Abstract
We analyzed the physiological role of forkhead box class O 1 (Foxo1) in adipocyte differentiation by suppressing Foxo1 mRNA with siRNA specific for Foxo1. Mouse 3T3-L1 preadipocytes infected with an adenovirus expressing Foxo1-siRNA showed a marked decrease in lipid droplet formation when induced to differentiate into adipocytes. Adipocyte differentiation was most severely inhibited by exposing cells to Foxo1-siRNA before induction of differentiation. The incorporation of fluorescent-labeled glucose and fatty acid was significantly inhibited in cells deficient in Foxo1. RT-PCR revealed that downregulation of Foxo1 decreased the expression of the transcription factors, PPAR-γ and C/EBP-α. By comparison, Foxo1-siRNA did not affect the expression of C/EBP-β or C/EBP-δ during the early period of adipocyte differentiation. These results indicate that Foxo1 plays an essential role in adipocyte differentiation, especially at the very early stage of terminal adipocyte differentiation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Adipose tissue functions as a storage organ to save surplus energy and also as a secretory organ of various bioactive substances. Adipocytes that constitute the adipose tissue secrete cytokines known as adipocytokines. Adipocytokines act on various organs to maintain homeostasis and in obesity play a role in life-style-related diseases, including diabetes mellitus, hyperlipemia, and hypertension (Rosen and Spiegelman 2006).
The 3T3-L1 mouse preadipocyte cell line has the capacity to differentiate into adipocytes when treated with a mixture of dexamethasone, 3-iso-butyl-1-methylxanthine, and insulin (DMI). After initiation of differentiation, 3T3-L1 cells pass through a transient cell proliferation stage, known as clonal expansion, before acquiring the functions of a mature adipocyte, including lipid droplet accumulation and the secretion of adipocytokines. Previous studies reveal that many transcription factors are associated with adipocyte differentiation. The ligand-dependent nuclear receptor type transcription factor PPAR-γ (Rosen et al. 1999) and the leucine zipper type transcription factor C/EBP family (Cao et al. 1991) play critical roles in primary adipocyte differentiation. The expression level of C/EBP-β, a member of the C/EBP family, rises 2–4 h after the induction of differentiation and gradually accumulates, then after about 14 h C/EBP-β becomes activated by acquiring DNA binding activity and contributes to the progression of clonal expansion (Tang and Lane 1999). Once clonal expansion is complete, C/EBP-β and C/EBP-δ induce the expression of C/EBP-α and PPAR-γ, and the differentiating adipocyte progresses into the terminal differentiation phase (Wu et al. 1996). C/EBP-α and PPAR-γ induce adipocyte-specific gene expression and mutual expression, and this leads to the development of the mature adipocyte with insulin sensitivity and the capacity to form lipid droplets.
The forkhead-type transcription factor family forkhead box class O (Foxo) is also thought to play a role in adipocyte differentiation (Nakae et al. 2003). The known Foxo subfamily consists of Foxo1, Foxo3a, Foxo4, and Foxo6 (Lam et al. 2006; Reagan-Shaw and Ahmad 2007). Foxo proteins have highly conservative phosphorylation sites (Thr-24, Ser-256, and Ser-319 in human FOXO1) and are phosphorylated by Akt and SGK through the insulin-dependent PI3K/Akt cascade (Czech 2003). Phosphorylated Foxo is transported from the nucleus to the cytosol resulting in its inactivation (Brunet et al. 1999). The target genes of Foxo are diverse, and Foxo contributes to various physiological phenomena, including apoptosis (Li et al. 2003), DNA repair (Tran et al. 2002), cell cycle arrest (Schmidt et al. 2002), carbohydrate metabolism (Nakae et al. 2001), fat metabolism (Kamei et al. 2003), differentiation (Hribal et al. 2003), and cellular protection from oxidative stress (Brunet et al. 2004).
The over-expression of activated Foxo1 in adipocyte progenitor cells induces the expression of the cell cycle arrest-associated factor p21and the C/EBP inhibitor CHOP10; these disturb the progression of clonal expansion, thereby inhibiting adipocyte differentiation (Nakae et al. 2003). Details of the physiological role of Foxo1 in adipocyte differentiation and the molecular mechanisms underlying this process are not known. By using Foxo1-specific siRNA, the present study demonstrates that Foxo1 is essential for adipocyte differentiation.
Materials and Methods
Materials.
The 3T3-L1 cell line was purchased from the Health Science Research Resources Bank (HSRRB, Osaka, Japan). HEK 293 and COS-7 cells were obtained from the cell bank of the RIKEN Institute of Physical and Chemical Research (Tsukuba, Ibaraki, Japan). The plasmid pcDNA3-Foxo1 was a gift from Dr. M Greenberg (Harvard Medical School, Boston, MA). The plasmids pEF-BOS-C/EBP-α, pEF-BOS-C/EBP-β, and pEF-BOS-C/EBP-δ were kindly provided by Dr. Takiguchi (Chiba University, Chiba, Japan).
Cell culture.
