Summary
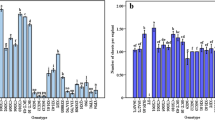
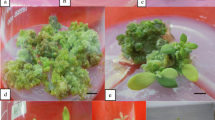
An in vitro method was developed for high-frequency shool regeneration and continuous production of rapid-cycling B. oleracea in large numbers. The high regenerative capacity was tissue-dependent. Developmental polarity (apical and basal ends) of the explants appeared to play a regulatory role in shoot morphogenesis in this system. High-frequency shoot regeneration was obtained with N6-benzyladenine or thidiazuron-supplemented media. Delayed and reduced regenerative ability of cultures in air-tight vessels and the dramatic suppression of shoot regeneration in internodal explants by the ethylene precursor 1-aminocyclopropane-1-carboxylic acid implicate a possible involvement of ethylene in shoot morphogenesis in this species. Rotting of regenerated shoots of B. oleracea occurred readily on α-naphthaleneacetic acid-supplemented media. Rooted plantlets were successfully established in soil and developed normal fertile flowers and viable seeds.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Arumuganathan, K.; Earle, E. D. Nuclear DNA content of some important plant species. Plant Mol. Biol. Rep. 9:208–218; 1991.
Aslam, F. N.; MacDonald, M. V.; Ingram, D. S. Rapid-cycling Brassica species: anther culture potential of B. campestris L. and B. napus L. New Phytol. 115:1–9; 1990.
Bajaj, Y. P. S.; Nietsch, P. In vitro propagation of red cabbage (Brassica oleracea L. var capitata). J. Exp. Bot. 26:883–890; 1975.
Berthomieu, P.; Jouanin, L. Transformation of rapid cycling cabbage (Brassica oleracea var capitata) with Agrobacterium rhizogenes. Plant Cell Rep. 11:334–338; 1992.
Chi, G. L.; Pua, E. C.; Goh, C. J. Role of ethylene on de novo shoot regeneration from cotyledonary explants of Brassica campestris spp. pekinensis (Lour) Olsson in vitro. Plant Physiol. 96:178–183; 1991.
Fosket, D. E. Plant growth and development. California: Academic Press; 1994:363–369.
Hansen, L. N.; Earle, E. D. Regeneration of plants from protoplasts of rapid cycling Brassica oleracea L. Plant Cell Rep. 13:335–339; 1994.
Kik, C.; Zaal, M. A. C. M. Protoplast regeneration from Brassica oleracea “rapid cycling’ and other varieties. Plant Cell Tiss. Organ Cult. 35:107–114; 1993.
Kumar, P. P.; Lakshmanan, P.; Thorpe, T. A. Regulation of morphogenesis in plant tissue culture by ethylene. In Vitro Cell Dev. Biol. Plant 34:94–103; 1998.
Lakshmanan, P.; Ng, S. K.; Loh, C. S.; Goh, C. J. Auxin, cytokinin and ethylene differentially regulate specific developmental states associated with shoot bud morphogenesis in leaf tissues of mangosteen (Garcinia mangostana L.) cultured in vitro. Plant Cell Physiol. 38:59–64; 1997.
Larkin, P. J.; Scowcroft, W. R. Somaclonal variation—a novel source of variability from cell cultures for plant improvement. Theor. Appl. Genet. 60:197–214; 1981.
Leung, H.; Williams, P. Cytoplasmic male sterile Brassica campestris breeding lines with resistance to clubroot, turnip mosaic, and downy mildew. HortScience 18:774–775; 1983.
Loudon, P. T.; Nelson, R. S.; Ingram, D. Studies of protoplast culture and plant regeneration from commercial and rapid-cycling Brassica species. Plant Cell Tiss. Organ Cult. 19:213–224; 1989.
Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant 15:473–497; 1962.
Narasimhulu, S. B.; Kirti, P. B.; Mohapatra, T.; Prakash, S.; Chopra, V. L. Shoot regeneration in stem explants and its amenability to Agrobacterium tumefaciens mediated gene transfer in Brassica carinata. Plant Cell Rep. 11:359–362; 1992.
Paterson, K. E. Polarity of regeneration in excised leaves of Crassula argentea. 1. A role of auxin. Can. J. Bot. 61:1058–1063; 1983.
Pua, E. C. Cellular and molecular aspects of ethylene on plant morphogenesis of recalcitrant Brassica species in vitro. Bot. Bull. Acad. Sinica 34:191–209; 1993.
Bood, S.; Hedden, P. Convergent pathways of gibberellin A1 biosynthesis in Brassica. Plant Growth Reg. 15:241–246; 1994.
Stringam, G. R. Regeneration in stem explants of haploid rapessed (Brassica napus L.) Plant Sci. Lett. 9:115–119; 1977.
Teo, W.; Lakshmanan, P.; Kumar, P.; Goh, C. J.; Swarup, S. Direct shoot formation and plant regeneration from cotyledon explants of rapid-cycling Brassica rapa. In Vitro Cell. Dev. Biol. Plant 33:288–292; 1997.
Williams, P. H.; Hill, C. B. Rapid-cycling populations of Brassica. Science 232:1385–1390; 1986.
Wong, R. S. C.; Zee, S. Y.; Swanson, E. B. Isolated microspore culture of Chinese flowering cabbage (Brassica campestris spp. parachinensis) Plant Cell Rep. 15:396–400; 1996.
Yang, M. Z.; Jia, S. R.; Pua E. C. High frequency of plant regeneration from hypocotyl explants of Brassica carinata. Plant Cell Tiss. Organ Cult. 24:79–82; 1991.
Zar, J. H. Biostatistical analysis 2nd edn. Englewood Cliffs, NJ: Prentice-Hall; 1984:718.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cheng, PK., Lakshmanan, P. & Swarup, S. High-frequency direct shoot regeneration and continuous production of rapid-cycling Brassica oleracea in vitro . In Vitro Cell.Dev.Biol.-Plant 37, 592–598 (2001). https://doi.org/10.1007/s11627-001-0104-0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11627-001-0104-0