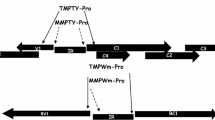
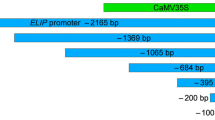
Summary
Two different promoters, a cauliflower mosaic virus (CaMV) 35S promoter with a 5′-untranslated leader sequence from alfalfa mosaic virus RNA4 (designated as CaMV 35S/AMV) and an E-8 fruit-ripening-specific promoter, were compared to evaluate their effects on expression of the uidA reporter gene in transgenic tomato plants. In order to generate sufficient numbers of transgenic tomato plants, both a reliable regeneration system and an efficient Agrobacterium transformation protocol were developed using 8-d-old cotyledons of tomato (Lycopersicon ecsulentum Mill. cv. Swifty Belle). Two sets of constructs, both derivatives of the binary vector pBI121, were used in transformation of tomato whereby the uidA gene was driven either by the CaMV 35S/AMV or the E-8 fruit-ripening-specific promoter. Southern blot hybridization confirmed the stable integration of the chimeric uidA gene into the tomato genome. Fruit and leaf tissues were collected from T0 and T1 plants, and assayed for β-glucuronidase (GUS) enzyme activity. As expected, both vegetative and fruit tissues of transgenic plants carrying the uidA gene under the control of CaMV 35S/AMV showed varying levels of GUS activity, while no expression was observed in vegetative tissues of transgenic plants carrying the uidA gene driven by the E-8 promoter. All fruits from transgenic plants produced with both sets of constructs displayed expression of the uidA gene. However, when this reporter gene was driven by the CaMV 35S/AMV, GUS activity levels were significantly higher than when it was driven by the E-8 fruit-specific promoter. The presence/absence of the uidA gene in T1 plants segregated in a 3∶1 Mendelian ratio.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Charest, P. J.; Calero, N.; Lachance, D.; Datla, R. S. S.; Duchesne, L. S.; Tsang, E. W. T. Microprojectile-DNA delivery in conifer species: factors affecting assessment of transient gene expression. Plant Cell Rep. 12:189–193; 1993.
Chyi, Y.-S.; Phillips, G. C. High efficiency Agrobacterium-mediated transformation of Lycopersicon based on conditions favorable for regeneration. Plant Cell Rep. 6:105–108; 1987.
Coupe, S. A.; Deikman, J. Characterization of a DNA-binding protein that interacts with a 5′ flanking region of two fruit-ripening genes. Plant Cell 11:1207–1218; 1997.
D'Aoust, M. A.; Nguyen-Quoc, B.; Le, V. Q.; Yelle, S. Upstream regulatory regions from the maize Sh1 promoter confer tissue-specific expression of the β-glucuronidase gene in tomato. Plant Cell Rep. 18:803–808; 1999.
Datla, R. S. S.; Bekkaoui, F.; Hammerlindl, J. K.; Pilate, G.; Dunstan, D. I.; Crosby, W. L. Improved high-level constitutive foreign gene expression in plants using an AMV RNA4 untranslated leader sequence. Plant Sci. 94:139–149; 1993.
Davis, M. E.; Miller, A. R.; Lineberger, R. D. Temporal competence for transformation of Lycopersicon esculentum L. (Mill.) cotyledons by Agrobacterium tumefaciens: relation to wound-healing and soluble plant factors. J. Exp. Bot. 42:359–364; 1991.
Deikman, J.; Xu, R.; Kneissi, M. L.; Ciardi, J. A.; Kim, K.-N.; Pelah, D. Separation of cis elements responsive to ethylene, fruit development, and ripening in the 5′-flanking region of the ripening-related E8 gene. Plant Mol. Biol. 37:1001–1011; 1998.
Filatti, J. J.; Kiser, J.; Rose, R.; Comai, L. Efficient transfer of a glyphosate tolerance gene into tomato using Agrobacterium tumefaciens vector. Bio/Technology 5:726–730; 1987.
Gamborg, O. G.; Miller, R. A.; Ojima, K. Nutrient requirements of suspension cultures of soybean root cells. Exp. Cell Res. 50:151–158; 1968.
Good, X.; Kellog, J. A.; Wagoner, W.; Landhoff, D.; Matsumura, W.; Bestwick, R. K. Reduced ethylene synthesis by transgenic tomatoes expressed S-adenosylmethionine hydrolase. Plant Mol. Biol. 26:781–790; 1994.
Haggman, H.; Aronen, T. Transgene expression in regenerating cotyledons and embryogenic cultures of Scots pine. J. Exp. Bot. 49:1147–1156; 1998.
Hamza, S.; Chupeau, Y. Re-evaluation of conditions for plant regeneration and Agrobacterium-mediated transformation from tomato (Lycopersicon esculentum). J. Exp. Bot. 44:1837–1845; 1993.
Huetteman, C. A.; Preece, J. E. Thidiazuron: a potent cytokinin for woody plant tissue culture. Plant Cell Tiss. Organ Cult. 33:105–119; 1993.
Jefferson, R. A. Assaying chimeric genes in plants: the GUS gene fusion system. Plant Mol. Biol. Rep. 5:387–405; 1987.
