Abstract
Background
The superiority of outcomes associated with anatomical resection (AR) versus those associated with non-anatomical resection (NAR) remains controversial in patients with hepatocellular carcinoma (HCC). The aim of this study was to evaluate the significance of AR on therapeutic outcomes of patients with small HCCs (≤ 5 cm), using propensity score–matched (PSM) analysis.
Methods
A total of 195 patients who had undergone elective hepatic resection for small HCCs (≤ 5 cm) were included in this study. We conducted PSM analysis for baseline characteristics (age, sex, hepatitis virus status, retention rate of indocyanine green at 15 min, and Child-Pugh grade), preoperative serum α-fetoprotein, and tumor characteristics (tumor size, tumor number, portal vein invasion, and surgical margin status) to eliminate potential selection bias. The prognostic significance of AR on the disease-free and overall survival was analyzed in patients selected by PSM analysis.
Results
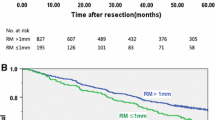
Applying PSM analysis, the patients were divided into PSM-AR (N = 66) and PSM-NAR (N = 66) groups. Disease-free survival was significantly better in the PSM-AR group than that of the PSM-NAR group (P = 0.018), while there was no significant difference in the overall survival between the PSM-AR and PSM-NAR groups (P = 0.292). The univariate HRs of the PSM-AR group were 0.55 (95% CI, 0.33–0.90) for disease-free survival and 0.61 (95% CI, 0.24–1.53) for overall survival, respectively. Remnant liver recurrence was significantly lower in the AR group (P = 0.014).
Conclusions
AR may improve the disease-free survival in HCC patients with tumors of ≤5 cm diameter.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hepatocellular carcinoma (HCC) is the seventh most common malignancy, but it is the second most common cause of cancer deaths worldwide.1 Advances in surgical techniques and perioperative management have made liver resection a potentially curative treatment for HCC. Among the several options for treatment, surgical resection may have a prognostic advantage over radiofrequency ablation, especially for solitary HCCs.2 However, the recurrence rate of HCC after curative hepatic resection remains high. Previous reports showed that even in patients with small HCCs, approximately 50–60% of those who underwent resection recurred within the first 3 years, and more than 70% recurred within the first 5 years.3,4
Anatomical resection (AR) is considered likely to improve outcome after liver resection since it may suppress tumor spread and micrometastasis through the portal vein by accomplishing systematic removal of the tumor-bearing portal region.5,6,7,8,9 On the other hand, several studies have suggested that AR provides no prognostic benefit over limited partial non-anatomical resection (NAR).10,11,12 The efficacy of AR remains controversial and may vary according to clinicopathological factors such as tumor size, portal vein invasion, and functional liver reserve.13,14 Therefore, in this study, we examined the prognostic significance of AR in patients with small HCCs (≤ 5 cm) by using a multi-institutional database. To minimize potential selection bias, we conducted propensity score–matched (PSM) analysis for baseline and tumor characteristics.
Patients and Methods
Patient Selection
Between April 2003 and December 2015, 306 patients with HCC underwent primary hepatic resection at the five institutions affiliated with the Jikei University (Department of Surgery, The Jikei University Hospital; The Jikei University Kashiwa Hospital; The Jikei University Daisan Hospital; The Jikei University Katsushika Medical Center; and Department of Digestive Surgery, Kawaguchi Municipal Medical Center). Of the original 306 patients, 29 were excluded: 16 patients for concomitant resection of other malignancies, 5 patients for lack of data, and 8 patients for postoperative mortality. Of the remaining 277 patients, 195 with available data and HCCs ≤5 cm in diameter were enrolled in the study. We performed a retrospective review of a prospectively maintained patient database. This study was approved by the Ethics Committee of the Jikei University School of Medicine.
