. Appl Soil Ecol 36:46–52
David T, Peter BR, Forest I (2012) Biodiversity impacts ecosystem productivity as much as resources, disturbance or herbivory. Proc Natl Acad Sci 109:10394–10397
De Deyn GB, Raaijmakers CE, Zoomer HR, Berg MP, de Ruiter PC, Verhoef HA, Martijn BT, Van der Putten WH (2003) Soil invertebrate fauna enhances grassland succession and diversity. Nature 422:711–713
Deng X, Zou S, Fu X, Yao T, Sheng C, Bai Z (2003) The impacts of land use practices on the communities of soil fauna in the Xishuangbanna rain forest, Yunnan, China. Acta Ecol Sin 23:130–138
Ekschmitta K, Liu M, Vettera S, Foxa O, Woltersa V (2005) Strategies used by soil biota to overcome soil organic matter stability—why is dead organic matter left over in the soil? Geoderma 128:167–176
Emmerling C (1995) Long-term effects of inundation dynamics and agricultural land-use on the distribution of soil macrofauna in fluvisols. Biol Fertil Soils 20:130–136
Fan H, Wang S, Ruan H, Tan Y, Zheng A, Xu Y, Xu K, Cao G (2014) Effects of soil fauna on litter decomposition and its community structure under different land use patterns in coastal region of northern Jiangsu province. J Nanjing For Univ (Natl Sci Ed) 38:1–7
Franklin E, Magnusson WE, Luizão FJ (2005) Relative effects of biotic and abiotic factors on the composition of soil invertebrate communities in an Amazonian savanna. Appl Soil Ecol 29:259–273
Galizzi MC, Zilli F, Marchese M (2012) Diet and functional feeding groups of Chironomidae (Diptera) in the Middle Paraná River floodplain (Argentina). Iheringia Série Zoologia 102:117–121
Ge B, Zhang D, Zhang H, Li Z, Liu Z, Zhou C, Tang B (2012) Community structure and functional groups of soil macrofauna in urban green spaces of Yancheng city, Jiangsu province in spring. Chin J Ecol 31:87–92
Geissen V, Peña-Peña K, Huerta E (2009) Effects of different land use on soil chemical properties, decomposition rate and earthworm communities in tropical Mexico. Pedobiologia 53:75–86
Hati KM, Swarup A, Dwivedi AK, Misra AK, Bandyopadhyay KK (2007) Changes in soil physical properties and organic carbon status at the topsoil horizon of a vertisol of central India after 28 years of continuous cropping, fertilization and manuring. Agric Ecosyst Environ 119:127–134
He R, Chen Y, Deng C, Yang W, Zhang J, Liu Y (2015a) Seasonal responses of the soil meso- and microfauna to litter decomposition in alpine meadow of western Sichuan. Chin J Appl Environ Biol 21:350–357
He R, Chen Y, Deng C, Yang W, Zhang J, Liu Y (2015b) Litter decomposition and soil faunal diversity of two understory plant debris in the alpine timberline ecotone of western Sichuan in a snow cover season. Chin J Appl Ecol 26:723–731
Hunter MD, Adl S, Pringle CM, Coleman DC (2003) Relative effects of macroinvertebrates and habitat on the chemistry of litter during decomposition. Pedobiologia 47:101–115
Illig J, Norton RA, Scheu S, Maraun M (2010) Density and community structure of soil- and bark-dwelling microarthropods along an altitudinal gradient in a tropical montane rainforest. Exp Appl Acarol 52:49–62
Irmler U (2000) Changes in the fauna and its contribution to mass loss and N release during leaf litter decomposition in two deciduous forests. Pedobiologia 44:105–118
Jaccard P (1908) Nouvelles recherches sur la distribution florale. Bulletin De La Societe Vaudoise Des Sciences Naturelles 44:223–270
Joo SJ, Yim MH, Nakane K (2006) Contribution of microarthropods to the decomposition of needle litter in a Japanese cedar (Cryptomeria japonica D. Don) plantation. For Ecol Manage 234:192–198
Koehler H, Born H (1989) The influence of vegetation structure on the development of soil mesofauna. Agric Ecosyst Environ 27:253–269
Lavelle P (2002) Functional domains in soils. Ecol Res 17:441–450
