Abstract
Heat stress–induced reductions in milk yield and the dysfunction of mammary glands are economically important challenges that face the dairy industry, especially during summer. The aim of the present study is to investigate the effects of heat stress on mitochondrial function by using dairy cow mammary epithelial cells (DCMECs) as an in vitro model. Live cell imaging shows that the mitochondria continually change shape through fission and fusion. However, heat stress induces the fragmentation of mitochondria, as well as the decreased of ATP level, membrane potential, and anti-oxidant enzyme activity and the increased of respiratory chain complex I activity. In addition, the cytosolic Ca2+ concentration and cytochrome c expression (Cyto-c) were increased after heat stress treatment. Both qRT-PCR and western blot analysis indicate that mitofusin1/2 (Mfn1/2) and optic atrophy protein-1 (Opa-1) are downregulated after heat stress, whereas dynamin-related protein 1 (Drp1) and fission 1 (Fis-1) are upregulated, which explains the observed defect of mitochondrial network dynamics. Accordingly, the present study indicated that heat stress induced the dysfunction of DCMEC through disruption of the normal balance of mitochondrial fission and fusion.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Heat stress, one of the major problems in tropical and subtropical regions, adversely affects large-size livestock farming production (Tao et al. 2018; Liu et al. 2019). Dairy cattle are one of the most sensitive livestock to high temperature, with excessive heat reducing their both reproductive performance and production potential (Herbut et al. 2018). Previous studies have reported that heat stress reduces the feed intake, milk yield and quality, and fertility and induces the metabolic disorders of dairy cattle. It also increases respiratory rates, heart rates, and peripheral blood flow (Takahashi 2012; Polsky and von Keyserlingk 2017; Tao et al. 2018). In addition, heat stress also affects the development of mammary glands, which is the main reason underlying observed reductions in milk yield during dry periods (Tao et al. 2018).
The negative effect of heat stress on mammary gland function is well known, and the resulting dysfunction is accompanied by apoptosis and autophagy, as well as reductions in mammary epithelial cells. Heat stress also reduces immunity and eventually increases the susceptibility of dairy cattle to mastitis (Wohlgemuth et al. 2016). Histological studies have shown that heat stress reduces levels of macrophages and lymphocytes and increases neutrophil count, as well as the increase of apoptotic cells in mammary gland, which is identified by the morphological changes (e.g., nuclear fragmentation, nuclear and cytoplasmic condensation, and the formation of apoptotic bodies) (Lindsay 2004).
Mammary epithelial cells (MECs) are the essential secretory cells of mammary glands, which involved in milk production (Boutinaud et al. 2015). Previous studies have reported that heat stress is associated with swollen mitochondria with broken cristae, low matrix density, and reduced myocardium ATP content, suggesting that the mitochondrial function is disrupted after heat stress (Qian et al. 2004). Mitochondria are double membrane–bound subcellular organelles that are the main source of cellular energy, owing to their generation of ATP through respiration and regulation of cellular metabolism. The morphology and population size of mitochondria were dynamically changed through continuous fission and fusion (Scott and Youle 2010). The imbalance of mitochondria fission and fusion can cause the dysfunction of mitochondria.
Mitochondrial fission and fusion processes are mediated by the dynamin family of large GTPases. The outer membrane protein Fis1 and the cytosolic protein Drp1, which are responsible for fission, are recruited to form spirals around mitochondria, thereby severing both inner and outer membranes (Knott et al. 2008). Meanwhile, the fusion of mitochondrial outer membranes is regulated by the mitochondrial outer membrane protein Mfn1/2, whereas fusion of mitochondrial inner membranes is mediated by Opa1, which located in the inner mitochondrial membrane (Hoppins et al. 2007). Depletion of Mfn1/2 results in severe mitochondrial fragmentation (Chen et al. 2003). A recent study showed that mitochondrial fusion and fission could eliminate the damaged material and maintain a healthy mitochondrial population (Liesa and Shirihai 2013). Furthermore, the skeletal muscle of Mfn1/2 transgenic mice exhibits several hallmarks of mitochondrial dysfunction, including mitochondrial swelling, loss of respiratory complex activity, and accumulation of mtDNA mutations (Chen et al. 2010). Meanwhile, mutation of Drp1 in mammalian cells has been reported to increase mitochondrial interconnectivity, which has similar mitochondrial morphology phenotype compared with Fis1-depleted cells, whereas overexpression of Drp1 or Fis1 increases mitochondrial fragmentation (Yoon et al. 2003). Overexpression of Mfn1 and Mfn2 could delay the occurrence of apoptosis (Sugioka et al. 2004), thereby mitochondrial fusion which regulated by Mfn1/2 may involve in apoptotic process. A previous study reported that heat stress could induce swollen mitochondria in bovine mammary epithelial cells (Du et al. 2008). However, the mechanism underlying the effects of heat stress on mitochondrial morphology and function in DCMEC remains unclear. Accordingly, the aim of the present study is to investigate the effects of high temperature on mitochondrial function by using DCMEC as an in vitro model.
