Abstract
Background
This study was performed to verify the superiority of a new “non-tensioning method” for avoiding stricture of the cervical esophagogastric anastomosis by circular stapling compared with the conventional method.
Methods
In total, 395 consecutive patients who underwent McKeown esophagectomy with gastric conduit (GC) reconstruction were reviewed. A 4 cm-wide GC was created and pulled up at the cervical site through the retrosternal route. The esophagogastrostomy site of the GC was planned as far caudally as possible on the greater curvature side. In the conventional technique, the stapler was fired while pulling the GC to avoid tissue slack. In the non-tensioning technique, the stapler was fired through the natural thickness of the stomach wall. The length of the blind end was changed from 4 to 2 cm in the non-tensioning technique. Anastomotic leakage and stricture formation were compared between the two techniques, and adjustment was performed using propensity score matching.
Results
The conventional group comprised 315 patients, and the non-tensioning group comprised 80 patients. Anastomotic leakage occurred in 22 (7%) and 2 (2.5%) patients, respectively (P = 0.134) [and in 9 (2.9%) and 2 (2.5%) patients, respectively, if leakage at the blind end was excluded]. Anastomotic stricture occurred in 92 (29.2%) and 3 (3.8%) patients, respectively (P < 0.001). The propensity score-matching analysis including 79 pairs of patients confirmed a lower stricture rate in the non-tensioning than conventional group (2.5% vs. 29.1%, P < 0.001).
Conclusions
The non-tensioning technique significantly reduced the incidence of anastomotic stricture compared with the conventional technique.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Esophagectomy with gastric conduit (GC) reconstruction is considered the standard surgical treatment for thoracic esophageal cancer worldwide. McKeown esophagectomy, which involves cervico-thoraco-abdominal lymph node dissection and cervical esophagogastric anastomosis, is generally recognized as a very invasive operation with a high risk of postoperative morbidity and mortality1,2. However, the incidence of severe postoperative complications such as pneumonia and anastomotic leakage has gradually decreased with improvements in surgical techniques and postoperative management3. Although not life-threatening, anastomotic stricture is a troublesome complication that causes dysphagia. It is often disruptive because it occurs at 2 to 3 months postoperatively4,5, when many patients who have undergone gastrointestinal reconstruction after esophagectomy are suffering from anorexia6.
The most basic and classic anastomotic technique is hand-sewn anastomosis. This technique has the advantage of being adaptable to all patients after esophagectomy; however, it is time-consuming and operator-dependent. A circular stapling device, the use of which is simple and uniform, has been developed to resolve these problems. Systematic reviews comparing the circular stapling technique with the hand-sewn technique showed that the incidence of anastomotic leakage was equivalent between the two (6–9% and 8–11%, respectively), but stricture formation was more frequent with the circular stapling technique than with the hand-sewn technique (23–31% and 14–16%, respectively)7,8. Therefore, to some degree, anastomotic stricture formation after use of a circular stapler has been recognized as an inevitable problem. A collard or side-to-side stapling method that creates a wide anastomotic opening using a linear stapler has been developed as a countermeasure9,10. Several studies comparing different stapling methods showed that linear stapling techniques were associated with a 3.0 to 10.9% incidence of anastomotic leakage and a 2.9 to 14.1% incidence of anastomotic stricture, leading to the conclusion that linear stapling is advantageous for prevention of anastomotic stricture formation11,12,13. However, a major disadvantage of linear stapling methods is the narrowness of their adaptation; they cannot be applied unless the cervical esophagus remains long. Thus, the optimal anastomosis technique for the cervical esophagus remains controversial14.
We have always considered that the simplicity and versatility of the circular stapling method are great advantages. Therefore, we have been improving and have now almost completed our technique to reduce anastomotic failure. As the next step in improving this technique, we attempted to prevent anastomotic stricture. We considered that one cause of anastomotic stricture was the return of the stomach wall to its original thickness after creation of the anastomosis because the GC wall had been anastomosed with a thin wall while the GC was pulled to avoid slack at the anastomosis site. Based on this theory, the conventional method was modified to a “non-tensioning method” to prevent anastomotic stricture formation. The purpose of this study was to verify the superiority of the non-tensioning method in preventing anastomotic stricture compared with the previously used conventional method in esophagogastric anastomosis after McKeown esophagectomy.
