Abstract
Objective
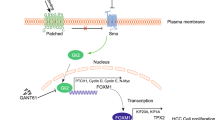
The activation of hedgehog (HH) pathway is implicated in the development of human malignancies including hepatocellular carcinoma (HCC). However, the clinical impact of HH activation in HCC patients is still unclear. This study was conducted to confirm whether the expression of HH pathway components was associated with HCC progression and clinical outcome.
Methods
This study was a sample-expanded and prolonged follow up of one of our previous studies. It included 46 HCC patients who underwent surgical treatment from 2002 to 2005. The expression of sonic HH (SHH), patched-1 (PTCH1), smoothened (SMOH) and glioma-associated oncogene-1 (GLI1) genes in tumor and adjacent normal tissues extracted from the patients were examined by reverse transcriptionpolymerase chain reaction (RT-PCR) to explore the relationship between these genes and the clinical prognosis of HCC.
Results
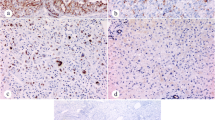
The expression levels of SHH, PTCH1, SMOH and GLI1 in HCC tissues were 60.87%, 50.00%, 32.61% and 54.35%, respectively. The expression levels of SHH-related molecules were relatively intense in cancer tissue, but insignificantly correlated with any clinicopathological factors of tumor. Transcriptional factor GLI1 was the only molecule associated with poor prognosis among the HCC patients. The expression of GLI1 gene in tumor tissues was significantly related with disease-free survival (DFS) (P=0.042) and overall survival (OS) (P=0.030). The simultaneous expression of GLI1 in tumor and adjacent normal liver tissues correlated with DFS (P<0.029) and OS (P<0.025).
Conclusions
HH signaling activation is an important event in the development of human HCC. The expression of GLI1 in SHH pathway is possibly involved in HCC progression, which may be a useful prognostic indicator of HCC.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Sicklick JK, Li YX, Jayaraman A, et al. Dysregulation of the hedgehog pathway in human hepatocarcinogenesis. Carcinogenesis 2006;27:748–757.
Patil MA, Zhang J, Ho C, et al. Hedgehog signaling in human hepatocellular carcinoma. Cancer Biol Ther 2006;5:111–117.
Huang S, He J, Zhang X, et al. Activation of the hedgehog pathway in human hepatocellular carcinomas. Carcinogenesis 2006;27:1334–1340.
Beachy PA, Karhadkar SS, Berman DM. Tissue repair and stem cell renewal in carcinogenesis. Nature 2004;432:324–331.
Nüsslein-Volhard C, Wieschaus E. Mutations affecting segment number and polarity in Drosophila. Nature 1980;287:795–801.
Johnson RL, Rothman AL, Xie J, et al. Human homolog of patched, a candidate gene for the basal cell nevus syndrome. Science 1996;272:1668–1671.
Hahn H, Wicking C, Zaphiropoulous PG, et al. Mutations of the human homolog of Drosophila patched in the nevoid basal cell carcinoma syndrome. Cell 1996;85:841–851.
Reifenberger J, Wolter M, Weber RG, et al. Missense mutations in SMOH in sporadic basal cell carcinomas of the skin and primitive euroectodermal tumors of the central nervous system. Cancer Res 1998;58:1798–1803.
Xie J, Murone M, Luoh SM, et al. Activating smoothened mutations in sporadic basal-cell carcinoma. Nature 1998;391:90–92.
Karhadkar SS, Bova GS, Abdallah N, et al. Hedgehog signalling in prostate regeneration, neoplasia and metastasis. Nature 2004;431:707–712.
Sanchez P, Clement V, Ruiz i Altaba A. Therapeutic targeting of the hedgehog- GLI pathway in prostate cancer. Cancer Res 2005;65:2990–2992.
Watkins DN, Berman DM, Burkholder SG, et al. Hedgehog signalling within airway epithelial progenitors and in small-cell lung cancer. Nature 2003;422:313–317.
Velcheti V, Govindan R. Hedgehog signaling pathway and lung cancer. J Thorac Oncol 2007;2:7–10.
Berman DM, Karhadkar SS, Maitra A, et al. Widespread requirement for Hedgehog ligand stimulation in growth of digestive tract tumors. Nature 2003;425:846–851.
Monzo M, Moreno I, Artells R, et al. Sonic hedgehog mRNA expression by real-time quantitative PCR in normal and tumor tissues from colorectal cancer patients. Cancer Lett 2006;233:117–123.
Dimmler A, Brabletz T, Hlubek F, et al. Transcription of sonic hedgehog, a potential factor for gastric morphogenesis and gastric mucosa maintenance, is upregulated in acidic conditions. Lab Invest 2003;83:1829–1837.
Isohata N, Aoyagi K, Mabuchi T, et al. Hedgehog and epithelial-mesenchymal transition signaling in normal and malignant epithelial cells of the esophagus. Int J Cancer 2009;125:1212–1221.
Ma X, Chen K, Huang S, et al. Frequent activation of the hedgehog pathway in advanced gastric adenocarcinomas. Carcinogenesis 2005;26:1698–705.