The 3T3-L1 cell line was maintained at 37°C in a humidified, CO2 incubator. The standard culture media was Dulbecco’s modified Eagle’s medium (DMEM, high-glucose; Sigma-Aldrich, St. Louis, MO) supplemented with 10% FBS (Sanko Junyaku, Chiyoda, Tokyo, Japan). We induced the differentiation of 3T3-L1 preadipocytes into adipocytes as previously described (Student et al. 1980); briefly, 3T3-L1 cells were cultured for 2 d to confluence then for a further 2 d in media supplemented with 0.25 μM dexamethasone (Sigma-Aldrich), 0.5 mM 3-iso-butyl-1-methylxanthine (Sigma-Aldrich), and 10 μg/ml insulin (Wako, Chuo, Osaka, Japan; DMI induction). Afterward, cells were cultured for 2 d in media containing 5 μg/ml insulin and then for 4 d in standard culture media. The HEK 293 and COS-7 cells were maintained in DMEM with 10% FBS.
Oil red O staining.
Cells were fixed with 4% paraformaldehyde for 1 h and then stained with 3 mg/ml Oil red O (in 60% isopropanol) for 10 min after washing cells with PBS. Stained cells were washed with sterile water then observed under a microscope (DMIRBE M2FLIII (Leica Microsystems Inc., Bannockburn, IL)). In addition, cells were stained with 0.3 mg/ml Oil red O, and the dye was then eluted for 10 min with 100% isopropanol. The concentration of the eluted dye was determined from measurements of absorbance (O.D. 420 nm).
Triglyceride assay.
Cells were lysed in lysis buffer (20 mM HEPES [pH 7.6], 420 mM NaCl, 1% triton X-100, 0.1% SDS), and total fat was extracted according to the Bligh and Dyer method (Bligh and Dyer 1959). The cell extract (600 μl) was incubated with 2 ml methanol and 1 ml chloroform for 1 h, and then 1 ml of chloroform and 1 ml of sterile water were added. The chloroform phase of the cell extract was collected by brief centrifugation, dried overnight, then dissolved in 10% triton-isopropanol solution. The triglyceride level was measured according to the method described in the manual of the Triglyceride E-test Kit (Wako).
RT-PCR.
The 3T3-L1 cells were cultured for 0, 2, 4, and 6 d after DMI induction, and RNA was extracted using the GTC-acid phenol method. RNA was incubated with 5 units of DNase (Takara, Otsu, Shiga, Japan) for 30 min and then treated twice with phenol/chloroform. After cDNA synthesis using M-MLV reverse transcriptase (Takara), PCR was performed using the primers indicated in Table 1 under optimal amplification conditions (cycle number and annealing temperature) for each gene.
Western blotting.
Cells were resuspended in sample buffer (50 mM Tris–HCl [pH 6.8], 2% SDS, 6% β-mercaptoethanol, 10% glycerol) containing protease inhibitors (1 mM AEBSF, 130 μM Bestatin, 14 μM E-64, 1 mM EDTA, 1 μM pepstatin A). The cells were then treated with a supersonicator (UP-50H, B. Braun Biotech International GmbH, Melsungen, Germany) for 2 min, and the cell extract was collected after centrifugation (4°C, 15,000×g, 10 min). The protein concentration was quantified using the BCA protein assay kit (Pierce, Rockford, IL). SDS-PAGE was performed with 5∼50 μg of protein, and the separated proteins were transferred to a PVDF membrane (Schleicher & Schuell BioScience, Keene, NH). Non-specific binding was blocked by soaking the membrane in 5% skim milk. The membrane was then incubated with the primary antibody overnight followed by incubation with the secondary antibody for 1 h and detection of specific binding using the lumiGLO reagent (Cell Signaling, Danvers, MA). Finally, each protein band was detected by chemiluminescence (Las1000, FujiFILM, Minato, Tokyo, Japan) and analyzed with Image Gauge Software (FUJI-FILM). The primary antibodies included anti-Foxo1 (Cell Signaling), anti-PPARγ (Santa Cruz Biotechnology Inc., Santa Cruz, CA), anti-C/EBPβ (Cell Signaling), anti-C/EBPδ (Cell Signaling), anti-C/EBPα (Cell Signaling), anti-β-actin (Sigma), and horseradish peroxidase-linked-anti-IgG (Cell Signaling).
Real-time PCR.
PCR was performed using SYBR Premix Ex Taq (Takara) on the Thermal Cycler Dice (Takara). Each cDNA was amplified (95°C for 5 s, 60°C for 30 s, 72°C for 30 s, for 40 cycles) using primers listed in Table 2. β-Actin was used as an internal standard for correction of the error determined between each sample. The PCR amplification of each cDNA was performed independently using three samples in triplicate.
Glucose transport.
The glucose transport capacity of 3T3-L1 cells was analyzed as follows. The cells were induced to differentiate by treatment with DMI; after 8 d, the cells were washed with PBS and treated with KRP-H buffer (131.2 mM NaCl, 4.7 mM KCl, 2.5 mM NaH2PO4, 2.5 mM CaCl2, 1 mM MgSO4, 1 mM HEPES (pH 7.4) containing 10 μM 2-NBDG (2-(N-(7-nitrobenz-2-oxa-1, 3-diazol-4-yl) amino)-2-deoxyglucose; Invitrogen Life Technologies, Carlsbad, CA) at 37°C for 5 min. After washing the cells with KRP-H buffer, cells were observed under a LEITZ DMRXA HC RXA-6 fluorescence microscope (Leica).
Fatty acid transport.
