Abstract
Objective
Red ginseng (RG, Panax ginseng C.A. Meyer) is one of the widely used herbs for treating type 2 diabetes mellitus (DM). However, no systematic review of the effectiveness of RG for type 2 DM is available. This systematic review aimed to evaluate the current evidence for the effectiveness of RG in patients with type 2 DM.
Methods
Electronic searches of 14 electronic databases were conducted without language restrictions. All randomized clinical trials (RCTs) with RG as a treatment for type 2 DM were considered for inclusion. Their methodological quality was assessed using the Cochrane criteria.
Results
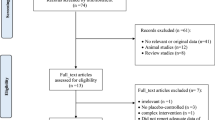
Four RCTs met our inclusion criteria. Their methodological quality was variable. Three of the RCTs compared the effectiveness of RG with placebo. The meta-analysis of these data failed to favor RG over placebo for fasting plasma glucose (FPG) [n =76, weighted mean difference (WMD): −0.43 mmol/L; 95% confidence interval (CI): −1.16 to 0.30, =0.25] and fasting plasma insulin (FPI) (n =76, WMD: −8.43 pmol/L; 95% CI: −19.54 to 2.68, P =0.14) for 12 weeks of treatment. One RCT compared the effects of RG with no treatment. The results did not suggest favorable effects of RG on FPG, hemoglobin A1c (HbA1c) or 2-h blood glucose after a meal (PP2h).
Conclusions
The evidence for the effectiveness of RG in controlling glucose in type 2 DM is not convincing. Few included studies with various treatment regimens prohibit definitive conclusions. More rigorous studies are needed to clarify the effects of RG on this condition.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
World Health Organization. WHO Diabetes. Available from: URL: http://www.who.int/mediacentre/factsheets/fs312/en/.
American Diabetes Association. Living with diabetes. Complications. Available from: URL: http://www.diabetes.org/living-with-diabetes/complications.
Amori RE, Lau J, Pittas AG. Efficacy and safety of incretin therapy in type 2 diabetes: systematic review and metaanalysis. JAMA 2007;298:194–206.
Lee MS, Lim HJ, Moon SR. Survey of the use of complementary and alternative medicine among Korean diabetes mellitus patients. Pharmacoepidemiol Drug Saf 2004;13:167–171.
National Center for Complementary and Alternative Medicine. Diabetes and CAM: A Focus on Dietary Supplements. Available from: URL: http://nccam.nih.gov/health/diabetes/CAM-and-diabetes.htm.
Park YM, Sohn CM, Jang HC. A study on status and subjective recognition of functional foods among diabetic patients. J Korean Diet Assoc 2005;11:216–222.
Kim HY, Kim K. Protective effect of ginseng on cytokineinduced apoptosis in pancreatic beta-cells. J Agric Food Chem 2007;55:2816–2823.
Luo JZ, Luo L. Ginseng on hyperglycemia: effects and mechanisms. Evid Based Complement Alternat Med 2009;6:423–427.
Vuksan V, Sievenpiper JL. Herbal remedies in the management of diabetes: lessons learned from the study of ginseng. Nutr Metab Cardiovasc Dis 2005;15:149–160.
Yun TK. Panax ginseng — a non-organ-specific cancer preventive? Lancet Oncol 2001;2:49–55.
Jang DJ, Lee MS, Shin BC, Lee YC, Ernst E. Red ginseng for treating erectile dysfunction: a systematic review. Br J Clin Pharmacol 2008;66:444–450.
Park KS, Ko SK, Chung SH. Comparisons of antidiabetic effect between Ginseng Radix alba, Ginseng Radix rubra and Panax Quinquefoli radix in MLD STZ-induced diabetic rats. J Ginseng Res 2003;27:56–61.
Buettner C, Yeh GY, Phillips RS, Mittleman MA, Kaptchuk TJ. Systematic review of the effects of ginseng on cardiovascular risk factors. Ann Pharmacother 2006;40:83–95.
Vogler BK, Pittler MH, Ernst E. The efficacy of ginseng. A systematic review of randomised clinical trials. Eur J Clin Pharmacol 1999;55:567–575.
Lee MS, Yang EJ, Kim JI, Ernst E. Ginseng for cognitive function in Alzheimer’s disease: a systematic review. J Alzheimers Dis 2009;18:339–344.
Higgins JPT, Green S, eds. Cochrane handbook for systematic reviews of interventions. Version 5.1.0 [updated March 2011]. The Cochrane Collaboration 2011.