Materials and Methods
Cell lines and culture
The DCMEC was provided by Prof. Gen-Lin Wang (Nanjing Agricultural University, Nanjing, China), and the separation of DCMEC was performed as described previously (Chen et al. 2019). In brief, the breast tissues were cut into 1.0 mm × 1.0 mm × 1.0 mm pieces and washed in PBS. Then, the tissue samples were treated with enzyme mixture (1.5 g/L type I collagenase, 1.5 g/L type II collagenase, and 1.5 g/L trypsin) (Sigma-Aldrich, St, Louis, MO; cat.: C0130 and C6885) and incubated in an oscillation incubator for 3 h (100 r/min). After filtration and centrifugation, the cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum (Gibco, Morgan Hill, CA;cat: 10438026) and 1% antibiotic-antimycotic solution in a humidified incubator with 5% CO2 at 37°C. The cells were cultured in DMEM supplemented with 10% fetal bovine serum and 1% antibiotic-antimycotic solution in a humidified incubator with 5% CO2 at 37°C. After 1 h, the DCMEC was selected based on means of differential attachment technique and identified with Cytokeratin 18 antibody staining (Abcam, Cambridge, MA;cat: ab52459, 1:300; Supplementary Figure 1).
Heat stress treatment
Heat stress treatment refers to previous studies. In briefly, DCMECs were seeded in a six-well plate and cultured for 24 h until they reached 80% confluence at 37°C in a 5% CO2 atmosphere, and the DCMECs were incubated at a high-temperature humidified incubator (42°C) for 2 h to induce heat stress (Collier et al. 2006) and then immediately for the next experiment.
Assessment of mitochondrial morphology
The DCMECs were seeded onto coverslips and incubated with MitoTracker Green (25 nM, Molecular Probes, Waltham, MA) in a cell culture incubator with 5% CO2 at 37°C. After 30-min incubation at 37°C, the cells were visualized using a confocal microscope, and mitochondrial morphology was quantified by ImageJ, according to previously reported methods (Merrill et al. 2017).
Measurement of mitochondrial membrane potential (JC-1)
The mitochondrial membrane potential of the DCMEC was measured using the JC-1 kit (Beyotime, Wuhan, China; cat.: c2006) according to the manufacturer’s instructions. Images were acquired using a fluorescence microscope (Olympus, Tokyo, Japan), and the fluorescence intensity was analyzed by ImageJ.
Measurement of the ATP content, Mn-SOD activity, and complex I activity
The DCMECs were homogenized, and the ATP content, Mn-SOD activity, and complex I activity of the supernatant were measured using an ATP kit (Beyotime, cat.: S0026), Mn-SOD kit (Beyotime, cat.: S0103), and complex I kit (Solarbio, Beijing, China; cat.: BC0515), respectively.
Total RNA extraction and qRT-PCR
The total RNA was extracted using TRIzol reagent (Invitrogen, Waltham, MA; cat.: 15596026). Reverse transcription was performed using PrimeScript RT Master Mix (TaKaRa, Shiga,Japan; cat.: RR036A), and mRNA expression was quantified using real-time PCR. The expression levels of all target genes were normalized to those of the endogenous reference gene β-actin using an optimized comparative Ct (2−ΔΔCt) value method, where ΔΔCt = ΔCttarget − ΔCtβ-actin. Primer sequences are listed in Supplementary Material Table S1.
