Abstract
Changes in land use cover, particularly from forest to agriculture, is a major contributing factor in increasing carbon dioxide (CO2) level in the atmosphere. Using satellite images of 1999 and 2011, land use and land use changes in the Kumrat valley KPK, Pakistan, were determined: a net decrease of 11.56 and 7.46 % occurred in forest and rangeland, while 100 % increase occurred in agriculture land (AL). Biomass in different land uses, forest land (FL), AL, and range land (RL) was determined by field inventory. From the biomass data, the amount of carbon was calculated, considering 50 % of the biomass as carbon. Soil carbon was also determined to a depth of 0–15 and 16–30 cm. The average carbon stocks (C stocks) in all land uses ranged from 28.62 ± 13.8 t ha−1 in AL to 486.6 ± 32.4 t ha−1 in pure Cedrus deodara forest. The results of the study confirmed that forest soil and vegetation stored the maximum amount of carbon followed by RL. Conversion of FL and RL to AL not only leads to total loss of about 56 % (from FL conversion) and 37 % (RL conversion) of soil carbon in the last decades but also the loss of a valuable carbon sink. In order to meet the emissions reduction obligations of the Kyoto Protocol, Conservation of forest and RL in the mountainous regions of the Hindu Kush will help Pakistan to meet its emissions reduction goals under the Kyoto Protocol.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Land-use, land-use change and forestry (LULUCF) contribute to ongoing anthropogenic climate change (Houghton 2003), and have consequently received increasing research attention over the last decade (Rokityanskiy et al. 2007; Running 2008; Smith 2008; Strassmann et al. 2008; Zomer et al. 2008). LULUCF is one of the five sources of greenhouse gases (GHGs) included in the United Nations Framework Convention on Climate Change (UNFCC) (UNFCCC 1992), and impact global GHGs emissions, biodiversity and land quality (Cowie et al. 2007). However, LULUCF can also make a significant contribution to the reduction of GHGs, by increasing the carbon storage of terrestrial ecosystems (carbon sequestration), by conserving existing C stocks (e.g. by avoiding deforestation or land degradation), and by providing renewable energy (biomass production) (Andersson et al. 2009). Such LULUCF activities are expected to provide a significant and cost-effective way by which atmospheric CO2 concentration can be reduced, at least in the short- to medium-term (Nabuurs et al. 2007). They can also assist countries in meeting part of their emissions reduction targets that are being proposed for the years after the Kyoto Protocol’s first compliance period (2008–2012) (Gainza-Carmenates et al. 2010). Furthermore, mitigation-driven LULUCF activities could have positive effects on the provision of other ecosystem services (Schroter et al. 2005).
The effects of mitigation-driven LULUCF activities are expected to be regionally unique, as changes in C stocks depend on many regional factors including suitability for different land uses, and the effectiveness of policy for carbon sequestration (Nabuurs et al. 2007). Detailed regional-level analyses are therefore needed to provide accurate estimates of LULUCF offset potentials, in order to help achieve the GHGs target reduction under a post-2012 climate agreement. Nevertheless, relatively few spatially explicit studies have been undertaken to date on the potential effects of land-use change on C stocks (Bolliger et al. 2008; Tappeiner et al. 2008).
The forests of Pakistan, especially the northern mountain forests, are rich in biodiversity and considered integral to the national economy. However they are under extreme threats from deforestation (Tanvir et al. 2007). The present study was carried out in the Kumrat valley of the Hindu Kush region. The aim of the study was to quantify C stock in different land uses and to find out the effect of land use conversion on C stocks. The objectives of the study were to estimate above-ground and below-ground biomass in different land uses and to estimate C stock in these land uses.
Materials and methods
Study area
The study was conducted in Kumrat valley Dir (U), Khyber Pukhtoonkhwa Khwa (KPK), which is located on the northwest side of KPK and to the north of Dir proper (Dir upper). The latitude and longitude of study area is 35°32′11.44″N 72°13′45.01″E. The elevation of the area ranges from 2,439 to 3,048 m. Average precipitation ranges from 1,200 to 600 mm. Major types of rocks in study area are granite, diorite, norites and schist. The soil is mostly loam or sandy loam.
