Summary
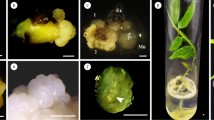
Plants were regenerated from cotyledon tissue of greenhouse grown seedlings of common buckwheat (Fagopyrum esculentum Moench.). Maximum callus regeneration was induced on Murashige and Skoog (MS) medium containing 2,4-D (2.0 mg l−1) and kinetin (KIN) (0.2 mg l−1) and either 3 or 6% sucrose. Friable callus was transferred to MS media containing KIN and benzylaminopurine (BAP) at varied concentrations for embryogenic callus induction. The optimum medium for embryogenic callus induction was found to be MS medium supplemented with 0.2 mg l−1 KIN, 2.0 mg l−1 BAP and 3% (w/v) sucrose. Variation of sucrose from 3 to 6% did not show any significant effect on callus induction or embryogenesis. Regeneration of embryonic callus varied from 13 to 32%. Whole plants were obtained at high frequencies when the embryogenic calluses with somatic embryos and organized shoot primordia were transferred to half-strength MS media with 3% sucrose. Regenerated plants after acclimation were transferred to greenhouse conditions, and both vegetative and floral characteristics were observed for variation. This regeneration system may be valuable for genetic transformation and cell selection in common buckwheat.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adachi, T.; Yamaguchi, A.; Miike, Y.; Hoffmann, F. Plant regeneration from protoplasts of common buckwheat (Fagopyrum esculentum). Plant Cell Rep. 8:247–250; 1989.
Bohanec, B.; Neskovic, M.; Vujicic, R. Anther culture and androgenic plant regeneration in buckwheat (Fagopyrum esculentum Moench.). Plant Cell Tiss. Organ. Cult. 35:259–266; 1993.
Kreft, I. Buckwheat breeding perspectives. In: Nagatomo, T.; Adachi, T., eds. Buckwheat Research. Proceedings of the 2nd International Symposium on Buckwheat. Japan: Miyazaki University; 1983:3–12.
Lachmann, S. Plant cell and tissue culture in buckwheat: an approach towards genetic improvement by means of unconventional breeding techniques. In: Adachi, T., ed. Overcoming breeding barriers by means of plant biotechnology. Proc. Intl. Coll., Miyazaki, Japan. 145–154; 1991.
Lachmann, S.; Adachi, T. Callus regeneration from hypocotyl protoplasts of Tartary buckwheat (Fagopyrum tataricum Gaertn). Fagopyrum 10:62–64; 1990.
Mijus-Djukic, J.; Neskovic, M.; Ninkovic, S.; Crkvenjakov, R. Agrobacterium-mediated transformation and plant regeneration of buckwheat (Fagopyrum esculentum Moench.). Plant Cell Tiss. Organ. Cult. 29:101–108; 1992.
Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant 15:473–497; 1962.
Neskovic, M.; Culafic, L.; Vujicic, R. Somatic embryogenesis in buckwheat (Fagopyrum Mill.) and sorrel (Rumex L.). Polygonaceae. In: Bajaj, Y. P. S., ed. Biotechnology in agriculture and forestry, Vol. 31. Somatic embryogenesis and synnetic seed II. Berlin, Heidelberg, New York: Springer-Verlag; 412–427; 1995.
Neskovic, M.; Vujicic, R.; Budimir, S. Somatic embryogenesis and bud formation from immature embryos of buckwheat (Fagopyrum esculentum Moench.). Plant Cell Rep. 6:423–426; 1987.
Rumyantseva, N. I.; Sergeeva, N. V.; Khakimova, L. E.; Salnikov, V. V.; Gumerova, E. V.; Lozovaya, V. V. Organogenesis and somatic embryogenesis in tissue culture of two buckwheat species. Fiziol. Rast. 36:187–194; 1989 (in Russian).
Srejovic, V.; Neskovic, M. Regeneration of plants from cotyledon fragments of buckwheat (Fagopyrum esculentum Moench.). Z. Pflanzenphysiol. 104:37–42; 1981.
Takahata, Y. Plant regeneration from cultured immature inflorescence of common buckwheat (Fagopyrum esculentum Moench.) and perennial buckwheat (F. cymosum Meisn.). Jpn J. Breed 38:409–413; 1988.
Yamane, Y. Induced differentiation of buckwheat plants from subcultured calluses in vitro. Jpn J. Genet. 48:139–146; 1974.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Woo, S.H., Nair, A., Adachi, T. et al. Plant regeneration from cotyledon tissues of common buckwheat (Fagopyrum esculentum Moench). In Vitro Cell.Dev.Biol.-Plant 36, 358–361 (2000). https://doi.org/10.1007/s11627-000-0063-x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11627-000-0063-x