Abstract
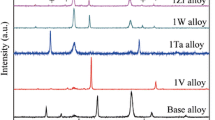
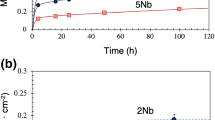
The oxidation of pure Sn and high Pb-Sn alloys was investigated under different oxidizing conditions of temperature and humidity. Both the chemical nature and the amount of oxides were characterized using electrochemical reduction analysis by measuring the electrolytic reduction potential and total transferred electrical charges. For pure tin, SnO grew faster under humid conditions than in dry air. A very thin (<10 Å) layer of SnO2 was formed on the top surface under humid conditions. The mixture of SnO and SnO2 was found for oxidation at 150°C. For oxidation of high Pb-Sn alloys, tin was preferentially oxidized on the surface, and tin content over the solubility limit suppressed the formation of lead oxide. For the evaporated Pb-3Sn alloy reflowed two times at 370°C under H2 atmosphere, tin was enriched on the surface as tin dendrites and lead oxide was formed in the tin-depleted region that surrounds tin dendrites.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
A.T. Fromhold, Jr., Theory of Metal Oxidation, Vol. 1 Fundamentals (New York: North-Holland Publishing Company, 1976), pp. 3–49.
Marcel Pourbaix, Atlas of Electrochemical Equilibria in Aqueous Solutions (New York: Pergamon Press, 1966), pp. 31–50.
Frederick G. Yost, F. Michael Hosking, and Darrel R. Frear, The Mechanics of Solder Alloy, Wetting & Spreading (New York: Van Nostrand Reinhold, 1993), pp. 155–189.
D. Morgan Tench, D.P. Anderson, and P. Kim, J. Appl. Electrochem. 24, 18 (1994).
Rao R. Tummala et al., Fundamentals of Microsystems Packaging (New York: McGraw-Hill, 2001), pp. 672–693.
D.A. Sluzewski, Y.A. Chang, and V.C. Marcotte: Mater. Res. Soc. Symp. Proc. 167, 353 (1990).
David R. Lide, Handbook of Chemistry and Physics, 73rd ed. (Boca Raton, FL: CRC Press, 1993), pp. 5–75.
Frederick G. Yost et al., The Mechanics of Solder Alloy “Wetting & Spreading” (New York: Van Nostrand Reinhold, 1993), pp. 155–189.
C.I. House and G.H. Kelsall, Electrochim. Acta 29, 1459 (1984).
R.J. Bird, J. Met. Sci. 7, 109 (1973).
T. Farrell, Met. Sci. 10, 87 (1976).
R.A. Konetzki and Y.A. Chang, J. Mater. Res. 3, 466 (1988).
A.J. Bevolo, J.D. Verhoeven, and M. Noack, Surf. Sci. 134, 499 (1983).
S.N. Shah and D. Eurof Davies: Electrochim. Acta 8, 663 (1963).
R.L. Opila, J. Vac. Sci. Technol. A4, 173 (1986).
E.E. de Kluizenaar, J. Vac. Sci. Technol. A 1, 1480 (1983).
M.L. Varsányi, J. Jaén, A. Vértes, and L. Kiss, Electrochim. Acta 30, 529 (1985).
D.M. Tench and D.P. Anderson, U.S. patent 5,262,022 (19 Nov. 1993).
S.E.S. El Wakkad, A.M. Shams Dl Din, and Jeannette A. El Sayed, J. Chem. Soc. London, 3103 (1954).
Peter Bratin, Michael Pavlov, and Gene Chalyt, “Surface Evaluation of the Immersion Tin Coatings via Sequential Electrochemical Reduction Analysis (SERA),” Research Report from Homepage of ECI technology (www.ecitechnology.com).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cho, S., Yu, J., Kang, S.K. et al. Oxidation study of pure tin and its alloys via electrochemical reduction analysis. J. Electron. Mater. 34, 635–642 (2005). https://doi.org/10.1007/s11664-005-0077-6
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11664-005-0077-6