Summary
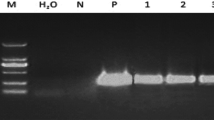
The expression of the green-fluorescent protein (GFP) gene from Aequorea victoria (jellyfish) was analyzed by transient and stable expression in sweet potato Ipomoea batatas L. (Lam.) ev. Beauregard tissues by electroporation and particle bombardment. Leaf and petiole segments from in vitro-raised young plantlets were used for protoplast isolation and electroporation. Embyrogenic callus was also produced from leaf segments for particle bombardment experiments. A buffer solution containing 1×106 protoplasts ml−1 was mixed with plasmid DNA containing the GFP gene, and electroporated at 375 V cm−1. Approximately 25–30% of electroporated mesophyll cell protoplasts subsequently cultured in KM8P medium regenerated cell walls after 48 h. Of these, 3% emitted bright green fluorescence when exposed to UV-blue light at 395 nm. Transformed cells continued to grow after embedding in KM8P medium solidifed with 1.2% SeaPlaque agarose. Stable expression of GFP was observed after 4 wk of culture in approximately 1.0% of the initial GFP positive cells (27.5 GFP positive micro callases out of 3024 cells which transiently expressed GFP 48 h after electroporation). In a separate experiment, 600–700 bright green spots were observed per plate 48 h after bombarding leaf segments or embryogenic cellus. In bombarded cultures, several stable GEP-expressing sectors were observed in leafderived embryogenic callus grown without selection for 4 wk. These results show that GFP gene expression can occur in various sweet potato tissues, and that it may be a useful sereenable marker to improve transformation efficiency and obtain transgenic sweet potato plants.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bowen, B. A. Markers for plant gene transfer. In: Kung, S. D.; Wec, R., eds. Transgenic plants: engineering and utilization, vol. I. San Diego: Academic Press: 1993:89–121.
Bower, R.; Elliott, A. R.; Potier, B. A. M.; Birch, R. G. High-efficiency microprojectile-mediated co-transformation of sugarcane using visible or selectable markers. Mol. Breeding 2:239–249; 1996.
Casper, S. J.; Holt, C. A. Expression of the green fluorescent proteinencoding gene from tobacco mosaic virus-based vector. Gene 173:69–73; 1996.
Chalfie, M.; Tu, Y.; Euskirchen, G.; Ward, W. W.; Prasher, D. C. Green fluorescent protein as a marker for gene expression. Science 263:802–805; 1994.
Chee, R. P.; Schultheis, J. R.; Cantliffe, D. J. Plant recovery from sweet potato embryos. Hortscience 25:795–797; 1990.
Dhir, S. K.; Dhir, S.; Hepburn, A.; Widholm, J. M. Factors affecting transient gene expression in electrophorated Glycine max protoplasts. Plant Cell Rep. 10:106–110; 1991.
Dhir, S. K.; Oglesby, J.; Bhagsari, A. S. Plant regeneration via somatic embryogenesis, and transient gene expression in sweet potato protoplasts. Plant Cell Rep. 17:665–669; 1998.
Dhir, S. K.; Pajeau, M. E.; Fromm, M. E.; Fry, J. E. Anthocyanin genes as visual markers for wheat transformation. In: Henry, R. J.; Ronalds, J. A., eds. Improvement of cereal quality by genetic engineering. New York: Plenum Press; 1994:71–75.
Elliott, A. B.; Campbell, J. A.; Dougdale, B.; Bretell, R. I. S.; Grof, C. P. L. Green fluorescent protein facitilates rapid in vivo detection of genetically transformed plant cells. Plant Cell Rep. 18:707–714; 1999.
Finer, J. J.; McMullen, M. D. Transformation of cotton (Gossypium hirsutum L.) via particle bomhardment. Plant Cell Rep. 8:586–589; 1990.
Frank, T.; Birch, R. C. Gene transfer into intact sugarcane cells using microprojectile bombardment, Aust. J. Plant Physiol. 18:471–480; 1991.
Galbraith, D. W. Flow cytometric and sorting of plant protoplasts and cells. Methods Cell Biol. 42:539–561; 1994.
Gama, I. C. S.; Leite, R. P., Jr.; Cordeiro, A. R.; Cantliffe, D. J. Transgenic sweetpotato plants obtained by Agrobacterium tumerfacriens-mediated transformation. Plant Cell Tiss. Organ Cult. 46:237–244; 1996.
Geest, A. H. M.; Petolino, J. F. Expression of a modified green fluorescent protein gene in transgenic maize plants and progeny. Plant Cell Rep. 17:760–764; 1998.
Grebenok, R. J.; Pierson, E.; Lambert, G. M.; Gong, F. C.; Afonso, C. L.; Haldeman-Cahill, R.; Carrington, J. C.; Galbraith, D. W. Greenfluorecent protein fusions for efficient characteization of nuclear targeting. Plant J. 11:573–586; 1997.
Haseloff, J.; Amos, B. GFP in plants. Trends Cenet. 11:328–329; 1995.