The fatty acid transport capacity of 3T3-L1 cells was analyzed as follows. The cells were induced to differentiate by treatment with DMI, and after 8 d, the cells were washed with PBS and treated with PBS (Ca2+/Mg2+) containing 20 μM BODIPY3823 (Invitrogen) and 20 μM BSA (fatty acid free) at 37°C for 2 min. After washing the cells twice with PBS containing 20 μM BSA, cells were observed under a LEITZ DMRXA HC RXA-6 fluorescence microscope (Leica).
Plasmid construction.
Plasmid pSilencer 2.1-U6 Hygro for siRNA was purchased from Ambion (St. Austin, TX). The following oligonucleotides were synthesized: mouse Foxo1-sense (1335∼1353): GATCCGACTTTGATAACTGGAGTACTGTGAAGCCACA-GATGGGTACTCCAGTTATCAAAGTCTTTTTTA, mouse FOXO1-anti-sense: AGCTTAAAAAA-GACTTTGATAACTGGAGTACCCATCTGTGGCTTCACAGTACTCCAGTTATCAAAGTCG, mouse FOXO3a-sense (2292∼2310): GATCCACTTGGACCTGGACATGTTCTGTGAAGCCAC-AGATGGGAACATGTCCAGGTCCAAGTTTTTTTA, mouse FOXO3a-anti-sense: AGCTTAAAA-AAACTTGGACCTGGACATGTTCCCATCTGTGGCTTCACAGAACATGTCCAGGTCCAAGTG.
Each oligonucleotide pair was annealed and subcloned into pSilencer-BamH1/HindIII (pSilencer-Foxo1Si, pSilencer-Foxo3aSi). A randomly arranged sequence (Ambion) was inserted into the same vector as a negative control for the siRNA vectors (pSilencer-NCSi).
Construction of adenovirus.
The siRNA expressing adenovirus vectors were constructed using the cosmid pAxcwit (Takara) and the pSilencer plasmids described above (pSilencer-Foxo1Si, pSilencer-Foxo3aSi, and pSilencer-NCSi). The pSilencer plasmids were linearized by digestion with ScaI, and full length plasmid DNA was cloned into the SmiI site of pAxcwit. The recombination adenovirus was made according to the procedure described by Takara. The pAxcwit/pSilencer clones were digested with BspT104 and transfected into HEK 293 cells using Lipofectamine™ 2000 (Invitrogen), resulting in the production of recombinant adenovirus Ad-Foxo1Si, Ad-Foxo3aSi, and Ad-NCSi. Adenovirus was amplified according to the procedure described by Takara. Virus-infected cells were cultured on a 96-well plate, then collected and subjected to six repeated freeze/thaw cycles to generate the primary virus solution. The primary virus solution was used to infected confluent cultures of HEK 293 cells on a 24-well plate. The secondary virus liquid was prepared as described above. Amplification of the virus was repeated until the fourth preparation of virus liquid was obtained. The titer was measured according to the TCID 50 method (Tissue Culture Infectious Dose).
Infection of 3T3-L1 cells with adenovirus.
Adenovirus solution at 3 × 108 pfu was diluted in 500 μl DMEM, mixed with polylysine (1 μg/ml), and left for 30 min. The adenovirus mixture was added to a confluent culture of 3T3-L1 cells on a 6-cm dish, incubated at 37°C for 1 h, then cultured for a further 2 d in 2.5 ml DMEM with 10% FBS.
Luciferase assay.
The pGL3-Basic (Promega, Madison, WI) reporter vector was used for luciferase assays. The sense oligonucleotide (AATAGATCTTTACTGCAATTTTAAAAAGCAATC-AATAAAGCTTAA) and anti-sense oligonucleotide (ATTAAGCTTTATTGATTGCTTTTTAAAAT-TGCAGTAAAGATCTATT) for the C/EBP responsive sequence, located in the promoter region of PPAR-γ, were synthesized and subcloned into the BglII–HindIII site of pGL3 (pGL3-C/EBP-RE). COS cells were seeded onto 4-cm-diameter plates at 8 × 104 cells/plate and 24 h later were transfected with 0.75 μg reporter plasmid (pGL3-C/EBP-RE) and 0.1 μg of each expression plasmid (pEF-BOS-C/EBP-α, pEF-BOS-C/EBP-β, pEF-BOS-C/EBP-δ, and pcDNA3-Foxo1) using 2.5 μl lipofectamine 2000 (Invitrogen) according to the manufacturer’s instructions. Luciferase activity was measured using the Luciferase Assay System (Promega). Co-transfection with the β-galactosidase expression plasmid and determination of β-galactosidase activity was used to reassess luciferase activity to correct for variation in transfection efficiency. The experiment was performed independently with three samples in triplicate.
Statistical analysis.
Results are expressed as mean ± standard deviation. Comparisons between groups were made by analysis of variance and, when significant, were examined by Tukey’s all-pairwise comparison test. Differences were considered significant when P < 0.05.