Materials and Methods
Patients
In total, 570 consecutive patients who underwent esophagectomy for esophageal cancer at the Department of Surgery, Kindai University Faculty of Medicine from July 2009 to December 2020 were reviewed in the present study. The inclusion criteria were performance of McKeown esophagectomy with GC reconstruction, cervical anastomosis using a 25-mm circular stapler, pulling up of the GC through the retrosternal route, and no residual tumor. The exclusion criteria were a two-stage operation, in-hospital mortality, follow-up period of less than 6 months, and postoperative radiation treatment at the cervical site. After exclusion of 175 patients, 395 patients were enrolled in the study and retrospectively analyzed (Fig. 1). The Institutional Review Board of Kindai University Faculty of Medicine approved this study (approval number R03-087).
Surgical Procedure
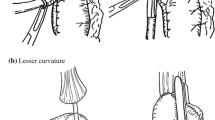
In July 2009, we established a GC reconstruction procedure to prevent anastomotic leakage in our institute, and this uniform procedure (described as follows) has been performed since that time. First, a GC was created using a linear cutter by a multiple cutting method in which the GC was cut by a few short pitches from the lesser curvature and then gradually cut by long pitches with sufficient stretch along the greater curvature. Since 2014, the first cut from the lesser curvature has been performed with a curved cutter-stapler device (Supplemental Fig. 1a-e). The 4 cm-wide GC was created as long as possible (Fig. 2a). The anvil of the circular stapler was secured with a purse-string suture at the proximal end of the esophagus. Second, the GC was pulled up to the cervical site through the retrosternal route, and esophagogastrostomy was planned using a 25-mm circular stapler at the GC as far caudally as possible on the greater curvature side, aiming for a site with good circulation (Fig. 2b). A circular stapled device was inserted through the blind end, and esophagogastrostomy was performed. Third, the blind end of the GC was cut down by a linear stapler and a buried suture was added (Fig. 2c). After slightly pulling down the GC, the dorsal anastomotic site, tracheal membranous portion, and blind end of the GC were covered with omentum (Fig. 2d, e). Finally, the dissected anterior cervical muscles were sutured to the sternocleidomastoid muscle to cover the blind end of the GC, and the surgical wound was closed without a drain at the anastomotic site. A nasogastric tube with automatic intermittent suction was inserted and used for decompression for 5 days.
Anastomotic procedure. a The 4 cm-wide gastric conduit was created as long as possible. b The esophagogastrostomy site was planned as far caudally as possible on the greater curvature side. c The blind end was cut down by a linear stapler. d, e The dorsal anastomotic site, tracheal membranous portion, and blind end of the gastric conduit were covered with omentum
The above-described steps were common to both the conventional and non-tensioning methods. The conventional technique was refined to the non-tensioning method for prevention of anastomotic stricture formation in January 2018. The differences between the two methods are as follows. In the conventional method, the anvil of the circular stapler was closed while pulling the GC to avoid tissue slack at the anastomosis site (Fig. 3a). In the non-tensioning method, the anvil of the circular stapler was closed through the natural thickness of the stomach wall without pulling (Fig. 3b). Additionally, the height of the B-form stapler was adjusted from ≤ 1.5 mm in the conventional method to ≥ 2.0 mm in the non-tensioning method to loosen the tissue pressure, and the length of the blind end was changed from 4 cm in the conventional method to 2 cm in the non-tensioning method. The conventional method was performed by Y.T. as the operator, and the non-tensioning method was performed by S.O. as the operator or instructor. Both doctors are board-certified esophageal surgeons. Y.T. and S.O. participated in both methods to maintain the quality of the operation. Therefore, both methods were equivalent in quality with the exception of tension at the anastomotic site and length of the blind end.