Protein extraction and western blot analysis
To extract total proteins, cells were incubated with lysis buffer and centrifuged at 15,000×g for 15 min at 4°C. The mitochondrial and cytoplasmic protein fractions in supernatant were isolated using an extraction kit (Beyotime, cat.: c3601) according to the manufacturer’s instructions. The protein concentration was quantified using a BCA protein assay kit (Beyotime, cat.: P0010), and the western blot procedures were performed as previously described (Chen et al. 2018). The intensity of each band was normalized against that of the respective β-actin or VDAC loading control. The following commercially available antibodies were used: anti-cytochrome C (ProteinTech, Wuhan, China, cat.: 10993-1-AP, 1:3000), anti-Mn-SOD (ProteinTech, cat.: 24127-1-AP, 1:3000), anti-β-actin (ProteinTech, cat.: 10494-1-AP, 1:4000), anti-HSP70 (ProteinTech, cat.: 10995-1-AP, 1:4000), anti-Mfn1 (Abcam, cat.: ab104274, 1:2000), anti-Mfn2 (ProteinTech, cat.: 12186-1-AP, 1:2000), anti-Fis1 (ProteinTech, cat.: 10956-1-AP, 1:2000), anti-Drp1 (Abcam, cat.: ab154879, 1:2500).
Ca2+ concentration assay
The levels of calcium in the cell cytoplasm were measured by using a cell-permeable calcium-sensitive fluorescent dye (Fluo-3/AM, Solarbio, cat.: F8841). DCMECs were incubated with 5 μM Fluo-3/AM for 30 min at 37°C. Fluorescent images were acquired by using a fluorescence microscope (Olympus), and the fluorescence intensity of five randomly selected visual fields was analyzed using ImageJ.
Statistical analysis
Statistical analysis was performed using GraphPad Prism 8.0.1 (La Jolla, CA). Data were compared using one-way analysis of variance (ANOVA) with two-tailed unpaired t tests, and p values < 0.05 were considered significant.
Results
Effect of heat stress on mitochondrial morphology in DCMEC
The present study found that mitochondria exhibit dynamic networks in DCMEC and undergo dynamic fusion and fission processes (Fig. 1A). In contrast, after heat stress treatment, the mitochondria became more fragmented, which indicated that heat stress could affect mitochondrial function by disrupting mitochondrial morphology (Fig. 1B). Indeed, the quantification data confirmed that the average mitochondrial area, aspect ratio, and perimeter were significantly reduced after heat stress treatment (Fig. 1B, C).
Effect of heat stress on the mitochondrial morphology in DCMEC. A Representative time-lapse images of mitochondria undergoing dynamic fusion (upper panels) and fission (lower panels) in wild-type DCMEC. Green, mitochondria, scale bar 10 μm. B Representative morphology of mitochondria from 37°C- and 42°C-treated cells. Green, mitochondria; scale bar, 10 μm. C Quantification of mitochondrial morphology, based on the average area (C), aspect ratio, and perimeter of mitochondria from 37°C- and 42°C-treated cells. Values and error bars represent means ± SD. Two-tailed unpaired t test, *p < 0.05, **p < 0.001.
Effect of heat stress on mitochondrial respiratory chain activity and oxidative stress in DCMEC
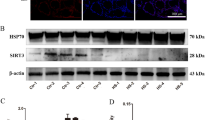
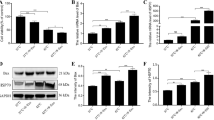
The change of mitochondrial morphology suggested that the mitochondrial function of DCMEC was disrupted under heat stress condition. In the meanwhile, the present study found that heat stress significantly reduced the ATP level (Fig. 2A) of DCMEC and significantly increased HSP70 expression (Fig. 2D–F). Complex I is the largest of the five enzyme supercomplexes in the mitochondrial electron transport chain. The present study found that heat stress significantly reduced complex I activity (Fig. 2B), as well as manganese superoxide dismutase (Mn-SOD) transcription, translation, and activity, thereby suggesting that heat stress causes defects in the mitochondrial electron transport chain (ETC) and oxidative phosphorylation system (OXPHOS; Fig. 2C–E). Interesting, heat stress also reduced the expression of PGC-1a, which indicated the disruption of mitochondrial biogenesis in DCMEC.