Results
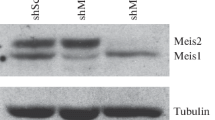
The differentiation of 3T3-L1 cells into adipocytes was induced by treating cells with DMI. RT-PCR revealed that expression of Foxo1 rose 2 d after DMI induction and increased gradually as differentiation progressed (Fig. 1A ). Under the same conditions, the level of Foxo3a expression was approximately constant throughout adipocyte differentiation. At the protein level, the expression of Foxo1 rose at days 2 and 4 after DMI induction, and it decreased a little by day 6; the observed peak level was on day 4 when the clonal expansion was completed (Fig. 1B ). By comparison, the protein level of Foxo3a was low before differentiation, increased as differentiation progressed, then was maintained at approximately constant level once clonal expansion was completed. We investigated the role of Foxo1 and Foxo3a in adipocyte differentiation by using siRNA specific to Foxo1 and Foxo3a to deplete the expression of these proteins in 3T3-L1 cells. We constructed siRNA-expression plasmids specific for Foxo1 and Foxo3a (pSilencer-Foxo1Si and pSilencer-Foxo3aSi) and incorporated these into adenovirus vectors (Ad-Foxo1Si and Ad-Foxo3aSi). The 3T3-L1 cells were infected with Ad-Foxo1Si or Ad-Foxo3aSi, and the level of Foxo1 and Foxo3a mRNA was analyzed by RT-PCR. We show marked suppression of Foxo1 and Foxo3a mRNA in infected 3T3-L1 cells (Fig. 1C ).

Figure 1. The RNA and protein were prepared on days 0, 2, 4 and 6 after DMI induction to analyze the expression of Foxo1 and Foxo3a by RT-PCR (A) and Western blot analysis (B). The mRNA expression of Foxo1 and Foxo3a was inhibited by introducing specific siRNA into the 3T3-L1 cells (C). Ad-NCSi (Ambion), which generates dsRNA of randomly arranged sequence, was used as a negative control for siRNA. GAPDH was used as an internal control for the level of RNA, and β-actin was used as a loading control for protein.
Four days before DMI induction, 3T3-L1 cells were infected with Ad-Foxo1Si or Ad-Foxo3aSi. The cells were stained with Oil red O 8 d after DMI induction to assess lipid accumulation. The siRNA-induced knockdown of Foxo1 expression significantly decreased lipid droplet accumulation (Figs. 2A , B). By comparison, knockdown of Foxo3a expression did not affect the accumulation of lipids or cellular form and size. We also analyzed the cumulative level of triglycerides during adipocyte differentiation of Foxo1-depleted 3T3-L1 cells: siRNS-induced suppression of Foxo1 in 3T3-L1 cells reduced the cumulative level of triglyceride by 30% compared to cells infected with the adenovirus expressing the negative control siRNA or the siRNA specific for Foxo3a (Fig. 2C ).

Figure 2. (A) The physiological effect on adipogenesis by suppression of Foxo mRNA was analyzed. The 3T3-L1 cells were infected with Ad-Foxo1Si, Ad-Foxo3aSi, or Ad-NCSi and then subjected to DMI induction. At day 8 after DMI induction, cells were dyed with Oil red O. (B) The concentration of the eluted dye was determined by measuring the absorbance at O.D. 420 nm. The bars on the graph indicate control NCSi (white), Foxo1Si (black), and Foxo3aSi (gray). An error bar shows standard deviation of the mean, n = 3. The experiments were performed independently in triple. Against a value of Ad-NCSi, **P < 0.01. (C) For the triglyceride assay, total lipid was extracted from the cells at day 8 after DMI induction. Against a value of Ad-NCSi, *P < 0.05.
The importance of Foxo1 expression in the terminal differentiation of adipocytes was investigated by examining the influence of siRNA-induced Foxo1 depletion on the incorporation of glucose and fatty acids. In these studies, we exposed Ad-Foxo1Si-, Ad-Foxo3aSi-, and Ad-NCSi-infected 3T3-L1 cells to fluorescent-labeled glucose (2-NBDG (2-(N-(7-nitrobenz-2-oxa-1, 3-diazol-4-yl) amino)-2-deoxyglucose)) 8 d after DMI induction. Observation with the fluorescence microscope revealed that glucose incorporation was inhibited markedly by the suppression of Foxo1 expression (Fig. 3A ); in contrast, the knockdown of Foxo3a did not affect glucose incorporation noticeably. In similar experiments, 3T3-L1 cells were exposed for 2 min to the fluorescent-labeled fatty acid analog, BODIPY3823, 8 d after DMI induction. Examination of fatty acids reveals that siRNA-induced suppression of Foxo1 expression remarkably reduced the incorporation of fatty acids compared to either the negative control or when FOXO3a expression was suppressed (Fig. 3B ).

Figure 3. Effect of Foxo1- and Foxo3a-siRNA on the uptake of glucose and fatty acid. The 3T3-L1 cells were infected with adenovirus (a, b Ad-Foxo1Si; c, d Ad-Foxo3aSi; or e, f Ad-NCSi) then subjected to DMI induction. (A) Glucose uptake was then analyzed by treating cells with fluorescent-labeled glucose 2-NBDG (10 μM) 8 d after DMI induction. (B) Fatty acid metabolism was analyzed by measuring fatty acid incorporation in virus-infected cells treated with 20 μM of the fluorescent-labeled fatty acid analog, BODIPY3823. The bright field (a, c, e) and fluorescent field (b, d, f) are indicated.
We then examined the effect of Foxo1 depletion on the expression of genes involved in adipocyte differentiation. Ad-Foxo1Si- and Ad-NCSi-infected 3T3-L1 cells were treated with DMI 4 d after infection. Protein was extracted from the cells at 0, 2, 4, and 6 d after DMI induction and subjected to Western blot analysis for detection of PPAR (-γ1 and -γ2) and C/EBP (-α, -β, and -δ) expression (Fig. 4A ). siRNA-induced suppression of Foxo1 expression significantly decreased the expression of PPAR-γ and C/EBP-α. By comparison, knockdown of Foxo1 expression had no notable effect on the expression of C/EBP-β or C/EBP-δ.