Jefferson, R. A.; Burgess, S. M.; Hirsh, D. β-Glucuronidase from Escherichia coli as a gene-fusion marker. Proc. Natl Acad. Sci. USA 83:8447–8451; 1986.
Jelesko, J. G.; Jenkins, S. M.; Rodriguez-Cincepcion, M.; Gruissem, W. Regulation of tomato HMG1 during cell proliferation and growth. Planta 208:310–318; 1999.
Joao, K. H. L.; Brown, T. A. Enhanced transformation of tomato cocultivated with Agrobacterium tumefaciens C58C1Rif::pGSFR1161 in the presence of acetosyrengone. Plant Cell Rep. 12:422–425; 1993.
Kosugi, S.; Ohashi, Y.; Nakajima, K.; Arai, Y. An improved assay for β-glucoronidase in transformed cells: methanol almost completely suppresses a putative endogenous β-glucoronidase activity. Plant Sci. 70:133–140; 1990.
Krasnyanski, S.; May, R. A.; Loskutov, A.; Sink, K. S. Transformation of the limonene synthase gene into peppermint (Mentha piperita L.) and preliminary studies on the essential oil profiles of single transgenic plants. Theor. Appl. Genet. 99:676–682; 1999.
Ling, H.-Q.; Kriseleit, D.; Ganan, M. W. Effect of ticarcillin/potassium clavulanate on callus growth and shoot regeneration in Agrobacterium-mediated transformation of tomato (Lycopersicon esculentum). Plant Cell Rep. 17:843–847; 1998.
Liu, T.-H.A.; Stephens, L. C.; Hannapel, D. J. Transformation of Solanum brevidens using Agrobacterium tumefaciens. Plant Cell Rep. 15:196–199; 1995.
McCormick, S.; Niederrneyer, J.; Fry, J.; Barnason, A.; Horsch, R.; Fraley, R. Leaf disc transformation of cultivated tomato (Lycopersicon esculentum) using Agrobacterium tumefaciens. Plant Cell Rep. 5:81–84; 1986.
Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15:473–479; 1962.
Nicholass, F. J.; Smith, C. J. S.; Schuch, W.; Bird, C. R.; Grierson, D. High levels of ripening-specific reporter gene expression directed by tomato fruit polygalacturonase gene-flanking regions. Plant Mol. Biol. 28:423–435; 1995.
Pawlowski, W. P.; Somers, D. A. Transgene inheritance in plants genetically engineered by microprojectile bombardment. Mol. Biotechnol. 6:17–30; 1996.
Sambrook, J.; Fritsch, E. F.; Maniatis, T. Molecular cloning: a laboratory manual, 2nd edn, vol. 2. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press; 1989.
Sandhu, J. S.; Krasnyanski, S. F.; Osadjan, M. D.; Domier, L. L.; Korban, S. S.; Buetow, D. E. Oral immunization of mice with transgenic tomato fruit expressing respiratory syncytial virus-F protein induces a systemic immune response. Transgenic Res. 9:127–135; 2000.
Sandhu, J. S.; Osadjan, M. D.; Krasnyanski, S. F.; Domier, L. L.; Korban, S. S.; Buetow, D. E. Enhanced expression of the human respiratory syncytial virus-F gene in apple protoplasts. Plant Cell Rep. 18:394–397; 1999.
Schnurr, J. A.; Guerra, D. J. The CaMV-35S promoter is sensitive to shortened photoperiod in transgenic tobacco. Plant Cell Rep. 19:279–282; 2000.
Serres, R.; McCown, B.; Zeldin, E. Detectable β-glucuronidase activity in transgenic cranberry is affected by endogenous inhibitors and plant development. Plant Cell Rep. 16:641–646; 1997.
Van Eck, J. M.; Blowers, A. D.; Earle, E. D. Stable transformation of tomato cell cultures after bombardment with plasmid and YAC DNA. Plant Cell Rep. 14:299–304; 1995.
Van Haaren, M. J. J.; Houck, C. M. A functional map of the fruit-specific promoter of the tomato 2A11 gene. Plant Mol. Biol. 21:625–640; 1993.
Van Roekel, J. S. C.; Damm, B.; Melchers, L. S.; Hoekema, A. Factors influencing transformation frequency of tomato (Lycopersicon esculentum). Plant Cell Rep. 12:644–647; 1993.
Warkentin, T. D.; Jordan, M. C.; Hobbs, S. L. A. Effect of promoter-leader sequence on transient reporter gene expression in particle bombardment pea (Pisum sativum L.) tissues. Plant Sci. 87:171–177; 1992.
Xu, R.; Goldman, S.; Coupe, S.; Deikman, J. Ethylene control of E4 transcription during tomato fruit ripening involves two cooperative cis elements. Plant Mol. Biol. 31:1117–1127; 1996.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Krasnyanski, S.F., Sandhu, J., Domier, L.L. et al. Effect of an enhanced CaMV 35S promoter and a fruit-specific promoter on uida gene expression in transgenic tomato plants. In Vitro Cell.Dev.Biol.-Plant 37, 427–433 (2001). https://doi.org/10.1007/s11627-001-0075-1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11627-001-0075-1