Treatment and Follow-up
Generally, the extent and type of hepatic resection was determined by preoperative tumor staging, retention rate of indocyanine green at 15 min (ICGR15) before surgery, and hepatic reserve, as described by Miyagawa et al..15 Nomenclature of the segment and types of operations followed the Brisbane 2000 terminology.16 Resection types were classified into two groups: AR (extended lobectomy, lobectomy, segmentectomy, or subsegmentectomy) and limited partial NAR. AR was defined as complete resection of the anatomical lesion identified by prior ischemia or dye staining. Liver parenchymal transection was performed using a Cavitron Ultrasonic Surgical Aspirator (CUSA™; Integra LifeSciences Corporation, Princeton, NJ, USA) with a few exceptions, and the method was standardized between institutions. Tumor-Node-Metastasis (TNM) classification was based on tumor pathology and the General Rules for the Clinical and Pathological Study of Primary Liver Cancer by the Liver Cancer Study Group of Japan.17 No adjuvant and/or neoadjuvant therapy was administered during this period.
The recurrence of HCC was defined as hypervascular hepatic or extrahepatic tumors newly detected by ultrasonography, computed tomography, magnetic resonance imaging, or angiography with or without an increase in serum α-fetoprotein, or protein induced by vitamin K absence or antagonist-II. Recurrent liver HCC was treated by repeated hepatic resection, local ablation therapy, or transarterial chemoembolization based on hepatic functional reserve assessed mainly by ICGR15 results. Extrahepatic recurrence was treated conservatively in most cases.
Statistical Analyses
All statistical analyses were conducted using IBM® SPSS statistics version 20.0 (IBM Japan, Tokyo, Japan), and all P-values were two-sided with α level of 0.05. To eliminate potential selection bias, we conducted PSM analysis. Our primary analyses assessed the association of AR with disease-free and overall survival. Secondary analyses consisted of all other tests, including assessment of risk estimates.
Data are expressed as median, range, or ratio. Continuous and categorical variables were compared using the Mann-Whitney U or chi-square tests, as appropriate.
We evaluated the prognostic significance of AR in patients with HCCs ≤5 cm. Univariate and multivariate Cox proportional-hazards regression models were used to estimate the hazard ratios (HR) for disease-free and overall survival. The multivariable Cox regression model initially included age (continuous), sex (female vs. male), hepatitis virus status (HBC vs. HCV vs. non-B non-C), ICGR15 (continuous), Child-Pugh grade (A vs. B), serum AFP level (continuous), tumor size (continuous), tumor number (single vs. multiple), portal vein invasion (yes vs. no), and surgical margin status (negative vs. positive). A backward elimination was conducted with a P threshold of 0.05 to select variables for the final models.
The propensity score was calculated using baseline characteristics in the logistic regression model. Baseline characteristics included age, sex, hepatitis virus status, ICGR15, Child-Pugh score, serum AFP level, tumor size, tumor number, portal vein invasion, and surgical margin status. The AR and NAR resection groups were matched in a 1:1 ratio by PSM using a caliper of 0.0335. The Kaplan-Meier method was used to estimate cumulative survival probabilities, and the differences between groups were compared using the log-rank test. Cox proportional-hazards regression models were used to calculate HR and 95% confidence interval (CI) for disease-free and overall survival.
Results
Baseline Characteristics of HCCs ≤5 cm According to Type of Resection
Baseline patient characteristics are specified in Table 1 as median, range, or ratio. Of 195 patients with HCCs ≤5 cm in diameter, 93 patients underwent AR, whereas 102 patients underwent limited partial NAR. AR included more than 2 segmentectomies in 11 patients, segmentectomy in 37 patients, and subsegmentectomy in 45 patients. Patients who underwent AR had lower ICGR15 values and larger tumor diameters. Among 195 patients, none exhibited lymph node metastases. In total; the 5-year disease-free survival and overall survival rates after hepatic resection for HCC were 39.5% and 80.4%, respectively.
Type of Resection in Relation to Disease-Free and Overall Survival for HCCs ≤5 cm
We examined the association of AR with disease-free and overall survival in patients with HCCs ≤5 cm (Table 2). In multivariate Cox regression analyses, AR was associated with lower recurrence (P = 0.001), but not with overall mortality (P = 0.426). The multivariate-adjusted HRs for recurrence and overall mortality were 0.46 (95% CI, 0.29–0.88) and 0.73 (95% CI, 0.34–1.58), respectively.
Baseline Characteristics of HCCs ≤5 cm Matched by Propensity Score According to Type of Resection
Based on a 1:1 PSM analysis, 132 patients were classified into propensity-matched AR (PSM-AR) (N = 66) and propensity-matched NAR groups (PSM-NAR) (N = 66). Baseline patient characteristics are outlined in Table 3. After PSM analysis, there were no significant differences in the baseline characteristics between the PSM-AR and PSM-NAR groups.