Feldmann G, Dhara S, Fendrich V, et al. Blockade of hedgehog signaling inhibits pancreatic cancer invasion and metastases: a new paradigm for combination therapy in solid cancers. Cancer Res 2007;67:2187–2196.
Kubo M, Nakamura M, Tasaki A, et al. Hedgehog signaling pathway is a new therapeutic target for patients with breast cancer. Cancer Res 2004;64:6071–6074.
Mukherjee S, Frolova N, Sadlonova A, et al. Hedgehog signaling and response to Cyclopamine differ in epithelialand stromal cells in benign breast and breast cancer. Cancer Biol Ther 2006;5:674–683.
Chen X, Horiuchi A, Kikuchi N, et al. Hedgehog signal pathway is activated in ovarian carcinomas, correlating with cell proliferation: It’s inhibition leads to growth suppression and apoptosis. Cancer Science 2007;98:68–76.
Deutsch G, Jung J, Zhang M, et al. A bipotential precursor population for pancreas and liver within the embryonic endoderm. Development 2001;128:871–881.
ten Haaf A, Bektas N, von Serenyi S, et al. Expression of the glioma-associated oncogene homolog (GLI) 1 in human breast cancer is associated with unfavourable overall survival. BMC Cancer 2009;9:298.
Li YC, Deng YH, Guo ZH, et al. Prognostic value of hedgehog signal component expressions in hepatoblastoma patients. Eur J Med Res 2010;15:468–474.
Souzaki M, Kubo M, Kai M, et al. Hedgehog signaling pathway mediates the progression of non-invasive breast cancer to invasive breast cancer. Cancer Sci 2011;102:373–381.
Saze Z, Terashima M, Kogure M, et al. Activation of the sonic hedgehog pathway and its prognostic impact in patients with gastric cancer. Dig Surg 2012;29:115–123.
He HC, Chen JH, Chen XB, et al. Expression of hedgehog pathway components is associated with bladder cancer progression and clinical outcome. Pathol Oncol Res 2012;18:349–355.
Che L, Ren J, Yuan YH, et al. Expression of genes related to Sonic Hedgehog signaling in human hepatocellular carcinomas. Beijing Da Xue Xue Bao (in Chinese) 2008;40:616–623.
Katoh Y, Katoh M. Hedgehog signaling pathway and gastrointestinalstem cell signaling network (Review). Int J Mol Med 2006;18:1019–1023.
Yang L, Xie G, Fan Q, et al. Activation of the hedgehogsignaling pathway in human cancer and the clinical implications. Oncogene 2010;29:469–481.
Hebrok M. Hedgehog signaling in pancreas development. Mech Dev 2003;120:45–57.
Watkins DN, Peacock CD. Hedgehog signalling in foregut malignancy. Biochem Pharmacol 2004;68:1055–1060.
Fattovich G, Giustina G, Degos F, et al. Morbidity and mortality in compensated cirrhosis type C: a retrospective follow-up study of 384 patients. Gastroenterology 1997;112:463–472.
Llovet JM, Burroughs A, Bruix J. Hepatocellular carcinoma. Lancet 2003;362:1907–1917.
Shackel NA, McGuinness PH, Abbott CA, et al. Identification of novel molecules and pathogenic pathways in primary biliary cirrhosis: cDNA array analysis of intrahepatic differential gene expression. Gut 2001;49:565–576.
Shackel NA, McGuinness PH, Abbott CA, et al. Insights into the pathobiology of hepatitis C virus-associated cirrhosis: analysis of intrahepatic differential gene expression. Am J Pathol 2002;160:641–654.
Ramalho-Santos M, Melton DA, McMahon AP. Hedgehog signals regulate multiple aspects of gastrointestinal development. Development 2000;127:2763–2772.
Jung Y, McCall SJ, Li YX, et al. Bile ductules and stromal cells express hedgehog ligands and/or hedgehog target genes in primary biliary cirrhosis. Hepatology 2007;45:1091–1096.
Keeler RF, Binns W. Teratogeneic compaunds of Veratrum californicum (Durand). V. Comparison of cyclopian effects of steroidal alkaloids from the plant and structurally related compounds from other sources. Tetratology 1968;1:5–10.
Incardona JP, Gaffield W, Kapur RP, et al. The teratogenic Veratrum alkaloid cyclopamine inhibits sonic hedgehog signal transduction. Development 1998;125:3553–3562.
Berman DM, Karhadkar SS, Hallahan AR, et al. Medulloblastoma growth inhibition by Hedgehog pathway blockade. Science 2002;297:1559–1561.
Chen JK, Taipale J, Cooper MK, et al. Inhibition of Hedgehog signaling by direct binding of cyclopamine to Smoothened. Genes Dev 2002;16:2743–2748.
Katano M. Hedgehog signaling pathway as a therapeutic target in breast cancer. Cancer Lett 2005;227:99–104.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Che, L., Yuan, YH., Jia, J. et al. Activation of sonic hedgehog signaling pathway is an independent potential prognosis predictor in human hepatocellular carcinoma patients. Chin. J. Cancer Res. 24, 323–331 (2012). https://doi.org/10.1007/s11670-012-0271-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11670-012-0271-z