Effect of heat stress on the mitochondrial respiratory chain activity and oxidative stress in DCMEC. A–C Effect of heat stress on the ATP content (A), complex I activity (B), and Mn-SOD (C) activity of DCMEC. Values and error bars represent means ± SD. Two-tailed unpaired t test, *p < 0.05. D Effect of heat stress on the relative expression of HSP70, Mn-SOD, and PGC-1α. Values and error bars represent means ± SD. Two-tailed unpaired t test, **p < 0.001, ***p < 0.0001. E, F Effect of heat stress on the levels of HSP70 and Mn-SOD proteins. Values and error bars represent means ± SD. Two-tailed unpaired t test, *p < 0.05.
Effect of heat stress on mitochondrial membrane potential in DCMEC
Because the generation of mitochondrial membrane potential is essential to oxidative phosphorylation, JC-1 dye was used to determine the effect of heat stress on mitochondrial membrane potential. As shown in Fig. 3A, the mitochondria exhibited a heterogeneous pattern of fluorescence, and most of the mitochondria exhibited red fluorescence in DCMEC. However, most of the heat-stressed DCMEC mitochondria exhibited high levels of green fluorescence and low levels of red fluorescence, which indicated the formation of mitochondrial monomers and the reduced membrane potential. Indeed, the quantification data showed that the ratio of red/green was reduced after heat stress in DCMEC (Fig. 3B).
Effect of heat stress on the mitochondrial membrane potential in DCMEC. A, B JC-1 staining (A) and red/green ratio (B) of 37°C- and 42°C-treated dairy cow mammary epithelial cells. Red, aggregate; green, monomer. The high red/green ratio indicates high membrane potential in cells. Scale bar 100 μm. Values and error bars represent means ± SD. Two-tailed unpaired t test, ***p < 0.0001.
Effect of heat stress on the balance of mitochondrial fission and fusion in DCMEC
Because mitochondria continually change morphology through dynamic fission and fusion, the expression of Drp1 and Fis1, which are involved in mitochondrial fission, were examined to evaluate the mechanism of heat stress–induced mitochondrial fragmentation. The results showed that the expression of Drp1 and Fis1 were upregulated after heat stress treatment (Fig. 4A, C), which is consistent with the result that the mitochondria are fragmented in heat-stressed cells. In contrast, Mfn1 and Mfn2, which are important in the fusion of outer mitochondrial membranes, were significantly downregulated by heat stress, thereby suggesting that defects in Mfn1 and Mfn2 function can promote the fragmentation of mitochondria after heat stress (Fig. 4B, D). In addition, the negative effect of heat stress on the expression of Opa1 further demonstrated the conclusion that the occurrence of mitochondrial fusion defects is the main mechanism underlying the fragmentation of mitochondria in heat-stressed DCMEC (Fig. 4B).
Effect of heat stress on the mitochondrial fission-fusion balance in DCMEC. A, C Effect of heat stress on the relative mRNA and protein levels of Drp1 and Fis1. Values and error bars represent means ± SD. Two-tailed unpaired t test, **p < 0.001, ***p < 0.0001. B Effect of heat stress on the relative mRNA levels of Mfn1, Mfn2, and Opa1. Values and error bars represent means ± SD. Two-tailed unpaired t test, *p < 0.05, **p < 0.001, ***p < 0.0001. D Effect of heat stress on the relative level of Mfn1 and Mfn2 proteins. Values and error bars represent means ± SD. Two-tailed unpaired t test, *p < 0.05.
Effect of heat stress on mitochondrial calcium uptake in DCMEC
Previous studies have suggested that disruption of calcium homeostasis could induce mitochondrial fragmentation (Xu et al. 2013). To investigate whether the heat stress–induced mitochondrial fragmentation was associated with calcium homeostasis, DCMECs were incubated with Fluo-3/AM for 30 min after heat stress. The increased fluorescence intensity of Fluo-3/AM demonstrated that the calcium homeostasis was disrupted after heat stress treatment in DCMEC, and the quantification data further confirmed that heat stress increased Ca2+ concentration in DCMEC (Fig. 5A, B).
Effect of heat stress on the calcium uptake by the mitochondria in DCMEC. A, B Representative images of intracellular free Ca2+ (A) and quantification of Ca2+ concentrations (B) in 37°C- and 42°C-treated dairy cow mammary epithelial cells. Values and error bars represent means ± SD. Two-tailed unpaired t test, ***p < 0.0001.