Figure 4. Effect of Foxo1-siRNA on gene expression. The 3T3-L1 preadipocytes were infected with adenovirus (Ad-Foxo1Si or Ad-NCSi) then subjected to DMI induction. (A) The protein levels of PPAR (-γ1 and -γ2) and C/EBP (-α, -β, and -δ) were analyzed. Total protein was extracted from cells at 0, 2, 4, and 6 d after DMI induction, then subjected to Western blot analysis. (B) Total RNA was prepared from cells at 0, 2, 4, and 6 d after DMI induction and subjected to RT-PCR for the following genes; PPAR-γ, Adipsin, CD36, Glut4, FATP4, C/EBP-δ, and CHOP10. (C) Total RNA was prepared from cells at 0, 2, 4, and 6 d after DMI induction and subjected to real-time PCR for the following genes; PPAR-γ, C/EBP-α, Glut4, and CD36. Ad-NCSi (closed circle) and Ad-Foxo1Si (closed triangle) are indicated.
Consistent with our findings by Western blot analysis, RT-PCR and real-time PCR analyses demonstrate that siRNA-induced Foxo1 depletion remarkably decreased the expression of PPAR-γ (Fig. 4). In addition, expression of Glut4, which is controlled by PPAR-γ, was significantly reduced by siRNA-induced Foxo1 depletion (Figs. 4B , C). Under the same conditions, the expression of Adipsin and CD36 was also reduced (Figs. 4B , C). The expression of C/EBP-δ remained unchanged (Fig. 4B ), whereas the expression of C/EBP-α was significantly reduced (Figs. 4A , C). In addition, the expression of CHOP10, an inhibitor of C/EBP, did not vary with knockdown of Foxo1 expression.
Next, we investigated whether Foxo1 directly affects the expression of PPAR-γ. Foxo1 is known to cooperate with C/EBP (Christian et al. 2002). It is possible that the presence of Foxo1 affects the transcriptional regulation of PPAR-γ by C/EBP. To test this possibility, we performed luciferase reporter assays by constructing a luciferase reporter vector harboring the C/EBP binding site (Zhu et al. 1995) from the PPAR-γ promoter region (Fig. 5A ). The role of Foxo1 in the transcriptional regulation of C/EBP was analyzed by over-expressing Foxo1 and C/EBP in COS7 cells. The transcriptional regulation of PPAR-γ was stimulated by over-expression of C/EBP-α and C/EBP-δ, whereas the over-expression of Foxo1 had no effect on PPAR-γ expression (Fig. 5A ). By comparison, C/EBP-β had no effect on the expression of PPAR-γ in the absence of DEX stimulation (Wiper-Bergeron et al. 2003). These data suggest that Foxo1 does not affect the expression of PPAR-γ by interacting with a C/EBP binding site in the PPAR-γ promoter. Knockdown of Foxo1 markedly reduced the expression of PPAR-γ and C/EBP-α, and it gave no notable effect on the expression of C/EBP-β or C/EBP-δ (Fig. 4). A similar result was reported by controlling an insulin signal cascade (Kim and Chen 2004; Naiki et al. 2007). Foxo1 is involved in the part of regulation of insulin signal cascade (Naïmi et al. 2007); thus, influence of Foxo1-RNAi to the expression of insulin signal-related gene was examined. RT-PCR revealed that knockdown of Foxo1 mRNA reduced the expression of IRS2, whereas it had no effect to that of TRB3 and mTOR (Fig. 5B ).

Figure 5. Role of Foxo1 on adipocyte differentiation. (A) COS7 cells were co-transfected with 0.1 μg each of the expression plasmids pEF-BOS-C/EBP (-α, -β, or -δ) and pcDNA3-Foxo1 together with 0.75 μg of the reporter plasmid pGL3-C/EBP-RE. The luciferase assay was performed 48 h after transfection to analyze the transcriptional activity of C/EBP in the presence (black bar) or absence (white bar) of Foxo1. The experiments were performed on two samples independently and in triplicate. An error bar shows standard deviation of the mean, n = 6. (B) The 3T3-L1 preadipocytes were infected with adenovirus (Ad-Foxo1Si or Ad-NCSi) before DMI induction. RNA was prepared from cells at 0, 2, 4, and 6 d after DMI induction, then subjected to RT-PCR for insulin signal-related genes.
Discussion
The expression level of Foxo1 reached a maximum at day 4 of differentiation, suggesting that Foxo1 plays an important role during the early stage of terminal differentiation that follows clonal expansion, not during the start of differentiation (Fig. 1). This conclusion is also supported by our findings that depletion of Foxo1 by Foxo1-siRNA has no effect on the expression of C/EBP-β and C/EBP-δ, which are essential for clonal expansion (Tang and Lane 1999), and that depletion of Foxo1 has a significant effect on the expression of PPAR-γ and C/EBP-α (Wu et al. 1999), which are essential for terminal differentiation at day 4 after DMI induction (Fig. 4). Together, these results indicate that Foxo1 plays an important role during adipocyte differentiation, at least by day 4. The siRNA-induced inhibition of Foxo1 expression inhibits terminal differentiation and simultaneously suppresses the expression of Glut4 via PPAR-γ and C/EBP-α (Fig. 4), resulting in the suppression of glucose and fatty acid incorporation (Fig. 3). From these results, it is clear that Foxo1 does not function from the start of differentiation through the first day of differentiation, and is essential after clonal expansion is complete. Foxo1 appears to play a role as a mediator to induce the transition from clonal expansion to terminal differentiation.