Type of Resection in Relation to Disease-Free and Overall Survival for HCCs ≤5 cm with PSM Analysis
Table 4 shows the association of AR with disease-free and overall survival by PSM analysis (Table 4). AR was associated with lower recurrence (P = 0.018, Fig. 1a), but not with overall mortality (P = 0.292, Fig. 1b). The HRs for recurrence and overall mortality were 0.55 (95% CI 0.33–0.90) and 0.61 (95% CI, 0.24–1.53), respectively.
Short- and Long-term Outcomes for HCCs ≤5 cm According to Type of Resection
Table 5 shows the relationship between type of resection and short- and long-term outcomes. In univariate analyses, the duration of operation was significantly longer in the AR group (P = 0.013). Moreover, remnant liver recurrence was significantly lower in the AR group (P = 0.014).
Discussion
In the current study, utilizing the PSM method, we found that anatomical resection was associated with lower recurrence in patients with HCCs ≤5 cm. Furthermore, remnant liver recurrence was significantly lower in the AR group. These findings support the significance of AR for the therapeutic outcome of small hepatocellular carcinoma (≤ 5 cm), and reinforce its role in preventing spread through the portal vein.
The effectiveness of AR varies across studies and decades.8 A Japanese nationwide survey showed that AR was an independent factor influencing disease-free survival for patients with HCCs of 2–5 cm in diameter.18 Two recent PSM analyses from Japan19 and China20 investigated HCCs with microvascular invasion. One study demonstrated that local recurrence around the resection site was suppressed by AR,20 whereas the other study revealed that AR did not significantly improve disease-free or overall survival in patients with HCCs exhibiting micro-portal invasion.19 This evidence suggests that the beneficial effect of AR may vary depending on a number of tumor factors such as size, portal vein invasion, and location.13,14 In addition, selection of a surgical approach could be influenced by liver functional reserve and volume. Thus, a PSM analysis minimizing potential selection bias enables a more accurate evaluation of the AR effect. In this study, we showed that AR improved disease-free survival, but did not improve overall survival in patients with small HCCs (≤ 5 cm). A possible reason for the discrepancy between disease-free and overall survival is that the prognosis for HCC strongly depends on the types and quality of repeated treatments for recurrent tumors.21 In addition, the balance between the extent of surgical excision and preservation of remnant liver function might affect overall survival because underlying liver function has previously been associated with prognosis.22,23
With recent developments in preoperative liver simulation using three-dimensional imaging, the concept of cone-unit liver resection has been described.6,24,25 Cone-unit resection is an AR procedure that attempts to remove tumor-bearing portal territory branches, enabling a parenchymal-sparing resection. Evidence suggests that cone-unit resection may confer oncological benefits.24 In addition, this approach can be performed using a laparoscopic approach.25 Given that the survival of patients with HCC depends on underlying liver function,22,23 a liver parenchymal-preserving approach might allow repeated liver resections for recurrent HCC, ultimately improving outcomes.
We acknowledge the potential limitations of this study. Although we attempted to minimize selection bias and the effect of baseline characteristics by applying PSM analysis, the possibility of other unconsidered biases still remains.
A core strength of our study was the access to a multi-institutional database. The study population was derived from five hospitals, increasing the generalizability of our findings. Nonetheless, our results will require validation in independent, randomized controlled studies in the new era of laparoscopic surgery, molecular targeted therapy, and immunotherapy.25,26
Conclusion
We demonstrated that AR was associated with lower recurrence in patients with HCCs ≤5 cm, suggesting it may be beneficial for preventing the spread of small tumors through the portal vein. Our results, obtained using data from multiple institutions, may inform future research regarding the influence of tumor characteristics, size, and pathological features on AR and patient outcomes.
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394-424.
Sakon M, Kobayashi S, Wada H, Eguchi H, Marubashi S, Takahashi H, Akita H, Gotoh K, Yamada D, Asukai K, Hasegawa S, Ohue M, Yano M, Nagano H. “Logic-Based Medicine” Is More Feasible than “Evidence-Based Medicine” in the Local Treatment for Hepatocellular Carcinoma. Oncology. 2020;98:259-266.