Li J, Zhang W, Liao C, Yang Y, Fu S (2009) Responses of earthworms to organic matter at different stages of decomposition. Pedosphere 19:382–388
Li Y, Luo C, Yang W, Hu J, Wu F (2011) Decomposition of eucalyptus-alder mixed litters and dynamics of soil faunal community. Chin J Appl Ecol 22:851–856
Liang W, Lou Y, Li Q, Zhong S, Zhang X, Wang J (2009) Nematode faunal response to long-term application of nitrogen fertilizer and organic manure in Northeast China. Soil Biol Biochem 41:883–890
Lin B, Liu Q, Wu Y, He H (2004) Advances in the studies of forest litter. Chin J Ecol 23:60–64
Lin Y, Zhang F, Liu H, Su H (2005) Fluctuation of soil fauna community in Baiwangshen during paper Mulberry leaf litter decomposition. Chin J Zool 40:60–66
Lin G, Zhao F, Chen G, Chen S, Su J, Zhang T (2012) Effects of different land-use types on larger-size soil animal communities in the northern region of Qinghai Lake. Acta Prataculturae Sin 21:180–186
Liu Q, Yin H, Cheng X, Lin B, Hu R, Zhao C, Yin C (2010) Problems and strategies of sustainable regeneration of plantation ecosystem in China. World For Res 23:71–75
Liu Y, Zhang A, Yan Y, Li K, Fang Y (2011) Diversity of soil animal community under different land-use types in Chongming Island. J Fudan Univy (Natl Sci) 50:288–295
Liu R, Li W, Yang W, Tan B, Wang W, Xu Z, Wu F (2013) Contributions of soil fauna to litter decomposition in alpine/subalpine forests. Chin J Appl Ecol 24:3354–3360
Mando A, Ouattara B, Sédogo M, Stroosnijder L, Ouattara K, Brussaard L, Vanlauwe B (2005) Long-term effect of tillage and manure application on soil organic fractions and crop performance under Sudano-Sahelian conditions. Soil Tillage Res 80:95–101
Maraun M, Scheu S (1996) Changes in microbial biomass, respiration and nutrient status of beech (Fagus sylvatica) leaf litter processed by millipedes (Glomeris marginata). Oecologia 107:131–140
Margalef DR (1958) Information theory in ecology. Gen Syst 3:36–71
Moore JC, Berlow EL, Coleman DC, Ruiter PCD, Dong Q, Alan H, Collins JN, Mccann KS, Kim M, Morin PJ (2004) Detritus, trophic dynamics and biodiversity. Ecol Lett 7:584–600
Pielou EC (1967) The measurement of diversity in different types of biological collection. J Theor Biol 15:131–144
Read DJ, Perez-Moreno J (2003) Mycorrhizas and nutrient cycling in ecosystems—A journey towards relevance? New Phytol 157:475–492
Ruan H, Li Y, Zou X (2005) Soil communities and plant litter decomposition as influenced by forest debris: variation across tropical riparian and upland sites. Pedobiologia 49:529–538
Seastedt TR (1984) The role of microarthropods in decomposition and mineralization processes. Annu Rev Entomol 29:25–46
Shannon CE (1948) A mathematical theory of communication. Bell Syst Tech J 27:379–423
Simpson EH (1949) Measurement of diversity. Nature 163:688
Swift MJ, Heal OW, Anderson JM (1979) Decomposition in terrestial ecosystems. Decompos Terr Ecosyst 5:607–612
Wall DH, Bradford MA, John MGS, Trofymow JA, Behan-Pelletier V, Bignell DE, Dangerfield JM, Parton WJ, Rusek J, Voigt W (2008) Global decomposition experiment shows soil animal impacts on decomposition are climate-dependent. Glob Change Biol 14:2661–2677
Wang G, Wang Y, Han L, Zhang M, Li B (2005) Soil animal communities of variously utilized in the Dongting Lake region. Acta Ecol Sin 25:2629–2636
Wang S, Ruan H, Wang B (2009) Effects of soil microarthropods on plant litter decomposition across an elevation gradient in the Wuyi Mountains. Soil Biol Biochem 41:891–897
Wang S, Ruan H, Han Y (2010) Effects of microclimate, litter type, and mesh size on leaf litter decomposition along an elevation gradient in the Wuyi Mountains, China. Ecol Res 25:1113–1120
Wang X, Yin X, Song B, Xin W, Li B, Ma H (2011) Main species litter decomposition and function of soil fauna in Leymus chinensis grassland. Acta Prataculturae Sin 20:143–149
