Summary
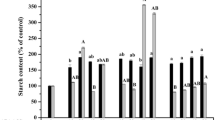
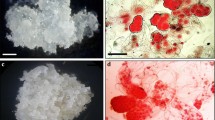
The influence of carbon sources and polyethylene glycol combined with 0.45 and 0.9% (w/v) of gellan gum on the maturation of maritime pine somatic embryos was tested. The effect of the carbon source and polyethylene glycol varied widely between lines. One out of the five lines tested showed a striking response to polyethylene glycol (PEG) treatment; the addition of this osmoticum limited the embryonal-suspensor mass (ESM) proliferation while it enhanced the maturation rate. Conversely, the ESM proliferation was stimulated by PEG in the other lines without subsequent improvement of the maturation rate. The use of a high concentration of gellan gum (0.9%) improved the maturation of the five ESM lines. It was concluded that the most efficient culture medium to recover cotyledonary embryos from all lines is one supplemented with sucrose at 6% (w/v) and gellan gum at 0.9% (w/v) without PEG. The determining factor in the maturation of maritime pine somatic embryos is the genotype and/or the quality of ESM. The possible relationship between maturation performances and ESM morphology, particularly the suspensor organization, is discussed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Attree, S.; Moore, D.; Sawhney, V. K.; Fowke, L. C. Enhanced maturation and desiccation tolerance of white spruce [Picea glauca (Moench) Voss] somatic embryos: effects of a non-plasmolysing water stress and abscisic acid. Ann. Bot. 68:519–525; 1991.
Bercetche, J.; Galerne, M.; Dereuddre, J. Efficient regeneration of plantlets from embryogenic callus of Picea abies (L.) Karst, after freezing in liquid nitrogen. C. R. Acad. Sci. 310:357–363; 1990.
Bercetche, J.; Pâques, M. Somatic embryogenesis in maritime pine (Pinus pinaster). In: Jain, S. M.; Gupta, P. K.; Newton, R. J., eds. Somatic embryogenesis in woody plants. Vol. 3. Gymnosperms. Dordrecht, Netherlands. Kluwer Academic Publishers; 1995; 221–242.
Chaperon, H.; Alazard, P.; Brunet, A. M. Place of the vegetative propagation by cuttings in the breeding programme of the maritime pine (Pinus pinaster). Proceedings of the IUFRO joint meeting of working parties on genetics about breeding strategies including multiclonal varieties. Sensenstein. Germany, September 6–10; 1982; 89.
Find, J. I.; Nørgaard, J.; Krogstrup, P. Growth parameters, nutrient uptake and maturation capacity of two cell-lines of Norway spruce (Picea abies) in suspension culture. J. Plant Physiol. 152:510–517; 1998.
Gupta, P. K. Method for reproducing, conifers by somatic embryogenesis using maltose enriched maintenance medium. International Patent WO 95/14373; 1995.
Gupta, P. K.; Durzan, D. J. Shoot multiplication from mature trees of Douglas fir (Pseudotsuga menziesii) and sugar pine (Pinus lambertiana). Plant Cell Rep. 4:177–179; 1985.
Gutmann, M.; von Aderkas, P.; Lelu, M-A. Effects of abscisic acid on somatic embryo maturation of hybrid larch. J. Exp. Bot. 47(305):905–1917; 1996.
Hakman, I.; von Arnold, S. Somatic embryogenesis and plant regeneration from suspension cultures of Picea glauca (White spruce). Physiol. Plant. 72:579–587; 1988.
Hristoforoglu, K.; Schmidt, J.; Bolhar-Nordenkampf, H. Development and germination of Abies alba somatic embryos. Plant Cell Tiss. Organ Cult. 40:277–284; 1995.
Jokinen, K. J.; Durzan, D. J. Properties of rescued embryonal suspensor masses of Norway spruce determined by the genotype and the environment in vitro. Silva Fennica 28(2):95–106; 1994.
Jones, N. B.; Ross, J.; van Staden, J. Improved somatic embryo production from embryonic tissue of Pinus patula. In: Proceedings of the IX IAPTC Congress. Plant biotechnology and in vitro biology in the 21st century. Jerusalem, Israel, June 14–19; 1998; 86.
Kapik, R. H.; Dinus, R. J.; Dean, J. F. D. Abscisic acid and zygotic embryogenesis in Pinus taeda. Tree Physiol. 15:485–490; 1995.
Klimaszewska, K.; Smith, D. R. Maturation of somatic embryos of Pinus strobus is promoted by a high concentration of gellan gum. Physiol. Plant. 100:949–957; 1997.
Klimaszewska, K.: Sutton, B. S.; Polonenko, D. R.; Cyr, D. R.; Stodola T. F. Maturation of somatic embryos. International Patent WO 99/46977; 1999.
Lelu, M-A.; Bastien, C.; Drugeault, A.; Gouez, M-L.; Klimaszewska, K. Somatic embryogenesis and plantlet development in Pinus sylvestris and Pinus pinaster on medium with or without growth regulators. Physiol. Plant. 105:719–728; 1999.
Li, X. Y.; Huang, F. H.; Murphy, J. B.; Gbur, E. E. Polyethylene glycol and maltose enhance somatic embryo maturation in loblolly pine (Pinus taeda). In Vitro Cell. Dev. Biol. Plant 34:22–26; 1998.
Nørgaard, J. V. Somatic embryo maturation and plant regeneration in Abies nordmanniana Lk. Plant Sci. 124:211–221; 1997.
Roth, R.; Ebert, J.; Schmidt, J. Trisomy associated with loss of maturation capacity in a long-term embryogenic culture of Abies alba. Theor. Appl. Genet. 95:353–358; 1997.
Salajova, T.; Salaj, J.; Kormutak, A. Initiation of embryogenic tissue and plantlet regeneration from somatic embryos of Pinus nigra Arn. Plant Sci. 145:33–40; 1999.
Scott, P.; Lyne, R. L.; Rees, T. Metabolism of maltose and sucrose by microspores isolated from barley (Hordeum vulgare). Planta 197:435–441; 1995.
Tremblay, L.; Tremblay, F. M. Maturation of black spruce somatic embryos: sucrose hydrolysis and resulting osmotic pressure of the medium. Plant Cell Tiss. Organ Cult. 42:39–46; 1995.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ramarosandratana, A., Harvengt, L., Bouvet, A. et al. Effects of carbohydrate source, polyethylene glycol and gellan gum concentration on embryonal-suspensor mass (ESM) proliferation and maturation of maritime pine somatic embryos. In Vitro Cell.Dev.Biol.-Plant 37, 29–34 (2001). https://doi.org/10.1007/s11627-001-0006-1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11627-001-0006-1