Land use and land use cover change assessment
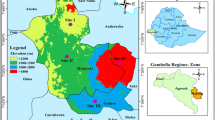
To assess land use and land use cover change, temporal images from 1999 and 2011 were downloaded from the website of US Geological Survey department http://glovis.usgs.gov/. These images were of 30 m spatial resolution. The images were put in ERDAS Imagine software and the area of interest was clipped from a complete scene. Image enhancement techniques, such as NDVI (Normalized Difference Vegetation Index), was applied to enhance the vegetation. Then signatures for different classes, including forests, agricultural lands, range lands (RLs), barren lands, and water bodies were taken in the software ERDAS. Using these signatures, the entire study area was organized into defined classes and then the classified images were imported to ArcGIS. The area for each class of each year was calculated in ArcGIS and two maps (1999 and 2011) were prepared (Fig. 1). In addition, forest maps, topographic sheets, and the 1995 working plan of the Upper Dir KPK were also used for land-use assessment.
Field enumeration in each land use
Overall 60 plots (10 plots each in RL and agricultural land, while 40 plots in forest land (FL) use; Fig. 1) were laid out according to terrain, time, and budget constraint. The size of each plot was 1 ha in FL, while in agriculture and RL, the size of each plot was 0.1 ha. Plots were randomly located in study area (Fig. 1). In each plot of FL, tree height (m), tree diameter (cm) at breast height (dbh) was measured. Tree height (m) was measured by Abney’s level. Tree volume (m3 ha−1) of all sampling sites in each forest stand was calculated as:
where SV is stem volume (m3); h is the height of the tree in meters; d 2 is the square of bdh and f is form factor. The calculated volume (m3 ha−1) in each stand was multiplied with the basic wood density (kg m−3) of respective tree species to calculate total stem biomass. Wood density of the respective tree species was sourced from the available literature (Haripriya 2000).
where SB is the stem biomass (kg), SV is the stem volume, and WD is the wood density
Biomass expension factors (BEF) of the respective species were sourced from available literature (Haripriya 2000). The BEF was used to calculate the total biomass of an entire forest. In order to calculate the biomass of under-story vegetation (Usv), rangeland, and AL, 10 sub plots of 1 m2 were laid out in each sample plot, then the vegetation was harvested and put in bags and their fresh weight was determined. The samples were dried in an oven at 72 °C for 48 h to obtain their dry weight.
Calculation of C stocks in each land use
The total C stock in each land use was calculated from total biomass. The total biomass of each land use was multiplied with conversion factor of 0.5 that has been used globally (Roy et al. 2001; Brown and lugo 1982; Malhi et al. 2004; Nizami 2012).
Soil sampling and analysis
To calculate soil carbon, soil samples were taken from all sample plots of the respective land uses. Three soil samples from each plot, at depths of 0–15 and 16–30 cm, were taken by auger yielding soil cores of known volume 198.29 cm3 (height = 7.25 cm and diameter = 5.9 cm). The weight of each sample was determined and the soil bulk density was calculated by dividing the weight of soil (gm) with the volume (cm3) of the core. Soil carbon was calculated, using the Walkley and Black method (1934). Total soil in t ha−1 was calculated by using the following formula (Nizami 2012).
where SC means Soil carbon (t ha−1), SOC-soil organic carbon (t ha−1), SBD-Soil Bulk density (gm cm−3), TH-Thickness of horizon (cm).
Results
Land use and land use cover change
Based on satellite images from 1999 and 2011, land use and land use cover changes were determined, including the total area of each land use (Table 1). In 2011, four overall land uses—water bodies, FL, barren lands, and RLs—were identified in addition to one new land use, agriculture. Based on ground verification and surveys, FL was further classified into pure Pinus wallichiana forest (PPWF), pure Cedrus deodara forest (PCDF), pure Abies pindrow forest (PAPF), and mixed conifer forest (MCF).
