Summary
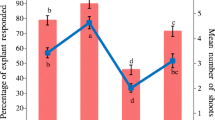
Plants were obtained via organogenesis from hypocotyl explants of Pithecellobium saman (Jacq.) Benth. (raintree) on Murashige and Skoog (1962) medium supplemented with N6-benzyladenine (BA). Adventitious bud induction was affected by mineral salts and benzyladenine concentrations, explant age, position of explant on the medium, and BA exposure time. The best results were obtained with explants cultured on half-strength MS medium containing 26.6 µM BA, with explants of 5 and 10 d after germination placed horizontally and a 7 d-exposure period to BA. The proximal and intermediate section of hypocotyls showed the highest organogenic response. Bud development and shoot elongation was promoted by increased concentrations of activated charcoal in the culture medium. Gibberellic acid had no effect on shoot development. Rooting percentages decreased when shoots were exposed for more than 24 h to indole-3-butyric acid (IBA). Maximum rooting was obtained with 369.0 µM IBA. The aerial and root systems of greenhouse plantlets were similar to those of seedlings.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Castillo de Meier, G. M.; Bovo, O. A. Obtención de plantas a partir del cultivo in vitro de microestacas de brotes epirrizos y brotes de un árbol adulto de Pithecellobium scalare (Tatane) (Leguminosae). Phyton 50:155–163; 1989.
Dumas, E.; Monteuuis, O. In vitro rooting of micropropagated shoots from juvenile and mature Pinus pinaster explants: influence of activated charcoal. Plant Cell Tissue Organ Cult. 40:231–235; 1995.
Fridborg, G.; Pederson, M.; Landstrom, L.-E., et al. The effect of activated charcoal on tissue cultures: adsorption of metabolites inhibiting morphogenesis. Physiol. Plant. 43:104–106; 1978.
George, E. F.; Sherrington, P. D. Plant propagation by tissue culture: handbook and directory of commercial laboratories. London: Exegectics Ltd.; 1984:371–420.
Goldfarb, B.; Strauss, S.; Howe, E., et al. Transient gene expression of microprojectile-introduced DNA in Douglas-fir cotyledons. Plant Cell Rep. 10:517–521; 1991.
Hartman, H.; Kester, D. Plant propagation: principles and practices. New York: Prentice Hall; 1975:165–192.
Martínez Pulido, C.; Harry, I. S.; Thorpe, T. A. In vitro regeneration of plantlets of Canary Islands pine (Pinus canariensis). Can. J. For. Res. 20:1200–1211; 1991.
Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol. Plant. 15:473–479; 1962.
Phillips, R. Cytodifferentiation. In: Vasil, I. K., ed. Perspectives in plant cell and tissue culture. Int. Rev. Cytol. Suppl. IIA. New York: Academic Press; 1980:145–175.
Rajadurai, D.; Rao, A. N.; Loh, C. S. In vitro culture studies on two leguminous species. In: Rao, A. N.; Yusoff, A. M., ed. Proceedings of the seminar on Tissue Culture of Forest Species. Kuala Lumpur, 15–18 June: FRIM-IDRC; 1987:104–128.
Salazar, R.; Boshier, D. Establecimiento y manejo de rodales de semilleros de especies forestales prioritarias en América Central. Serie técnica. Informe técnico No. 148. Turrialba: CATIE; 1989:77.
SAS Institute. SAS’s user’s guide: statistics version 5. Cary, North Carolina: SAS Institute; 1985:597.
Saxena, P. K.; Gill, R. Plant regeneration from mesophyll protoplasts of the tree legume Pithecellobium dulce Benth. Plant Sci. 53:257–262; 1987.
Van Waes, J. Effect of activated charcoal on in vitro propagation of western European orchids. Acta Hort. 212:131–138.
Villalobos, V. M.; Leung, D. W. M.; Thorpe, T. A. Light-cytokinin interaction in shoot formation in cultured cotyledon explant of radiate pine. Physiol. Plant. 61:497–504; 1984.
Webb, D. T.; Flinn, B. S.; Georgis, W. Micropropagation of Eastern white pine (Pinus strobus L.). Can. J. For. Res. 18:1570–1580; 1988.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Valverde-Cerdas, L., Dufour, M. & Villalobos, V.M. In vitro propagation of Pithecellobium saman (Raintree). In Vitro Cell.Dev.Biol.-Plant 33, 38–42 (1997). https://doi.org/10.1007/s11627-997-0038-2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11627-997-0038-2