Effect of heat stress on the release of cytochrome c from mitochondria in DCMEC
Cytochrome c is a component of the electron transport chain, which is involved in the initiation of apoptosis. It has been shown that inhibition of Drp1-mediated mitochondrial fission could prevent the release of cytochrome c and, thus, delay the apoptosis (Estaquier and Arnoult 2007). The present study found that heat stress significantly increased levels of cytochrome c in the cytoplasm, but not in the mitochondria, suggesting that heat stress induced the release of cytochrome c from mitochondria and, eventually, induced apoptosis (Fig. 6).
Discussion
The results of the present study provide mechanistic insight into the effects of heat stress on mitochondrial dysfunction in DCMEC. The results demonstrate that heat stress causes mitochondrial fragmentation by disrupting the balance of mitochondrial fission and fusion, thus contributing to mitochondrial disorders and dysfunction, which further induce the apoptosis of DCMEC and the dysfunctional of dairy mammary glands.
Mitochondria are dynamic organelles that constantly undergo fission and fusion processes, which are essential for mitochondrial inheritance and maintenance, when cells are subject to metabolic or environmental stresses (Westermann 2010). Recent studies have reported that the change of mitochondrial morphology is associated with disease occurrence. For example, the expression of Mfn1, Mfn2, and Opa1 promote the elongation of mitochondria, and further protect cells during embryonic development or nutrient starvation (Chen et al. 2003; Rambold et al. 2011). In contrast, expression of Drp1 promoted mitochondrial fragmentation, cell death, and apoptosis (Frank et al. 2001; Liot et al. 2009). However, it remains unclear whether mitochondrial morphology is associated with heat stress–induced mitochondrial dysfunction. The present study demonstrated that heat stress induces mitochondrial fragmentation, through mediating the expression levels of Mfn1/2, Opa1, Drp1, and Fis1. Previous studies have reported that mitochondrial fusion is important for maintaining healthy mitochondrial populations and that the process can ameliorate environment–induced stress by allowing functional mitochondria to complement dysfunctional mitochondria and sharing the components (Youle and van der Bliek 2012).
The destruction of dynamic mitochondrial networks is associated with mitochondrial dysfunction, including reductions in mitochondrial membrane potential and respiration and increases in the formation of mitochondrial reactive oxygen species (ROS) (Nagdas and Kashatus 2017; Jezek et al. 2018). Depletion of Mfn1 and Mfn2 altered mitochondrial metabolism along with the loss of mitochondrial membrane potential and reduction in endogenous respiration (Chen et al. 2005), which coincides with the present study’s observation that heat stress reduces complex I activity and mitochondrial membrane potential, along with Mfn1 and Mfn2 expression. Heat stress has also been reported to modulate the activity of anti-oxidant enzymes and heat stress response, which is characterized by reduced Mn-SOD activity and increased HSP70 expression, and that is coupled with increased ROS production (Slimen et al. 2014). Several studies have shown that increased ROS production is correlated with mitochondrial fission, suggesting that oxidative stress can induce mitochondrial fragmentation. However, when cells are under hyperglycemic conditions, mitochondria undergo Drp1-dependent fission, which further increases the release of ROS. Therefore, there is a reciprocal relationship between oxidative stress and mitochondrial morphology dynamics (Yu et al. 2006; Jendrach et al. 2008; Fan et al. 2010; Wu et al. 2011).
Calcium ions (Ca2+) function as a primary intracellular second messenger that directly or indirectly regulate a variety of cellular processes. In fact, it has been reported that heat stress induces large increases in intracellular Ca2+ concentrations (Han et al. 2010), which could potentially disrupt the balance between mitochondrial fission and fusion (Hom et al. 2010). Conditions that reduce cytosolic Ca2+ levels are sufficient to prevent mitochondrial fragmentation, and this could explain the result of the present study that heat stress–induced increases in cytoplasmic Ca2+ could contribute to mitochondrial fragmentation in DCMEC. However, the characterization of the mechanisms underlying this process will still need to be explored (Pinton et al. 2001).