Stem density, stem volume, stem biomass, and total tree biomass in FL
In each forest, the stem density (No. of trees ha−1), stem volume (m3 ha−1 ), upper-story vegetation biomass (USVB), and under-story vegetation biomass (uSVB; t ha−1) was calculated (Table 2). Stem density was maximum in MCF and minimum in PCDF. Stem density decreased with increases in diameter (Fig. 2). The maximum basal area (m2 ha−1) and volume (m3 ha−1) was recorded in PCDF, followed by PAPF. The minimum basal area (m2 ha−1) and volume (m3 ha−1) was recorded in PPWF. The average stem biomass in the forest stand ranges from 304.2 ± 37.7 to 548.33 ± 36 (t ha−1). The highest stem biomass was recorded in PCDF and lowest in PPWF.
In the present study, two forests (PCDF and PAPF) consist of old-age trees with large diameters, which resulted in more basal area, stem volume, and stem biomass as compared to PPWF and MCF. Stem volume and stem biomass has a direct relationship with basal area. In each forest stand, the relation of stem volume and biomass with basal area is given in Figs. 3, 4). In PPWF and MCF, there were fewer old-age trees. Total tree biomass in each forest was calculated using the allomatric equations. The value of above and below-ground biomass was calculated as 459.2.9 ± 56.9 t ha−1 in PPWF to 827.9 ± 55.4 t ha−1 in PCDF.
Understory vegetation (uSV) in each forest stand mainly consisted of various grasses, forbs and shrubs. Among grasses Cynodon dacttylon, Agropyron dentatum, A. canaliculatum and Poa species dominates while major forbs are Caltha alba, Bergenia ciliate, Rumux dentatus, Pomi emodia, and Plantago major. In PPWF, the dominant uSV were grasses with an average understorey biomass equals to 1.69 t ha−1. The mean biomass of uSV in PCDF was 1.1 t ha−1. The mean uSV biomass in MCF and PAPF was 1.6 and 2.6 t ha−1. In PPWF and MCF, the USV mainly consisted of grasses with associated woody shrubs and forbs, which resulted more biomass as compared to PCDF and PAPF.
Total biomass in agricultural land (AL) and rangeland (RL)
The mean biomass of AL was 2.91 ± 0.870 t ha−1. The mean biomass of RL was 5.86 ± 2.788 t ha−1. In RL, grasses were the dominant vegetation. Species like Artemisia spp, Indigofera wallichiana, Rosa webbiana and Berberis lyceum were also found in RL.
Calculation of C stocks in each land use
C stock in each land use was calculated from the total biomass. Details of C stocks in each land use are given in Table 3. The study revealed that among the different land uses FL holds maximum C stocks followed by RL. The forests of the study sites consisted of mature trees. The value of % CV in case of PCDF and PAPF yielded little variation as compared to PPWF and MCF. In PCDF and PAPF, the uniform nature (same age and diameter) of vegetation resulted in little variation. In RL, the sample plots where the vegetation was mixed (grasses, forbs and woody spp) had more biomass and higher value of C stocks. The sites where dominant vegetation was either grasses or forbs contained a minimum value of carbon. Similarly in AL in some sites, the contribution of weed biomass was greater, which gave higher values of biomass and carbon, while on other sites, the weed contribution in biomass was less, therefore responsible for minimum value of biomass and carbon.
Soil carbon calculation in all land uses
The mean soil carbon in PPWF, PAPF, PCDF, MCF, AL and RL was 63.3, 72.12, 75.03, 71.4 and 27, 47.1 t ha−1, respectively. Among all land uses, FL holds the maximum soil carbon (70.78 t ha−1), followed by RL (47.05 t ha−1) while the AL hold minimum soil carbon of 28.62 t ha−1. FL, particularly PPWF and RL, were converted to AL in last 10 years. It can be concluded from the present study that forest and RL conversion to AL resulted in a loss of 43.78 t ha−1 at the rate of 3.46 t ha−1 a−1 in the forest and 20.05 t ha−1 (at the rate of 1.673.46 t ha−1 a−1) in rangeland from 1999 to 2011.