The knockdown of Foxo1 expression by Foxo-siRNA significantly suppressed the expression of C/EBP-α and PPAR-γ, but had no effect on the expression of C/EBP-β and C/EBP-δ. Similarly, the suppression of insulin signaling by the over-expression of TRB (Naiki et al. 2007) or the mTOR inhibitor (Kim and Chen 2004) coincides with suppression of C/EBP-α and PPAR-γ expression but has no influence on the expression C/EBP-β or C/EBP-δ. So, the effect of Foxo against the expression of the insulin signal-related gene was analyzed, and it revealed that the expression of IRS2 was suppressed by Foxo1-RNAi (Fig. 5B ). Because IRS2 is known to effect the differentiation of the adipocyte (Miki et al. 2001), the repression of the differentiation due to the Foxo1-RNAi might be partially caused by IRS2.
Our finding that knockdown of Foxo1 expression inhibits adipocyte differentiation is not consistent with the report that over-expression of the activated form of Foxo1 inhibits adipocyte differentiation (Nakae et al. 2003). Other studies investigating the effect of over-expression or knockdown of expression on adipocyte differentiation are also contradictory. For example, the over-expression of ERK suppresses adipocyte differentiation via the phosphorylation of PPAR-γ (Hu et al. 1996), whereas the functional obstruction of ERK suppresses adipocyte clonal expansion and differentiation (Tang et al. 2003). Furthermore, dephosphorylated-RB suppresses the transcriptional activity of PPAR-γ through binding with PPAR-γ and HDAC3 (Fajas et al. 2002a, b), and knockdown of RB expression fails to induce adipocyte differentiation (Classon et al. 2000). It is generally accepted that the effect of RB phosphorylation depends on the stage of differentiation of the adipocytes and that RB is specifically phosphorylated from clonal expansion to the beginning of terminal differentiation. Together, these findings suggest that many regulatory factors are essential for adipocyte differentiation and require tight temporal control for the correct progression of the differentiation process. Hence, it is possible that regulatory factors have the reverse effect if expressed at an improper stage of adipocyte differentiation. Although a previous study shows that over-expression of Foxo1 in preadipocytes suppresses adipocyte development (Nakae et al. 2003), we demonstrate that the expression level of Foxo1 is extremely low in preadipocytes and only increases from day 2 of DMI-induced adipocyte differentiation, reaching a maximum level at day 4 (Fig. 1B ). Furthermore, we show that knockdown of Foxo1 mRNA 4 d after DMI induction significantly suppresses adipocyte differentiation. It is possible that the study by Nakae et al. demonstrated inhibition of clonal expansion and terminal differentiation by introducing an excess level of Foxo1 during an early phase of differentiation when Foxo1 does not normally exist (Nakae et al. 2003). Foxo1 plays a potentially important role as a stage-specific regulator of adipocyte differentiation.
The terminal differentiation of adipocytes follows clonal expansion. As adipocyte differentiation involves cooperation between two distinct phenomena, cell proliferation and differentiation, the progression of differentiation requires tight control of the expression and activation of specific transcription factors. Notably, transcription factors that induce cell proliferation, including E2F, CDK4, and RB, are involved in the induction of gene expression and activation of PPAR-γ, which is essential for cell cycle arrest at the transition stage between clonal expansion and terminal differentiation (Hansen et al. 1999; Fajas et al. 2002a, b; Abella et al. 2005). The activation of PPAR-γ causes the subsequent inactivation of the accelerator of cell proliferation (Morrison and Farmer 1999). C/EBP-α also plays an important role in cell cycle arrest (Schuster and Porse 2006). The relay of information from factors controlling cell proliferation to factors controlling cell differentiation is essential for the induction of cell cycle arrest following clonal expansion to allow progression of the differentiation process to terminal differentiation. The significant accumulation of Foxo1 at the completion of clonal expansion enhances the expression of PPAR-γ and C/EBP-α (Figs. 1 and 4). This result suggests that Foxo1 may function as a key regulator of the transition from clonal expansion to terminal differentiation during adipocyte differentiation.
In summary, the present study reveals that Foxo1 is essential for adipocyte differentiation. Details of the role of Foxo1 in the mechanisms controlling adipocyte differentiation require further investigation.
References
Abella A.; Dubus P.; Malumbres M.; Rane S. G.; Kiyokawa H.; Sicard A.; Vignon F.; Langin D.; Barbacid M.; Fajas L. Cdk4 promotes adipogenesis through PPARgamma activation. Cell. Metab. 2: 239–249; 2005. doi:10.1016/j.cmet.2005.09.003.
Bligh E. G.; Dyer W. J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 37: 911–917; 1959.
Brunet A.; Bonni A.; Zigmond M. J.; Lin M. Z.; Juo P.; Hu L. S.; Anderson M. J.; Arden K. C.; Blenis J.; Greenberg M. E. Akt promotes cell survival by phosphorylating and inhibiting a forkhead transcription factor. Cell 96: 857–868; 1999. doi:10.1016/S0092-8674(00)80595-4.
