Abstract
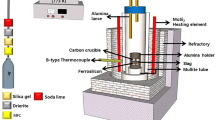
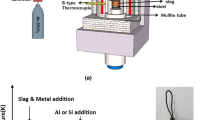
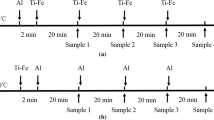
A thermodynamic equilibrium between the Fe-16Cr melts and the CaO-Al2O3-MgO slags at 1823 K as well as the morphology of inclusions was investigated to understand the formation behavior of the MgO-Al2O3 spinel-type inclusions in ferritic stainless steel. The calculated and observed activities of magnesium in Fe-16Cr melts are qualitatively in good agreement with each other, while those of aluminum in steel melts exhibit some discrepancies with scatters. In the composition of molten steel investigated in this study, the log (X MgO/X Al 2O3) of the inclusions linearly increases by increasing the log [a Mg/a 2Al ·a 2O ] with the slope close to unity. In addition, the relationship between the log (X MgO/X Al 2O3) of the inclusions and the log (a MgO/a Al 2O3) of the slags exhibits the linear correlation with the slope close to unity. The compositions of the inclusions are relatively close to those of the slags, viz. the MgO-rich magnesia-spinel solid solutions were formed in the steel melts equilibrated with the highly basic slags saturated by CaO or MgO. The spinel inclusions nearly saturated by MgO were observed in the steel melts equilibrated with the slags doubly saturated by MgO and MgAl2O4. The spinel and the Al2O3-rich alumina-spinel solid solutions were formed in the steel melts equilibrated with the slags saturated by MgAl2O4 and MgAl2O4-CaAl2O4 phases, respectively. The apparent modification reaction of MgO to the magnesium aluminate inclusions in steel melts equilibrated with the highly basic slags would be constituted by the following reaction steps: (1) diffusion of aluminum from bulk to the metal/MgO interface, (2) oxidation of the aluminum to the Al3+ ions at the metal/intermediate layer interface, (3) diffusion of Al3+ ions and electrons through the intermediate layer, and (4) magnesium aluminate (MgAl2O4 spinel, for example) formation by the ionic reaction.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
B. Harkness and D. Dyson: Proc. METEC Cong. ’94, Düsseldorf, June 20–22, 1994, VDEh, Düsseldorf, 1994, pp. 70–77.
J.W. Kim, S.K. Kim, D.S. Kim, Y.D. Lee, and P.K. Yang: Iron Steel Inst. Jpn. Int., 1996, vol. 36, pp. S140-S143.
Y. Ehara, S. Nakamura, and Y. Habara: Proc. 4th Eur. Stainless Steel Science and Market Cong., Paris, Jun. 10–13, 2002, ATS, Paris, 2002, vol. 2, pp. 176–81.
T. Nishi and K. Shinme: Tetsu-to-Hagané, 1998, vol. 84, pp. 837–43.
G. Okuyama, K. Yamaguchi, S. Takeuchi, and K. Sorimachi: Iron Steel Inst. Jpn. Int., 2000, vol. 40, pp. 121–28.
H. Todoroki and K. Mizuno: Trans. ISS, 2003, Mar., pp. 60–67.
M. Kowalski, P.J. Spencer, and D. Neuschutz: Slag Atlas, 2nd ed., Verlag Stahleisen GmbH, Düsseldorf, 1995.
H. Itoh, M. Hino, and S. Ban-ya: Tetsu-to-Hagané, 1997, vol. 83, pp. 623–28.
H. Itoh, M. Hino, and S. Ban-ya: Tetsu-to-Hagané, 1997, vol. 83, pp. 773–78.
Steelmaking Data Sourcebook, The Japan Society for the Promotion of Science, The 19th Committee of Steelmaking, Gordon and Breach Science Publications, New York, NY, 1988, pp. 273–325.
H. Ohta and H. Suito: Iron Steel Inst. Jpn. Int., 2003, vol. 43, pp. 1301–08.
Z. Hong, X. Wu, and C. Kun: Steel Res., 1995, vol. 66, pp. 72–76.
S.K. Jo, B. Song, and S.H. Kim: Metall. Mater. Trans. B, 2002, vol. 33B, pp. 703–09.
J.D. Seo and S.H. Kim: Steel Res., 2000, vol. 71, pp. 101–06.
T. Itoh, T. Nagasaka, and M. Hino: Iron Steel Inst. Jpn. Int., 2000, vol. 40, pp. 1051–58.
K. Takahashi and M. Hino: High Temp. Mater. Proc., 2000, vol. 19, pp. 1–10.
H. Ohta and H. Suito: Iron Steel Inst. Jpn. Int., 1996, vol. 36, pp. 983–90.
M. Hino, S. Kinoshita, Y. Ehara, H. Itoh, and S. Ban-ya: Proc. 5th. Int. Conf. Molten Slags, Fluxes and Salts ’97, Sydney, Jan. 5–8, 1997, ISS-AIME, Warrendale, PA, 1997, pp. 53–57.
K. Mukai, Z. Li, and M. Zeze: Mater. Trans., 2002, vol. 43, pp. 1724–31.
B.J. Keene: Slag Atlas, 2nd ed., Verlag Stahleisen GmbH, Düsseldorf, 1995, pp. 513–40.
J.H. Park, D.S. Kim, and S.B. Lee: Metall. Mater. Trans. B, 2005, vol. 36B, pp. 67–73.
L. Pauling: The Nature of the Chemical Bond and the Structure of Molecules and Crystals: An Introduction to Modern Structural Chemistry, 3rd ed., Cornell University Press, New York, NY, 1960, pp. 64–107.
J.H. Park and D.J. Min: Metall. Mater. Trans. B, 2001, vol. 32B, pp. 297–303.
J. Livage, F. Babonneau, M. Chatry, and L. Coury: Ceram. Int., 1997, vol. 23, pp. 13–18.
M.K. Naskar and M. Chatterjee: J. Eur. Ceram. Soc., 2004, vol. 24, pp. 3499–3508.
M.H. Zayan, O.M. Jamjoom, and N.A. Razik: Oxid. Met., 1990, vol. 34, pp. 323–33.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Park, J.H., Kim, D.S. Effect of CaO-Al2O3-MgO slags on the formation of MgO-Al2O3 inclusions in ferritic stainless steel. Metall Mater Trans B 36, 495–502 (2005). https://doi.org/10.1007/s11663-005-0041-0
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11663-005-0041-0