Haseloff, J.; Siemering, K. R.; Prasher, D. C. Removal of eryptic intron and subcellular localization of green fluorescent protein are required to mark transgenic Arabidopsis plant brightly. Proc. Natl Acad. Sci. USA 94:2122–2127; 1997.
Heim, R.; Cubitt, A. B.; Tsien, R. Y. Improved green fluorescence. Nature 373:663–664; 1995.
Hunold, R.; Bronner, R.; Hahne, G. C. Early events in microprojectile bombardment: cell viability and particle location. Plant J. 5:593–604; 1994.
Jefferson, R. A.; Kavangh, T. A.; Bevan, M. W. GUS fusion: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J. 6:3901–3907; 1987.
Kao, K. N.; Michayluk, M. R. Nutritional requirements for growth of Vicia hajastana cell and protoplasts at very low population density in liquid media. Planta 126:105–110; 1975.
Kohler, R. H.; Zipfel, W. R.; Webb, W. W.; Hanson, M. R. The green fluorescent protein marker to visualize plant mitochondria in vivo. Plant J. 11:613–621; 1997.
Ludwig, S. R.; Bowen, B.; Beach, L.; Wessler, S. R. A regulatory gene as a novel visible marker for maize transformation. Science 247:449–450; 1990.
Mitchell, T.; Bhagsari, A. S.; Ozias-Akins, P.; Dhir, S. K. Electroporationmediated transient gene expression in intact cells of sweet potato. In Vitro Cell. Dev. Biol. Plant 34:319–324; 1998.
Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15:473–479; 1962.
Neidz, R. P.; Sussman M. R.; Satterlee, J. S. Green fluorescent protein: an in vivo reporter of plant gene expression. Plant Cell Rep. 14:403–406; 1995.
Newell, C. A.; Lowe, J. M.; Merryweather, A.; Rooke, L. M.; Hamilton, W. D. O. Transformation of sweetpotato (Ipomoea balatas (L.) Lam.) with Agrobacterium tumefaciens and regeneration of plants expressing cowpea trypsin inhibitor and snowdrop lectin. Plant Sci. 107:215–227; 1995.
Pang, S.; DeBoer, D. L.; Wan, Y.; Ye, G.; Layton, J. G.; Neher, M. K.; Armstrong, C. L.; Fry, J. C.; Hinchee, M. A. W.; Fromm, M. E. An improved green fluorescent protein gene as a vital marker in plants. Plant Physiol. 112:893–900; 1996.
Pines, J. GFP in mammalian cells. Trends Genet. 11:326–327; 1995.
Plautz, J. D.; Richard, N. D.; Dailey, G. N.; Welsh, S. B.; Hall, J. C.; Halpain, S.; Kay, S. A. Green fluorescent protein in and its derivatives as versatile markers for gene expression in living Drosophila melanogaster, plant and mammalian cells. Gene 173:83–87; 1996.
Prakash, C. S.; Varadarajan, U. Genetic transformation of sweet potato by particle bombardment. Plant Cell Rep. 11:53–57; 1992.
Prasher, D. C.; Eckenrode, V. K.; Ward, W. W.; Prendergast, F. G.; Cornier, M. J. Primary structure of the Aequorea victoria green-fluorescent protien. Gene 111:229–233; 1992.
Reichel, C.; Mathur, J.; Eckes, P.; Langenkemper, K.; Konez, C.; Schell, J.; Reiss, B.; Mass, C. Enhauced green fluorescencer by the expression of an Aequorea victoria green fluorecent protein mutant in mono-and dicotyledoneous plant cells. Proc. Nalt Acad. Sci. USA 93:5888–5893; 1996.
Sambrook, K. J.; Fritsch, E. F.; Maniatis, T. C. Molecular cloning: a laboratory manual. 2nd edn. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press; 1989.
Saunders, J. A.; Matthews, B. F.; Miller, P. D. Plant gene transfer using electrofusion and electroporation. In: Neumann, E.; Sowers, A.; Jordan, C., eds. Electroporation and electrofusion in cell biology. New York: Plenum Press; 1996:343–354.
Sheen, J.; Hwang, S.; Niwa, Y.; Kobyashi, H.; Galbraith, D. W. Greenfluorescent protein as a new vital marker in plant cells. Plant J. 8:777–784; 1995.
Suter-Crazzolara, C.; Klemm, M.; Reiss, B., In: Galbraith, D. W., ed. Methods in cell biology: methods in plant cell biology, part B. San Diego: Academic Press: 1995:425–438.
Tian, L.; Sequin, A.; Charest, P. J.; Expression of the green fluorescent protein gene in conifer tissues. Plant Cell Rep. 16:267–271; 1997.
Wang, S.; Hazelrigg, T. Implication for bcd mRNA localization from spatial distribution of exu protein in Drosophila oogenesis. Nature 369:400–403; 1994.
Youvan, D. C.; Larrick, J. W. Special issue on CFP. Gene 173:1–117; 1996.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Winfield, S., Lawton, R., Daniell, H. et al. Transformation of sweet potato tissues with green-fluorescent protein gene. In Vitro Cell.Dev.Biol.-Plant 37, 648–653 (2001). https://doi.org/10.1007/s11627-001-0113-z
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11627-001-0113-z