Brunet A.; Sweeney L. B.; Sturgill J. F.; Chua K. F.; Greer P. L.; Lin Y.; Tran H.; Ross S. E.; Mostoslavsky R.; Cohen H. Y.; Hu L. S.; Cheng H. L.; Jedrychowski M. P.; Gygi S. P.; Sinclair D. A.; Alt F. W.; Greenberg M. E. Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science 303: 2011–2015; 2004. doi:10.1126/science.1094637.
Cao Z.; Umek R. M.; McKnight S. L. Regulated expression of three C/EBP isoforms during adipose conversion of 3T3-L1 cells. Genes Dev. 5: 1538–1552; 1991. doi:10.1101/gad.5.9.1538.
Christian M.; Zhang X.; Schneider-Merck T.; Unterman T. G.; Gellersen B.; White J. O.; Brosens J. J. Cyclic AMP-induced forkhead transcription factor, FKHR, cooperates with CCAAT/enhancer-binding protein beta in differentiating human endometrial stromal cells. J. Biol. Chem. 277: 20825–20832; 2002. doi:10.1074/jbc.M201018200.
Classon M.; Kennedy B. K.; Mulloy R.; Harlow E. Opposing roles of pRB and p107 in adipocyte differentiation. Proc. Natl. Acad. Sci. U. S. A. 97: 10826–10831; 2000. doi:10.1073/pnas.190343597.
Czech M. P. Insulin’s expanding control of forkheads. Proc. Natl. Acad. Sci. U. S. A. 100: 11198–11200; 2003. doi:10.1073/pnas.2135308100.
Fajas L.; Egler V.; Reiter R.; Hansen J.; Kristiansen K.; Debril M. B.; Miard S.; Auwerx J. The retinoblastoma-histone deacetylase 3 complex inhibits PPARgamma and adipocyte differentiation. Dev. Cell. 3: 903–910; 2002a. doi:10.1016/S1534-5807(02)00360-X.
Fajas L.; Landsberg R. L.; Huss-Garcia Y.; Sardet C.; Lees J. A.; Auwerx J. E2Fs regulate adipocyte differentiation. Dev. Cell. 3: 39–49; 2002b. doi:10.1016/S1534-5807(02)00190-9.
Hansen J. B.; Petersen R. K.; Larsen B. M.; Bartkova J.; Alsner J.; Kristiansen K. Activation of peroxisome proliferator-activated receptor gamma bypasses the function of the retinoblastoma protein in adipocyte differentiation. J. Biol. Chem. 274: 2386–2393; 1999. doi:10.1074/jbc.274.4.2386.
Hribal M. L.; Nakae J.; Kitamura T.; Shutter J. R.; Accili D. Regulation of insulin-like growth factor-dependent myoblast differentiation by Foxo forkhead transcription factors. J. Cell. Biol. 162: 535–541; 2003. doi:10.1083/jcb.200212107.
Hu E.; Kim J. B.; Sarraf P.; Spiegelman B. M. Inhibition of adipogenesis through MAP kinase-mediated phosphorylation of PPARgamma. Science 274: 2100–2103; 1996. doi:10.1126/science.274.5295.2100.
Kamei Y.; Mizukami J.; Miura S.; Suzuki M.; Takahashi N.; Kawada T.; Taniguchi T.; Ezaki O. A forkhead transcription factor FKHR up-regulates lipoprotein lipase expression in skeletal muscle. FEBS Lett. 536: 232–236; 2003. doi:10.1016/S0014-5793(03)00062-0.
Kim J. E.; Chen J. Regulation of peroxisome proliferator-activated receptor-gamma activity bymammalian target of rapamycin and amino acids in adipogenesis. Diabetes 53: 2748–2756; 2004. doi:10.2337/diabetes.53.11.2748.
Lam E. W.; Francis R. E.; Petkovic M. FOXO transcription factors: key regulators of cell fate. Biochem. Soc. Trans. 34: 722–726; 2006. doi:10.1042/BST0340722.
Li P.; Lee H.; Guo S.; Unterman T. G.; Jenster G.; Bai W. AKT-independent protection of prostate cancer cells from apoptosis mediated through complex formation between the androgen receptor and FKHR. Mol. Cell. Biol. 23: 104–118; 2003. doi:10.1128/MCB.23.1.104-118.2003.
Miki H.; Yamauchi T.; Suzuki R.; Komeda K.; Tsuchida A.; Kubota N.; Terauchi Y.; Kamon J.; Kaburagi Y.; Matsui J.; Akanuma Y.; Nagai R.; Kimura S.; Tobe K.; Kadowaki T. Essential role of insulin receptor substrate 1 (IRS-1) and IRS-2 in adipocyte differentiation. Mol. Cell. Biol. 21: 2521–2532; 2001. doi:10.1128/MCB.21.7.2521-2532.2001.
Morrison R. F.; Farmer S. R. Role of PPARgamma in regulating a cascade expression of cyclin-dependent kinase inhibitors, p18(INK4c) and p21(Waf1/Cip1), during adipogenesis. J. Biol. Chem. 274: 17088–17097; 1999. doi:10.1074/jbc.274.24.17088.
Naiki T.; Saijou E.; Miyaoka Y.; Sekine K.; Miyajima A. TRB2, a mouse Tribbles ortholog, suppresses adipocyte differentiation by inhibiting AKT and C/EBPbeta. J. Biol. Chem. 282: 24075–24082; 2007. doi:10.1074/jbc.M701409200.