Follmann D, Elliott P, Suh I, Cutler J. Variance imputation for overviews of clinical trials with continuous response. J Clin Epidemiol 1992;45:769–773.
Sievenpiper JL, Sung MK, Di Buono M, Seung-Lee K, Nam KY, Arnason JT, et al. Korean red ginseng rootlets decrease acute postprandial glycemia: results from sequential preparation- and dose-finding studies. J Am Coll Nutr 2006;25:100–107.
Shin SK. Effects of cheonggukjang and red ginseng cheonggukjang in mild hyperglycemic subjects [Dissertation]. Daegu: Kyungpook National University, 2009.
Son BR, Kim YK, Choi DS. The effect of red ginseng on platelet aggregation in noninsulin-dependent diabetes mellitus patients. J Clin Pathol Qual Control 1994;16:89–96.
Yoon SY, Kim YK. The study on the effect of red ginseng on levels of protein C and protein S in non-insulindependent diabetes mellitus patients. Korean J Clin Pathol 1995;15:397–404.
Kim HY. Effects of fermented red ginseng on blood glucose and insulin resistance in patients with type 2 diabetes mellitus [Dissertation]. Pusan: Pusan National University, 2008.
Vuksan V, Sung MK, Sievenpiper JL, Stavro PM, Jenkins AL, Di Buono M, et al. Korean red ginseng (Panax ginseng) improves glucose and insulin regulation in well-controlled, type 2 diabetes: results of a randomized, double-blind, placebo-controlled study of efficacy and safety. Nutr Metab Cardiovasc Dis 2008;18:46–56.
Choi KM, Lee EJ, Kim YH, Baik SH. Effects of red ginseng on the lipid peroxidation of erythrocyte and antioxidant superoxide dismutase (SOD) activity in NIDDM patients. Korean J Ginseng Sci 1997;21:153–159.
Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJ, Gavaghan DJ, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials 1996;17:1–12.
Rattinger G, Bero L. Factors associated with results and conclusions of trials of thiazolidinediones. PLoS One 2009;4:e5826.
Freiman JA, Chalmers TC, Smith HJ, Kuebler RR. The importance of beta, the type II error and sample size in the design and interpretation of the randomized control trial. Survey of 71 “negative” trials. N Engl J Med 1978;299:690–694.
Begg C, Cho M, Eastwood S, Horton R, Moher D, Olkin I, et al. Improving the quality of reporting of randomized controlled trials. The CONSORT statement. JAMA 1996;276:637–639.
Vickers A, Goyal N, Harland R, Rees R. Do certain countries produce only positive results? A systematic review of controlled trials. Control Clin Trials 1998;19:159–166.
Lee HJ, Lee YH, Park SK, Kang ES, Kim HJ, Lee YC, et al. Korean red ginseng (Panax ginseng) improves insulin sensitivity and attenuates the development of diabetes in Otsuka Long-Evans Tokushima fatty rats. Metabolism 2009;58:1170–1177.
Attele AS, Zhou YP, Xie JT, Wu JA, Zhang L, Dey L, et al. Antidiabetic effects of Panax ginseng berry extract and the identification of an effective component. Diabetes 2002;51:1851–1858.
Hong SJ, Fong JC, Hwang JH. Effects of crude drugs on glucose uptake in 3T3-L1 adipocytes. Kaohsiung J Med Sci 2000;16:445–451.
Park MY, Lee KS, Sung MK. Effects of dietary mulberry, Korean red ginseng, and banaba on glucose homeostasis in relation to PAR-a, PPAR-g, and LPL mRNA expressions. Life Sci 2005;77:3344–3354.
Yamasaki K, Murakami C, Ohtani K, Kasai R, Kurokawa T, Ishibashi S. Effects of standardized Panax ginseng extract G115 on the D-glucose transport by Ehrlich ascites tumour cells. Phytother Res 1993;7:200–202.
Hasegawa H, Matsumiya S, Murakami C, Kurokawa T, Kasai R, Ishibashi S, et al. Interactions of ginseng extract, ginseng separated fractions, and some triterpenoid saponins with glucose transporters in sheep erythrocytes. Planta Med 1994;60:153–157.
Suzuki Y, Ito Y, Konno C, Furuya T. Effects of tissue cultured ginseng on gastric secretion and pepsin activity. Yakugaku Zasshi 1991;111:770–774
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, S., Shin, BC., Lee, M.S. et al. Red ginseng for type 2 diabetes mellitus: A systematic review of randomized controlled trials. Chin. J. Integr. Med. 17, 937–944 (2011). https://doi.org/10.1007/s11655-011-0937-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-011-0937-2