Total C stocks in all land uses
The total C stocks in each land use was determined from respective components of carbon. The components of carbon in each land use were the total biomass of vegetation and soil carbon (Table 3). Among the land uses, FL hold the maximum amount of carbon of 349.84 ± 30.79 t ha−1, followed by RL (50 ± 6.5 t ha−1). Minimum carbon stocks among all land was recorded in AL (28.62 ± 13.85 t ha−1). It can be concluded from the results of present study that forest soil and vegetation stored the highest level of carbon as compared to other land uses. Deforestation in the study area not only caused an addition release of carbon to the atmosphere but also destruction of valuable sink of carbon.
Discussion
The value of total biomass in the present study ranged from 437.76 ± 76 t ha−1 MCF to 809.91 ± 76.03 t ha−1 PCDF. The highest level of biomass was recorded in PCDF followed by PAPF. These biomass values are comparable with those in Gupta and Sharma (2011) in India. The forest stand was comprised of old age trees with high tree density (fully stocked), which resulted in more biomass and C stocks. PCDF and PAPF stored more carbon compared to other two forests (PPWF and MCf) that contained 405.77 ± 38.24 and 266.50 ± 19. t ha−1, respectively. Similar results in Cedrus deodara and Abies pindrow forest have been reported by Gupta and Sharma (2011). Old growth forest had more carbon stocks because of more tree layer biomass, which is a time-dependent accretion of carbon (Zang et al. 2012).
Soil carbon is an integral part of a particular ecosystem. Soil carbon ranged from 35.033 t ha−1(AL) to 70.78 t ha−1 (FL). The present study revealed that among different land uses, FL had more soil carbon followed by RL. The results of the present study also showed that in all four forest stands, PAPF held the maximum soil carbon (75.02 t ha−1). Gupta and Sharma (2011) reported soil carbon of 132 ± 22.73, 120.35 ± 25.86, and 85.67 ± 30.20 t ha−1 in the Abies pindrow and Picia smithina mixed forest, Cedrus deodara and Pinus wallichiana forests of India respectively. In FL, they estimated an average soil carbon as 78.49 t ha−1, while in horticulture and grass land, they estimated an average soil carbon of 45.13 ± 27.25 and 75.76 ± 44.00 t ha−1, respectively. The present study also showed the same pattern of higher soil carbon in respective forest stands and other land uses. The conversion of forest and RL to agricultural land can be linked to the rapid increase in population and migration of people to upland area. The present study revealed that the area was under high pressure from local people regarding forest and RL degradation and conversion to AL. The results indicated that FL stored the highest level of carbon followed by RL. These differences in total carbon in each land use confirmed that conversion of FL and RL to AL caused enormous carbon losses. Across the land uses, total mean C stocks range from 28.62 ± 13 t ha−1 in AL to 477.82 ± 39.5 t ha−1 in PCDF. The differences of C stocks in different land uses are consistent with Sharma and Rai (2007).
Land use change, particularly from forest to AL, is the most significant factor in terms of change in C stocks around the globe (Li et al. 2008). Forests have the ability to store 20 to 100 times more carbon ha−1 than cropland and upon conversion of forest to agricultural land (Houghton 2003). The same results were recorded in the present study where the PCDF contain about 20 times more carbon as compare to AL.