Evaluation
Anastomotic leakage was identified by the existence of free air outside the intestine as shown by computed tomography or by performing drainage at the cervical wound. The site of leakage was confirmed by endoscopy. An anastomotic stricture was defined as stenosis impeding the passage of a 9-mm endoscope, for which endoscopic dilatation was needed without anastomotic relapse. Clavien–Dindo grade ≥ II pneumonia and other severe grade ≥ IIIb complications were considered postoperative complications that may affect the general condition.
Propensity Score-Matched Analysis
Propensity score-matched analysis was conducted using a logistic regression model and the following covariates presumed to be risk factors for anastomotic stricture: sex, age, body mass index, cardiovascular disease, chronic obstructive pulmonary disease, diabetes mellitus, tumor location, clinical stage, preoperative treatment, approach in the abdominal operation, and field of cervical lymph node dissection.
Statistical Analysis
Continuous variables are expressed as mean or median. The Mann–Whitney U test was used to compare nonparametric variables. Categorical data were compared using Fisher’s exact test or Pearson’s chi-square test. We investigated the risk factors for anastomotic stricture using logistic regression analysis. Several clinicopathological variables were selected according to our clinical experience, and variables with P values of < 0.1 in the univariate analysis were included in the multivariable analysis. Differences were considered statistically significant at P < 0.05. All statistical analyses were performed using JMP data analysis software, version 9.0.2 (SAS Institute Inc., Cary, NC, USA).
Results
The characteristics of the patients in the non-tensioning group and conventional group are shown in Table 1. There were no major differences in age, body mass index, cardiovascular disease, chronic obstructive pulmonary disease, diabetes mellitus, tumor location, clinical stage, or field of cervical lymph node dissection between the two groups. However, the non-tensioning group had significantly more female patients, neoadjuvant chemotherapy, and hand-assisted laparoscopic surgery than the conventional group. After propensity score matching, 79 patients were respectively selected in each group, and all variables became equivalent in both groups.
Postoperative Complications and Stricture Formation
Postoperative complications before and after propensity score matching are shown in Table 2. Comparison of all patients showed that 2 patients (2.5%) in the non-tensioning group and 22 patients (7%) in the conventional group developed anastomotic leakage. Examination of the leakage site showed equivalent rates of leakage between the groups at the esophagogastric anastomosis [two patients (2.5%) in the non-tensioning group and nine patients (2.9%) in the conventional group]. Leakage at the blind end occurred in 13 patients (4.1%), increasing the total leakage rate in the conventional group. The rates of pneumonia, severe grade ≥ IIIb complications, and hospitalization were similar between the two groups. Anastomotic stricture as the primary endpoint occurred in only 3 patients (3.8%) in the non-tensioning group but in 23 patients (29.1%) in the conventional group (P < 0.001). Most cases were resolved by treatment with bougies. Additionally, refractory strictures requiring ≥ 5 bougies occurred in no patients in the non-tensioning group but in 4 patients in the conventional group. After propensity score matching, the results were similar to those of all patients; there was no difference in the rate of anastomotic leakage, but the rate of anastomotic stricture was 3.8% in the non-tensioning group and 29.1% in the conventional group.
Risk Factors for Anastomotic Stricture
The multivariable logistic regression analysis showed that three-field lymphadenectomy, anastomotic leakage, and the conventional method were significant risk factors for anastomotic stricture (Table 3).
Discussion
The present study demonstrated that the non-tensioning anastomotic method significantly reduced the incidence of anastomotic stricture compared with the conventional method. Reducing the rate of anastomotic stricture to 3.8% with the use of a circular stapler is very important because it would allow for the use of a simple and versatile circular stapled device and improve the patient’s quality of life.