Naïmi M.; Gautier N.; Chaussade C.; Valverde A. M.; Accili D.; Van Obberghen E. Dual role of transcription factor FoxO1 in controlling hepatic insulin sensitivity and lipid metabolism. Endocrinology 148: 2424–2434; 2007. doi:10.1210/en.2006-1411.
Nakae J.; Kitamura T.; Kitamura Y.; Biggs W. H. 3rd; Arden K. C.; Accili D. The forkhead transcription factor Foxo1 regulates adipocyte differentiation. Dev. Cell. 4: 119–129; 2003. doi:10.1016/S1534-5807(02)00401-X.
Nakae J.; Kitamura T.; Silver D. L.; Accili D. The forkhead transcription factor Foxo1 (Fkhr) confers insulin sensitivity onto glucose-6-phosphatase expression. J. Clin. Invest. 108: 1359–1367; 2001.
Reagan-Shaw S.; Ahmad N. A target for the management of cancer. Toxicol. Appl. Pharmacol. 224: 360–368; 2007. doi:10.1016/j.taap.2006.12.003.
Rosen E. D.; Sarraf P.; Troy A. E.; Bradwin G.; Moore K.; Milstone D. S.; Spiegelman B. M.; Mortensen R. M. PPAR gamma is required for the differentiation of adipose tissue in vivo and in vitro. Mol. Cell. 4: 611–617; 1999. doi:10.1016/S1097-2765(00)80211-7.
Rosen E. D.; Spiegelman B. M. Adipocytes as regulators of energy balance and glucose homeostasis. Nature 444: 847–853; 2006. doi:10.1038/nature05483.
Schmidt M.; Fernandez de Mattos S.; van der Horst A.; Klompmaker R.; Kops G. J.; Lam E. W.; Burgering B. M.; Medema R. H. Cell cycle inhibition by FoxO forkhead transcription factors involves downregulation of cyclin D. Mol. Cell. Biol. 22: 7842–7852; 2002. doi:10.1128/MCB.22.22.7842-7852.2002.
Schuster M. B.; Porse B. T. C/EBPalpha: a tumour suppressor in multiple tissues? Biochim. Biophys. Acta 1766: 88–103; 2006.
Student A. K.; Hsu R. Y.; Lane M. D. Induction of fatty acid synthetase synthesis in differentiating 3T3-L1 preadipocytes. J. Biol. Chem. 255: 4745–4750; 1980.
Tang Q. Q.; Lane M. D. Activation and centromeric localization of CCAAT/enhancer-binding proteins during the mitotic clonal expansion of adipocyte differentiation. Genes Dev. 13: 2231–2241; 1999. doi:10.1101/gad.13.17.2231.
Tang Q. Q.; Otto T. C.; Lane M. D. Mitotic clonal expansion: a synchronous process required for adipogenesis. Proc. Natl. Acad. Sci. U. S. A. 100: 44–49; 2003. doi:10.1073/pnas.0137044100.
Tran H.; Brunet A.; Grenier J. M.; Datta S. R.; Fornace A. J. Jr.; DiStefano P. S.; Chiang L. W.; Greenberg M. E. DNA repair pathway stimulated by the forkhead transcription factor FOXO3a through the Gadd45 protein. Science 296: 530–534; 2002.
Wiper-Bergeron N.; Wu D.; Pope L.; Schild-Poulter C.; Haché R. J. Stimulation of preadipocyte differentiation by steroid through targeting of an HDAC1 complex. EMBO J. 22: 2135–2145; 2003. doi:10.1093/emboj/cdg218.
Wu Z.; Bucher N. L.; Farmer S. R. Induction of peroxisome proliferator-activated receptor gamma during the conversion of 3T3 fibroblasts into adipocytes is mediated by C/EBPbeta, C/EBPdelta, and glucocorticoids. Mol. Cell. Biol. 16: 4128–4136; 1996.
Wu Z.; Rosen E. D.; Brun R.; Hauser S.; Adelmant G.; Troy A. E.; McKeon C.; Darlington G. J.; Spiegelman B. M. Cross-regulation of C/EBP alpha and PPAR gamma controls the transcriptional pathway of adipogenesis and insulin sensitivity. Mol. Cell. 3: 151–158; 1999. doi:10.1016/S1097-2765(00)80306-8.
Zhu Y.; Qi C.; Korenberg J. R.; Chen X. N.; Noya D.; Rao M. S.; Reddy J. K. Structural organization of mouse peroxisome proliferator-activated receptor gamma (mPPAR gamma) gene: alternative promoter use and different splicing yield two mPPAR gamma isoforms. Proc. Natl. Acad. Sci. U. S. A. 92: 7921–7925; 1995. doi:10.1073/pnas.92.17.7921.
Acknowledgments
We wish to thank Dr. M. Greenberg (Harvard Medical School) for providing expression plasmid of Foxo1, pcDNA3-Foxo1. We are also grateful to Dr. Takiguchi (Chiba University) for providing plasmids pEF-BOS-C/EBP-α, -β, and -δ. This work was supported in part by Grants-in-Aid for Scientific Research from the Ministry of Education, Science, Sports, and Culture of Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: J. Denry Sato
Rights and permissions
About this article
Cite this article
Munekata, K., Sakamoto, K. Forkhead transcription factor Foxo1 is essential for adipocyte differentiation. In Vitro Cell.Dev.Biol.-Animal 45, 642–651 (2009). https://doi.org/10.1007/s11626-009-9230-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-009-9230-5