The morphology of mitochondria is known to change during the early stages of apoptosis, resulting in small and fragmented mitochondria (Youle and Karbowski 2005). Mitochondrial fragmentation generally occurs at the same time that Bax, a pro-apoptotic member of the BCL-2 family, translocates from the cytoplasm to mitochondria, but before caspase activation (Frank et al. 2001; Capano and Crompton 2002; Karbowski et al. 2004). Inhibition of Drp1 or Fis1 before the induction of apoptosis blocks mitochondrial fission and delays the activation of caspase and apoptosis (Frank et al. 2001; Karbowski et al. 2002; Breckenridge et al. 2003; Lee et al. 2004). Downregulation of Mfn1/2 has been reported to promote the development of shorter mitochondria and to enhance apoptosis (Sugioka et al. 2004); meanwhile, depletion of Opa1 has been reported to induce mitochondrial fragmentation and the spontaneous release of cytochrome c (Olichon et al. 2003). The present study revealed that heat stress can induce the release of cytochrome c from mitochondria to the cytosol, thereby confirming that mitochondrial fusion and fission machinery participates in heat stress–induced apoptosis. Under stress conditions, Bax and Bak coalesce on the surface of the outer mitochondrial membrane (OMM) into large foci (Nechushtan et al. 2001; Valentijn et al. 2003) and that these foci co-localize with Mfn2 and Drp1 at mitochondrial fission sites (Karbowski et al. 2002), suggesting that Bax and Bak are involved in Drp1- or Mfn2-regulated mitochondrial fission during early apoptosis.
Conclusion
Taken together, the present study demonstrates that heat stress induces both oxidative stress and apoptosis was related with the mitochondrial fission- and fusion-regulated network (Fig. 7), thereby suggesting that mitochondrial network dynamics are important for physiological homeostasis and disease states. Accordingly, the results of the present study indicate that maintaining the normal balance of mitochondrial fission and fusion could help to attenuate the heat stress–induced oxidative stress and apoptosis in DCMEC.
References
Boutinaud M, Herve L, Lollivier V (2015) Mammary epithelial cells isolated from milk are a valuable, non-invasive source of mammary transcripts. Front Genet 6:323
Breckenridge DG, Stojanovic M, Marcellus RC, Shore GC (2003) Caspase cleavage product of BAP31 induces mitochondrial fission through endoplasmic reticulum calcium signals, enhancing cytochrome c release to the cytosol. J Cell Biol 160(7):1115–1127
Capano M, Crompton M (2002) Biphasic translocation of Bax to mitochondria. Biochem J 367(Pt 1):169–178
Chen H, Chomyn A, Chan DC (2005) Disruption of fusion results in mitochondrial heterogeneity and dysfunction. J Biol Chem 280(28):26185–26192
Chen H, Detmer SA, Ewald AJ, Griffin EE, Fraser SE, Chan DC (2003) Mitofusins Mfn1 and Mfn2 coordinately regulate mitochondrial fusion and are essential for embryonic development. J Cell Biol 160(2):189–200
Chen H, Vermulst M, Wang YE, Chomyn A, Prolla TA, McCaffery JM et al (2010) Mitochondrial fusion is required for mtDNA stability in skeletal muscle and tolerance of mtDNA mutations. Cell 141(2):280–289
Chen KL, Li L, Li CM, Wang YR, Yang FX, Kuang MQ et al (2019) SIRT7 regulates lipopolysaccharide-induced inflammatory injury by suppressing the NF-kappaB signaling pathway. Oxidative Med Cell Longev 2019:3187972
Chen KL, Li L, Yang FX, Li CM, Wang YR, Wang GL (2018) SIRT7 depletion inhibits cell proliferation, migration, and increases drug sensitivity by activating p38MAPK in breast cancer cells. J Cell Physiol 233(9):6767–6778
Collier RJ, Stiening CM, Pollard BC, VanBaale MJ, Baumgard LH, Gentry PC et al (2006) Use of gene expression microarrays for evaluating environmental stress tolerance at the cellular level in cattle. J Anim Sci 84(Suppl):E1–E13