Wang W, Yang W, Tan B, Liu R, Wu F (2015) Effects of soil fauna to nitrogen and phosphorus releases during litter decomposition at different phenological stages in the subtropical evergreen broad-leaved forest in Sichuan Basin. Scientia Silvae Sinicae 51:1–11
Wei X, Hao M, Shao M, Gale WJ (2006) Changes in soil properties and the availability of soil micronutrients after 18 years of cropping and fertilization. Soil Tillage Res 91:120–130
Wood CT, Schlindwein CCD, Soares GLG, Araujo PB (2012) Feeding rates of Balloniscus sellowii (Crustacea, Isopoda, Oniscidea): the effect of leaf litter decomposition and its relation to the phenolic and flavonoid content. Zookeys 24:231–245
Wu T (2013) Effects of global change on soil fauna diversity: a review. Chin J Appl Ecol 24:581–588
Wu D, Zhang B, Chen P (2006) Community structure and composition of soil macrofauna under different land use in Changchun City. Acta Zool Sin 52:279–287
Wu Y, Cai Q, Lin C, Huang J, Cheng X (2009) Effects of terrace hedgerows on soil macrofauna diversity. Acta Ecol Sin 29:5320–5329
Wu F, Yang W, Zhang J, Deng R (2010) Litter decomposition in two subalpine forests during the freeze–thaw season. Acta Oecol 36:135–140
Xia L, Wu F, Yang W, Tan B (2012) Contribution of soil fauna to the mass loss of Betula albosinensis leaf litter at early decomposition stage of subalpine forest litter in western Sichuan. Chin J Appl Ecol 23:301–306
Xu G, Kuster TM, Günthardt-Goerg MS, Matthias D, Li M (2012) Seasonal exposure to drought and air warming affects soil Collembola and mites. PLoS ONE 7:1–9
Xuluc-Tolosa FJ, Vester HFM, Ramĺrez-Marcial N, Castellanos-Albores J, Lawrence D (2003) Leaf litter decomposition of tree species in three successional phases of tropical dry secondary forest in Campeche, Mexico. For Ecol Manag 174:401–412
Yang X, Zou X (2006) Soil Fauna and leaf litter decomposition in tropical rain forest in XiShuangBanNa, SW China: effects of Mesh Size of Litterbags. J Plant Ecol 30:791–801
Yang X, Chen J (2009) Plant litter quality influences the contribution of soil fauna to litter decomposition in humid tropical forests, southwestern China. Soil Biol Biochem 41:910–918
Yin W, Hu S, Shen Y (1998) Pictorial keys to soil animals of China. Science Press, Beijing
Yin X, Zhong W, Wang H, Chen P (2002) Decomposition of forest defoliation and role of soil animals in Xiao Hinggan Mountains. Geogr Res 21:689–699
Yin X, Song B, Dong W, Xin W, Wang Y (2010) A review on the eco-geography of soil fauna in China. J Geogr Sci 20:333–346
Yu Q, Wu J, Liang D, Zhang J, Li Z, Zhang S (2015) Effects of flooding condition and mesh size on leaf litter decomposition of the dominant species, Carex atrofusca, in an alpine swamp meadow in Tibetan Plateau. Chin J Ecol 34:2785–2791
Zhang J, Qin Z, Li Q (2011) Clustering and ordination of soil animal community under different land-use types. Chin J Ecol 30:2849–2856
Acknowledgements
We thank Guobing Wang, Yueqin Chen, Yuanyuan Li and Danyan Zhou for their assistance. Special thanks go to the Dongtai Forest Farm for its support in the field trials.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project funding: This research was supported by the National Basic Research Program of China (973 Program, 2012CB416904) and partially supported by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
The online version is available at http://www.springerlink.com
Corresponding editor: Chai Ruihai.
Rights and permissions
About this article
Cite this article
Yang, B., Zhang, W., Xu, H. et al. Effects of soil fauna on leaf litter decomposition under different land uses in eastern coast of China. J. For. Res. 29, 973–982 (2018). https://doi.org/10.1007/s11676-017-0521-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-017-0521-5