Zang et al. (2012) pointed out that soil disturbance due to site preparation and tree planting reduces soil carbon. Sharma and Rai (2007) reported that soil carbon loss is greater (92 %) in cropped areas as compared to forests due to disturbances. The grass land conversion to cropland decreases carbon content in soil (Yan et al. 2009). Conversion of forest into cropland reduced the amount of soil carbon by 32 % over 15 years (Fantaw et al. 2007). Similar results were found in the present study. Conversion of FL resulted in a loss of 56 % of soil carbon at a depth of 30 cm over the last decade. The conversion of RL to AL resulted loss of 37 % of soil carbon at a depth of 30 cm. Agricultural practices like cultivation, removal of vegetation cover, and exposure of soil to erosion caused a significant reduction in soil carbon.
The results of the present study corroborated that forest soil and vegetation sustained more carbon than AL. Similarly, RL soil and vegetation hold more carbon than AL, soil and vegetation. The results of the study confirmed that conversion of FL and RL to AL caused loss of 29.15 and 4.16 t ha−1 a−1 carbon from 1999 to 2011 respectively. Afforestation, reforestation, and rehabilitation of degraded forest and RL can play in important role in mitigation of atmospheric carbon dioxide. The area has great potential in term of carbon trading under CDM of Article 12 of Kyoto protocol. Proper management of land, control of deforestation, reforestation of degraded land, and rehabilitation of degraded RL can be significant steps for carbon sequestration in the study area and inclusion in carbon trading.
References
Andersson K, Evans TP, Richards KR (2009) National forest carbon inventories: policy needs and assessment capacity. Clim Change 93:69–101
Bolliger J, Hagedorn F, Leifeld J, Böhl J, Zimmermann S, Soliva R, Kienast F (2008) Effects of land-use change on carbon stocks in Switzerland. Ecosystems 11:895–907
Brown S, Lugo AE (1982) The storage and production of organic matter in tropical forest and their role in global carbon cycle. Biotropica 14:161–187
Cowie A, Schneider UA, Montanarella L (2007) Potential synergies between existing multilateral environmental agreements in the implementation of land use, land-use change and forestry activities. Environ Sci Policy 10:335–352
Fantaw Y, Stig L, Abdu A (2007) Changes in soil organic carbon and total nitrogen contents in three forest covers and land use types in the Bale Mountains, south-eastern highlands of Ethiopia. For Ecol Manage 242:337–342
Gainza-Carmenates R, Carlos Altamirano-Cabrera J, Thalmann P, Drouet L (2010) Trade-offs and performances of a range of alternative global climate architectures for post-2012. Environ Sci Policy 13:63–71
Gupta MK, Sharma SD (2011) Sequestrated carbon: organic carbon pool in the soils under different land uses in Garhwal Himalayan Region of India. Int J Agricu For 1(1):14–20
Haripriya GS (2000) Estimates of biomass in Indian forests. Biomass Bioenerg 19:245–258
Houghton RA (2003) Revised estimates of the annual net flux of carbon to the atmosphere from changes in land use and land management 1850–2000. Tellus, Ser B 55:378–390
Li H, Ma Y, Aide TM, Liu W (2008) Past, present and future land-use in Xishuangbanna, China and the implications for carbon dynamics. For Ecol Manage 255:16–24
Malhi Y, Baker TR, Phillips OL, Almeida S, Alvarez E, Arroyo L, Chave J, CzimczikI CI, Fiore AD, Higuchi N, Killeen TJ, Laurance SG, Laurance WF, Lewis SL, Montoya LMM, Lloyd J (2004) The above ground coarse wood productivity of 104 Neotropical forest plots. Glob Change Biol 10:563–591