Du J, Di H-S, Guo L, Li Z-H, Wang G-L (2008) Hyperthermia causes bovine mammary epithelial cell death by a mitochondrial-induced pathway. J Therm Biol 33(1):37–47
Estaquier J, Arnoult D (2007) Inhibiting Drp1-mediated mitochondrial fission selectively prevents the release of cytochrome c during apoptosis. Cell Death Differ 14(6):1086–1094
Fan X, Hussien R, Brooks GA (2010) H2O2-induced mitochondrial fragmentation in C2C12 myocytes. Free Radic Biol Med 49(11):1646–1654
Frank S, Gaume B, Bergmann-Leitner ES, Leitner WW, Robert EG, Catez F, Smith CL, Youle RJ (2001) The role of dynamin-related protein 1, a mediator of mitochondrial fission, in apoptosis. Dev Cell 1(4):515–525
Han AY, Zhang MH, Zuo XL, Zheng SS, Zhao CF, Feng JH, Cheng C (2010) Effect of acute heat stress on calcium concentration, proliferation, cell cycle, and interleukin-2 production in splenic lymphocytes from broiler chickens. Poult Sci 89(10):2063–2070
Herbut P, Angrecka S, Walczak J (2018) Environmental parameters to assessing of heat stress in dairy cattle-a review. Int J Biometeorol 62(12):2089–2097
Hom J, Yu T, Yoon Y, Porter G, Sheu SS (2010) Regulation of mitochondrial fission by intracellular Ca2+ in rat ventricular myocytes. Biochim Biophys Acta 1797(6–7):913–921
Hoppins S, Lackner L, Nunnari J (2007) The machines that divide and fuse mitochondria. Annu Rev Biochem 76:751–780
Jendrach M, Mai S, Pohl S, Voth M, Bereiter-Hahn J (2008) Short- and long-term alterations of mitochondrial morphology, dynamics and mtDNA after transient oxidative stress. Mitochondrion 8(4):293–304
Jezek J, Cooper KF, Strich R (2018) Reactive oxygen species and mitochondrial dynamics: the yin and yang of mitochondrial dysfunction and cancer progression. Antioxidants (Basel) 7(1)
Karbowski M, Arnoult D, Chen H, Chan DC, Smith CL, Youle RJ (2004) Quantitation of mitochondrial dynamics by photolabeling of individual organelles shows that mitochondrial fusion is blocked during the Bax activation phase of apoptosis. J Cell Biol 164(4):493–499
Karbowski M, Lee YJ, Gaume B, Jeong SY, Frank S, Nechushtan A et al (2002) Spatial and temporal association of Bax with mitochondrial fission sites, Drp1, and Mfn2 during apoptosis. J Cell Biol 159(6):931–938
Knott AB, Perkins G, Schwarzenbacher R, Bossy-Wetzel E (2008) Mitochondrial fragmentation in neurodegeneration. Nat Rev Neurosci 9(7):505–518
Lee YJ, Jeong SY, Karbowski M, Smith CL, Youle RJ (2004) Roles of the mammalian mitochondrial fission and fusion mediators Fis1, Drp1, and Opa1 in apoptosis. Mol Biol Cell 15(11):5001–5011
Liesa M, Shirihai OS (2013) Mitochondrial dynamics in the regulation of nutrient utilization and energy expenditure. Cell Metab 17(4):491–506
Lindsay, S.J. (2004). Hand-held pneumatic impact handpiece for delicate, e.g. hand engraving operation, of jewelry, has variable fuel meter and work energy supply which regulate supply of pressurized fluid and of work energy based on pressure sensor output. US patent application 10/267630. 17 Feb 2004
Liot G, Bossy B, Lubitz S, Kushnareva Y, Sejbuk N, Bossy-Wetzel E (2009) Complex II inhibition by 3-NP causes mitochondrial fragmentation and neuronal cell death via an NMDA- and ROS-dependent pathway. Cell Death Differ 16(6):899–909
Liu W, Yuan Y, Sun C, Balasubramanian B, Zhao Z, An L (2019) Effects of dietary betaine on growth performance, digestive function, carcass traits, and meat quality in indigenous yellow-feathered broilers under long-term heat stress. Animals (Basel) 9(8)
Merrill, R.A., Flippo, K.H., and Strack, S. (2017). Measuring mitochondrial shape with ImageJ. 123, 31–48
Nagdas S, Kashatus DF (2017) The interplay between oncogenic signaling networks and mitochondrial dynamics. Antioxidants (Basel) 6(2)
Nechushtan A, Smith CL, Lamensdorf I, Yoon SH, Youle RJ (2001) Bax and Bak coalesce into novel mitochondria-associated clusters during apoptosis. J Cell Biol 153(6):1265–1276