Shim JH, Jun MJ, Han S, Lee YJ, Lee SG, Kim KM, Lim YS, Lee HC. Prognostic nomograms for prediction of recurrence and survival after curative liver resection for hepatocellular carcinoma. Ann Surg. 2015;261:939-946.
Marubashi S, Gotoh K, Akita H, Takahashi H, Sugimura K, Miyoshi N, Motoori M, Kishi K, Noura S, Fujiwara Y, Ohue M, Nakazawa T, Nakanishi K, Ito Y, Yano M, Ishikawa O, Sakon M. Analysis of Recurrence Patterns After Anatomical or Non-anatomical Resection for Hepatocellular Carcinoma. Ann Surg Oncol. 2015;22:2243-2252.
Shindoh J, Makuuchi M, Matsuyama Y, Mise Y, Arita J, Sakamoto Y, Hasegawa K, Kokudo N. Complete removal of the tumor-bearing portal territory decreases local tumor recurrence and improves disease-specific survival of patients with hepatocellular carcinoma. J Hepatol. 2016;64:594-600.
Shindoh J, Kobayashi Y, Umino R, Kojima K, Okubo S, Hashimoto M. Successful Anatomic Resection of Tumor-Bearing Portal Territory Delays Long-Term Stage Progression of Hepatocellular Carcinoma. Ann Surg Oncol. 2021;28:844-853.
Zhao H, Chen C, Gu S, Yan X, Jia W, Mao L, Qiu Y. Anatomical versus non-anatomical resection for solitary hepatocellular carcinoma without macroscopic vascular invasion: A propensity score matching analysis. J Gastroenterol Hepatol. 2017;32:870-878.
Jiao S, Li G, Zhang D, Xu Y, Liu J, Li G. Anatomic versus non-anatomic resection for hepatocellular carcinoma, do we have an answer? A meta-analysis Int J Surg. 2020;80:243-255.
Zhao H, Ding WZ, Wang H, Gu S, Yan XP, Sun SQ, Mao L, Jin HH, Qiu YD. Prognostic value of precise hepatic pedicle dissection in anatomical resection for patients with hepatocellular carcinoma. Medicine (Baltimore). 2020;99:e19475.
Marubashi S, Gotoh K, Akita H, Takahashi H, Ito Y, Yano M, Ishikawa O, Sakon M. Anatomical versus non-anatomical resection for hepatocellular carcinoma. Br J Surg. 2015;102:776-784.
Feng X, Su Y, Zheng S, Xia F, Ma K, Yan J, Li X, Tang W, Wang S, Bie P, Dong J. A double blinded prospective randomized trial comparing the effect of anatomic versus non-anatomic resection on hepatocellular carcinoma recurrence. HPB (Oxford). 2017;19:667-674.
Hirokawa F, Kubo S, Nagano H, Nakai T, Kaibori M, Hayashi M, Takemura S, Wada H, Nakata Y, Matsui K, Ishizaki M, Uchiyama K. Do patients with small solitary hepatocellular carcinomas without macroscopically vascular invasion require anatomic resection? Propensity score analysis. Surgery. 2015;157:27-36.
Yamamoto T, Yagi S, Uryuhara K, Kaihara S, Hosotani R. Clinical factors that affect the outcomes after anatomical versus non-anatomical resection for hepatocellular carcinoma. Surg Today. 2017;47:193-201.
Kaibori M, Kon M, Kitawaki T, Kawaura T, Hasegawa K, Kokudo N, Ariizumi S, Beppu T, Ishizu H, Kubo S, Kamiyama T, Takamura H, Kobayashi T, Kim DS, Wang HJ, Kim JM, Han DH, Park SJ, Kang KJ, Hwang S, Roh Y, You YK, Joh JW, Yamamoto M. Comparison of anatomic and non-anatomic hepatic resection for hepatocellular carcinoma. J Hepatobiliary Pancreat Sci. 2017;24:616-626.
Miyagawa S, Makuuchi M, Kawasaki S, Kakazu T. Criteria for safe hepatic resection. Am J Surg. 1995;169:589-594.