Nabuurs GJ, Masera O, Andrasko K, Benitez-Ponce P, Boer R, Dutschke M, Elsiddig E, Ford-Robertson J, Frumhoff P, Karjalainen T, Krankina O, Kurz W, Matsumoto M, Oyhantcabal W, Ravindranath NH, Sanz Sanchez MJ, Zhang X (2007) Forestry. In: Metz B, Davidson OR, Bosch PR, Dave R, Meyer LA (eds) Climate change 2007: mitigation. Contribution of working group III to the fourth assessment report of the intergovernmental panel on climate change, Cambridge University Press, Cambridge, UK
Nizami SM (2012) The inventory of the carbon stocks in sub-tropical forest of Pakistan for reporting under Kyoto Protocol. J For Res 23(2):377–384
Philip MS (1994) Measuring trees and forests, 2nd edn. CAB. International, Wallingford
Rokityanskiy D, Benitez PC, Kraxner F, McCallum I, Obersteiner M, Rametsteiner E, Yamagata Y (2007) Geographically explicit global modeling of land-use change, carbon sequestration, and biomass supply. Technol Forecast Soc Chang 74:1057–1082
Roy J, Saugier B, Mooney H (2001) Terrestrial global productivity: past. Academic Press, San Diego
Running SW (2008) Climate change: ecosystem disturbance, carbon, and climate. Science 321:652–653
Schroter D, Cramer W, Leemans R, Prentice IC, Araujo MB, Arnell NW, Bondeau A, Bugmann H, Carter TR, Gracia CA, de la Vega-Leinert AC, Erhard M, Ewert F, Glendining M, House JI, Kankaanpaa S, Klein RJT, Lavorel S, Lindner M, Metzger MJ, Meyer J, Mitchell TD, Reginster I, Rounsevell M, Sabate S, Sitch S, Smith B, Smith J, Smith P, Sykes MT, Thonicke K, Thuiller W, Tuck G, Zaehle S, Zierl B (2005) Ecosystem service supply and vulnerability to global change in Europe. Science 310:1333–1337
Sharma S, Rai SC (2007) Carbon sequestration with land-use cover change in a Himalayan watershed. Geoderma 139:371–378
Smith P (2008) Land use change and soil organic carbon dynamics. Nutr Cycl Agroecosyst 81:169–178
Strassmann KM, Joos F, Fischer G (2008) Simulating effects of land use changes on carbon fluxes: past contributions to atmospheric CO2 increases and future commitments due to losses of terrestrial sink capacity. Tellus, Ser B 60:583–603
Tanvir A, Ahmad M, Shahbaz B, Suleri A (2007) Impact of participatory forest management on vulnerability and livelihood assets of forest-dependent communities in northern Pakistan. Int J Sustain Dev World Ecol 14(2):211–223
Tappeiner U, Tasser E, Leitinger G, Cernusca A, Tappeiner G (2008) Effects of historical and likely future scenarios of land use on above-and belowground vegetation carbon stocks of an Alpine valley. Ecosystems 11:1383–1400
UNFCCC 1992 united nations framework convention on climate change, article 4. Available at http://unfccc.int/resource/docs/convkp/conveng.pdf. Accessed 10 March 2013
Walkley A, Black JA (1934) An examination of the degtjaref method for determining soil organic matter and proposed modification of the chromic. titration method. Soil Sci 37:29–38
Yan J, Zhu X, Jiaohong Z (2009) Effects of grassland conversion to cropland and forest on soil organic carbon and dissolved organic carbon in farming pastoral ecotone of Inner Mongolia. Acta Ecol Sin 29:150–154
Zang Y, Fengxue G, Shirong L, Yanchun L, Chao L (2012) Variation of carbon stock with forest types subalpine region of South Western China. For Ecol Manage 30:88–95
Zomer RJ, Trabucco A, Bossio DA, Verchot LV (2008) Climate change mitigation: a spatial analysis of global land suitability for clean development mechanism afforestation and reforestation. Agric Ecosyst Environ 126:67–80
Author information
Authors and Affiliations
Corresponding author
Additional information
The online version is available at http://www.springerlink.com
Corresponding editor: Hu Yanbo
Rights and permissions
About this article
Cite this article
Ahmad, A., Nizami, S.M. Carbon stocks of different land uses in the Kumrat valley, Hindu Kush Region of Pakistan. J. For. Res. 26, 57–64 (2015). https://doi.org/10.1007/s11676-014-0008-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-014-0008-6