Circular stapled anastomosis is an easy method to perform. The head diameter of the circular stapler device reaches 21 to 28 mm, and it can be adapted to almost all cases of cervical anastomosis during McKeown esophagectomy. This versatility is a strong advantage; however, the frequent occurrence of anastomotic stricture has been recognized as an intractable problem7,8,15. New anastomotic methods such as the collard and side-to-side techniques were developed as countermeasures. Although they are excellent techniques with a low stricture rate, they have poor versatility because they cannot be adapted to cases with a short remnant cervical esophagus11,12,13. If a special technique for the hand-sewn method can be mastered, the stricture rate may be lowered; however, this is a highly operator-dependent and distant goal. The ideal is to develop a simple circular stapling method without stricture formation.
The strongest causative factor in the formation of anastomotic stricture is anastomotic leakage as indicated by our multivariable logistic analysis and previous reports 4,5,16. Leakage results in tissue necrosis and collapse of the tissue structure at the esophagogastric anastomosis site. The tissue is reconstituted with collagen and fibronectin produced by fibroblasts in the granulation phase, after which re-epithelialization occurs. Contraction occurs simultaneously with re-epithelization; during this process, myofibroblasts that have been recruited to the area around the wound site pull against each other to reduce the anastomotic diameter17. Therefore, eradication of anastomotic leakage is the most important factor in avoiding anastomotic stricture. We considered that an important point in avoiding anastomotic leakage was to perform the anastomosis as proximally as possible along the greater curvature using a long GC, aiming for a region of good circulation18,19. As a result, at the time of the conventional method, the anastomotic leakage rate of the esophagogastric anastomosis site had already been successfully suppressed to 2.9%. Regardless of this low esophagogastric anastomotic leakage rate, anastomotic stricture occurred at an incidence of 29.2%. Therefore, this procedure alone was not enough to avoid anastomotic stricture.
The cause of anastomotic stricture formation is multifactorial, and the reported risk factors for stricture (other than leakage) include older age, a high body mass index, upper thoracic tumor location, and cardiovascular disease4,5,13,20. However, we suspected that the major cause of stricture was our own surgical technique. Surgeons may have unknowingly formed an anastomotic ring that is prone to stenosis from the beginning of the procedure. Esophagogastric anastomosis using a circular stapled device had been conventionally performed with pulling of the GC to avoid tissue slack at the anastomosis site (Fig. 3a). As a result, the staples were fired into the stretched stomach wall. We show models mimicking the esophagogastric anastomosis using surgical gloves to verify the mechanism of stricture formation in Fig. 4. The difference between the conventional method and the non-tensioning method is the degree of extension on the gastric tube side due to traction. The black line, which is the anastomotic line in the non-tensioning method, moves outward by traction in the conventional method (Fig. 4a, e). After the staples are fired into the stretched stomach wall, the stomach wall returns to its original thickness. Although the anastomosis caliber is essentially the same in both methods because of immobilization by the stapler (Fig. 4b, f), the staple arrangement in the conventional technique is denser than that in the non-tensioning method because of the shrinking mechanism (Fig. 4c, g). This dense staple arrangement might decrease the blood flow in the stapled part, resulting in ischemia and subsequent cicatricial contracture. Thus, the anastomotic ring in the conventional technique not only has the power to shrink the original anastomotic caliber but is also prone to the development of cicatricial contracture. In addition, reducing the pressure placed on the tissue by changing the B-form stapler height from 1.5 to 2.0 mm might have helped to prevent tissue necrosis, although this is only speculation. In fact, narrow ring-shaped necrosis of the staple line was sometimes confirmed by endoscopic observation around 7 days after surgery using the conventional technique (Supplemental Fig. 2a). However, ring-shaped necrosis was rarely observed with use of the non-tensioning method (Supplemental Fig. 2b). One proposed mechanism of stricture formation states that circular stapled anastomosis, which involves the application of serosal apposition stitches outside the walls of the stomach and esophagus, corresponds to secondary union and that the anastomosis ring develops into a stenosis during the process of necrosis and repair21,22. However, the effect of secondary union is considered to be minimized if ring-shaped necrosis does not occur.