Strasberg SM. Nomenclature of hepatic anatomy and resections: a review of the Brisbane 2000 system. J Hepatobiliary Pancreat Surg. 2005;12:351-355.
Ueno S, Tanabe G, Nuruki K, Hamanoue M, Komorizono Y, Oketani M, Hokotate H, Inoue H, Baba Y, Imamura Y, Aikou T. Prognostic performance of the new classification of primary liver cancer of Japan (4th edition) for patients with hepatocellular carcinoma: a validation analysis. Hepatol Res. 2002;24:395-403.
Eguchi S, Kanematsu T, Arii S, Okazaki M, Okita K, Omata M, Ikai I, Kudo M, Kojiro M, Makuuchi M, Monden M, Matsuyama Y, Nakanuma Y, Takayasu K. Comparison of the outcomes between an anatomical subsegmentectomy and a non-anatomical minor hepatectomy for single hepatocellular carcinomas based on a Japanese nationwide survey. Surgery. 2008;143:469-475.
Hidaka M, Eguchi S, Okuda K, Beppu T, Shirabe K, Kondo K, Takami Y, Ohta M, Shiraishi M, Ueno S, Nanashima A, Noritomi T, Kitahara K, Fujioka H. Impact of Anatomical Resection for Hepatocellular Carcinoma With Microportal Invasion (vp1): A Multi-institutional Study by the Kyushu Study Group of Liver Surgery. Ann Surg. 2020;271:339-346.
Zhong XP, Zhang YF, Mei J, Li SH, Kan A, Lu LH, Chen MS, Wei W, Guo RP. Anatomical versus Non-anatomical Resection for Hepatocellular Carcinoma with Microscope Vascular Invasion: A Propensity Score Matching Analysis. J Cancer. 2019;10:3950-3957.
Shindoh J, Kawamura Y, Kobayashi Y, Akuta N, Kobayashi M, Suzuki Y, Ikeda K, Hashimoto M. Time-to-Interventional Failure as a New Surrogate Measure for Survival Outcomes after Resection of Hepatocellular Carcinoma. J Gastrointest Surg. 2020;24:50-57.
Johnson PJ, Berhane S, Kagebayashi C, Satomura S, Teng M, Reeves HL, O’Beirne J, Fox R, Skowronska A, Palmer D, Yeo W, Mo F, Lai P, Iñarrairaegui M, Chan SL, Sangro B, Miksad R, Tada T, Kumada T, Toyoda H. Assessment of liver function in patients with hepatocellular carcinoma: a new evidence-based approach-the ALBI grade. J Clin Oncol. 2015;33:550-558.
Haruki K, Shiba H, Saito N, Horiuchi T, Shirai Y, Fujiwara Y, Furukawa K, Sakamoto T, Yanaga K. Risk stratification using a novel liver functional reserve score of combination prothrombin time-international normalized ratio to albumin ratio and albumin in patients with hepatocellular carcinoma. Surgery. 2018;164:404-410.
Imura S, Yamada S, Saito Y, Ikemoto T, Morine Y, Shimada M. Utility of cone unit liver resection for small hepatocellular carcinoma: a propensity score matched analysis. HPB (Oxford). 2020. https://doi.org/10.1016/j.hpb.2020.09.010.
Berardi G, Igarashi K, Li CJ, Ozaki T, Mishima K, Nakajima K, Honda M, Wakabayashi G. Parenchymal Sparing Anatomical Liver Resections With Full Laparoscopic Approach: Description of Technique and Short-term Results. Ann Surg. 2019. https://doi.org/10.1097/SLA.0000000000003575.
Kabashima A, Shimada S, Shimokawa M, Akiyama Y, Tanabe M, Tanaka S. Molecular and immunological paradigms of hepatocellular carcinoma: Special reference to therapeutic approaches. J Hepatobiliary Pancreat Sci. 2021;28:62-75.
Acknowledgements
We would like to thank Editage (www.editage.com) for English language editing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Haruki, K., Furukawa, K., Fujiwara, Y. et al. Effectiveness of Anatomical Resection for Small Hepatocellular Carcinoma: a Propensity Score–Matched Analysis of a Multi-institutional Database. J Gastrointest Surg 25, 2835–2841 (2021). https://doi.org/10.1007/s11605-021-04985-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-021-04985-4