Olichon A, Baricault L, Gas N, Guillou E, Valette A, Belenguer P, Lenaers G (2003) Loss of OPA1 perturbates the mitochondrial inner membrane structure and integrity, leading to cytochrome c release and apoptosis. J Biol Chem 278(10):7743–7746
Pinton P, Ferrari D, Rapizzi E, Di Virgilio F, Pozzan T, Rizzuto R (2001) The Ca2+ concentration of the endoplasmic reticulum is a key determinant of ceramide-induced apoptosis: significance for the molecular mechanism of Bcl-2 action. EMBO J 20(11):2690–2701
Polsky L, von Keyserlingk MAG (2017) Invited review: effects of heat stress on dairy cattle welfare. J Dairy Sci 100(11):8645–8657
Qian L, Song X, Ren H, Gong J, Cheng S (2004) Mitochondrial mechanism of heat stress-induced injury in rat cardiomyocyte. Cell Stress Chaperones 9(3):281–293
Rambold AS, Kostelecky B, Elia N, Lippincott-Schwartz J (2011) Tubular network formation protects mitochondria from autophagosomal degradation during nutrient starvation. Proc Natl Acad Sci U S A 108(25):10190–10195
Scott I, Youle RJ (2010) Mitochondrial fission and fusion. Essays Biochem 47:85–98
Slimen, I.B., Najar, T., Ghram, A., Dabbebi, H., Ben Mrad, M., and Abdrabbah, M. (2014). Reactive oxygen species, heat stress and oxidative-induced mitochondrial damage. A review. Int J Hyperthermia 30(7), 513–523
Sugioka R, Shimizu S, Tsujimoto Y (2004) Fzo1, a protein involved in mitochondrial fusion, inhibits apoptosis. J Biol Chem 279(50):52726–52734
Takahashi M (2012) Heat stress on reproductive function and fertility in mammals. Reprod Med Biol 11(1):37–47
Tao S, Orellana RM, Weng X, Marins TN, Dahl GE, Bernard JK (2018) Symposium review: the influences of heat stress on bovine mammary gland function. J Dairy Sci 101(6):5642–5654
Valentijn AJ, Metcalfe AD, Kott J, Streuli CH, Gilmore AP (2003) Spatial and temporal changes in Bax subcellular localization during anoikis. J Cell Biol 162(4):599–612
Westermann B (2010) Mitochondrial fusion and fission in cell life and death. Nat Rev Mol Cell Biol 11(12):872–884
Wohlgemuth SE, Ramirez-Lee Y, Tao S, Monteiro APA, Ahmed BM, Dahl GE (2016) Short communication: effect of heat stress on markers of autophagy in the mammary gland during the dry period. J Dairy Sci 99(6):4875–4880
Wu S, Zhou F, Zhang Z, Xing D (2011) Mitochondrial oxidative stress causes mitochondrial fragmentation via differential modulation of mitochondrial fission-fusion proteins. FEBS J 278(6):941–954
Xu S, Pi H, Chen Y, Zhang N, Guo P, Lu Y et al (2013) Cadmium induced Drp1-dependent mitochondrial fragmentation by disturbing calcium homeostasis in its hepatotoxicity. Cell Death Dis 4:e540
Yoon Y, Krueger EW, Oswald BJ, McNiven MA (2003) The mitochondrial protein hFis1 regulates mitochondrial fission in mammalian cells through an interaction with the dynamin-like protein DLP1. Mol Cell Biol 23(15):5409–5420
Youle RJ, Karbowski M (2005) Mitochondrial fission in apoptosis. Nat Rev Mol Cell Biol 6(8):657–663
Youle RJ, van der Bliek AM (2012) Mitochondrial fission, fusion, and stress. Science 337(6098):1062–1065
Yu T, Robotham JL, Yoon Y (2006) Increased production of reactive oxygen species in hyperglycemic conditions requires dynamic change of mitochondrial morphology. Proc Natl Acad Sci U S A 103(8):2653–2658
Funding
This work was supported by the China Postdoctoral Science Foundation (grant number 2019M651755), the Natural Science Foundation of Jiangsu Province (grant number BK20190254), and the National Natural Science Foundation of China (grant number 31372290).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Editor: Tetsuji Okamoto
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Chen, KL., Wang, HL., Jiang, LZ. et al. Heat stress induces apoptosis through disruption of dynamic mitochondrial networks in dairy cow mammary epithelial cells. In Vitro Cell.Dev.Biol.-Animal 56, 322–331 (2020). https://doi.org/10.1007/s11626-020-00446-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-020-00446-5