The models mimicking esophagogastric anastomosis with surgical gloves. a, e The black line is the anastomotic line in the non-tensioning method. It moved outward by traction in the conventional method. b, f The anastomosis caliber size was essentially the same in both methods because of immobilization by the stapler. c, g However, the staple arrangement in the conventional method was denser than that in the non-tensioning method because of the shrinking mechanism. d, h The punched stomach wall tissue shrank in the conventional method
The present study had several limitations. As a retrospective, single-center study, the results generated are dependent on the reliability of the data. First, with respect to the stomach wall thickness at the time of anastomosis in the conventional group, it is difficult to know exactly how thin the stomach wall became and in how many cases a dense staple arrangement developed because of pulling on the stomach when firing the stapler. Second, it was unclear how frequent narrow ring-shaped necrosis occurred because we did not examine all patients by endoscopy after the operation. A prospective randomized study is required to determine the incidence of necrosis. However, despite these limitations, the present study is noteworthy in that the efficacy of the non-tensioning method was verified with as little bias as possible by enforcing strict eligibility for the GC reconstructive procedures and performing propensity score matching. Furthermore, considering the facts that most of the patients were re-examined at our hospital without loss to follow-up and that we rarely performed endoscopic dilatation treatment with a bougie after using the non-tensioning method, our study results present a true picture of anastomotic stricture formation after GC reconstruction with McKeown esophagectomy in our institute.
Conclusion
The present study demonstrated that the non-tensioning method significantly reduced the anastomotic stricture rate to 3.8% compared with the conventional method. It is very important that the subsequent reconstruction can be performed easily because surgical treatment of thoracic esophageal cancer is a great burden for patients and requires substantial effort by surgeons. In this sense, we believe that the non-tensioning method should be recommended as a simple-to-perform and versatile circular stapled anastomosis method that is associated with fewer postoperative complications.
References
Berry MF, Atkins BZ, Tong BC, Harpole DH, D’Amico TA, Onaitis MW. A comprehensive evaluation for aspiration after esophagectomy reduces the incidence of postoperative pneumonia. J Thorac Cardiovasc Surg 2010; 140: 1266-1271.
Takeuchi H, Miyata H, Gotoh M, et al. A risk model for esophagectomy using data of 5354 patients included in a Japanese nationwide web-based database. Ann Surg 2014; 260: 259-266.
Law S, Wong KH, Kwok KF, Chu KM, Wong J. Predictive factors for postoperative pulmonary complications and mortality after esophagectomy for cancer. Ann Surg 2004; 240: 791-800.
van Heijl M, Gooszen JA, Fockens P, Busch OR, van Lanschot JJ, van Berge Henegouwen MI. Risk factors for development of benign cervical strictures after esophagectomy. Ann Surg 2010; 251: 1064-1069.
Tanaka K, Makino T, Yamasaki M, et al. An analysis of the risk factors of anastomotic stricture after esophagectomy. Surg Today 2018; 48: 449-454.
Baker M, Halliday V, Williams RN, Bowrey DJ. A systematic review of the nutritional consequences of esophagectomy. Clin Nutr 2015. doi:https://doi.org/10.1016/j.clnu.2015.08.010.
Beitler AL, Urschel JD. Comparison of stapled and hand-sewn esophagogastric anastomoses. Am J Surg 1998; 175: 337-340.
Markar SR, Karthikesalingam A, Vyas S, Hashemi M, Winslet M. Hand-sewn versus stapled oesophago-gastric anastomosis: systematic review and meta-analysis. J Gastrointest Surg 2011; 15: 876-884.
Collard JM, Romagnoli R, Goncette L, Otte JB, Kestens PJ. Terminalized semimechanical side-to-side suture technique for cervical esophagogastrostomy. Ann Thorac Surg 1998; 65: 814-817.
Orringer MB, Marshall B, Iannettoni MD. Eliminating the cervical esophagogastric anastomotic leak with a side-to-side stapled anastomosis. J Thorac Cardiovasc Surg 2000; 119: 277-288.
Wang ZQ, Jiang YQ, Xu W, et al. A novel technique for cervical gastro-oesophageal anastomosis during minimally invasive oesophagectomy. Int J Surg 2018; 53: 221-229.
Sugimura K, Miyata H, Matsunaga T, et al. Comparison of the modified Collard and hand-sewn anastomosis for cervical esophagogastric anastomosis after esophagectomy in esophageal cancer patients: A propensity score-matched analysis. Ann Gastroenterol Surg 2019; 3: 104-113.
Hirano Y, Fujita T, Sato K, et al. Totally Mechanical Collard Technique for Cervical Esophagogastric Anastomosis Reduces Stricture Formation Compared with Circular Stapled Anastomosis. World J Surg 2020; 44: 4175-4183.
Kamarajah SK, Bundred JR, Singh P, Pasquali S, Griffiths EA. Anastomotic techniques for oesophagectomy for malignancy: systematic review and network meta-analysis. BJS Open 2020; 4: 563-576.
Rice TW. Anastomotic stricture complicating esophagectomy. Thorac Surg Clin 2006; 16: 63-73.
Honkoop P, Siersema PD, Tilanus HW, Stassen LP, Hop WC, van Blankenstein M. Benign anastomotic strictures after transhiatal esophagectomy and cervical esophagogastrostomy: risk factors and management. J Thorac Cardiovasc Surg 1996; 111: 1141-1146; discussion 7-8.
Nishikawa K, Fujita T, Yuda M, et al. Early prediction of complex benign anastomotic stricture after esophagectomy using early postoperative endoscopic findings. Surg Endosc 2020; 34: 3460-3469.
Sasaki K, Omoto I, Uchikado Y, et al. Comparison of greater curvature and lesser curvature circular-stapled esophagogastrostomy after esophagectomy in patients with esophageal cancer: a prospective randomized controlled trial. Surg Today 2021; 51: 575-581.
Shen Y, Wang H, Feng M, Tan L, Wang Q. The effect of narrowed gastric conduits on anastomotic leakage following minimally invasive oesophagectomy. Interact Cardiovasc Thorac Surg 2014; 19: 263-268.
Xu QR, Wang KN, Wang WP, Zhang K, Chen LQ. Linear stapled esophagogastrostomy is more effective than hand-sewn or circular stapler in prevention of anastomotic stricture: a comparative clinical study. J Gastrointest Surg 2011; 15: 915-921.
Wang WP, Gao Q, Wang KN, Shi H, Chen LQ. A prospective randomized controlled trial of semi-mechanical versus hand-sewn or circular stapled esophagogastrostomy for prevention of anastomotic stricture. World J Surg 2013; 37: 1043-1050.
Liu QX, Qiu Y, Deng XF, Min JX, Dai JG. Comparison of outcomes following end-to-end hand-sewn and mechanical oesophagogastric anastomosis after oesophagectomy for carcinoma: a prospective randomized controlled trial. Eur J Cardiothorac Surg 2015; 47: e118-123.
Acknowledgements
We thank Angela Morben, DVM, ELS, from Edanz for editing a draft of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information

Fig. S1
Procedure used to create gastric conduit. (a) The first cut from the lesser curvature was performed with a curved cutter-stapler device. (b–e) The stomach was then sequentially cut off with a linear cutter according to the straightened distance based on the stretched greater curvature.(PNG 4225 kb)

Fig. S2
(a) In some cases, narrow ring-shaped necrosis of the staple line was confirmed by endoscopic observation around 7 days after surgery with the conventional method. (b) No ring-shaped necrosis was observed in the non-tensioning method.(PNG 4474 kb)
Rights and permissions
About this article
Cite this article
Shiraishi, O., Yasuda, T., Kato, H. et al. Circular Stapler Method for Avoiding Stricture of Cervical Esophagogastric Anastomosis. J Gastrointest Surg 26, 725–732 (2022). https://doi.org/10.1007/s11605-022-05266-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-022-05266-4