Abstract
Background and Aim
Two-stage hepatectomy (TSH) with or without portal vein ligation (PVL) or portal vein embolization (PVE) and associated liver partition and portal vein ligation for staged hepatectomy (ALPPS) are surgical strategies in the treatment of advanced colorectal liver metastases (CRLM). The role of each strategy is yet ill defined. The aim of this analysis is to share our center experience with conventional TSH with or without PVL/PVE and ALPPS in patients with advanced bilateral CRLM.
Patients and Methods
Data were extracted from a prospectively collected institutional database. Complication rates according to the Dindo-Clavien classification, overall and recurrence-free survival data were analyzed.
Results
Between 2008 and 2017, 790 liver resections were performed in 611 patients with CRLM. Out of 320 patients with bilateral disease, TSH (as right or extended right hepatectomy) with or without PVL/PVE was performed in 50 patients and ALPPS in 8. Stage 2 was completed in 36 (72%) out of 50 TSH/PVL/PVE and in all ALPPS patients (100%). Median follow-up was 15.8 months (0.9 to 111.9 months). On an intention-to-treat basis, the median overall survival was 26.7 (21.8–35.1 range) months after TSH/PVL/PVE and 36.2 months (11.3–61.2 range) after ALPPS (p = 0.809). In the TSH/PVL/PVE cohort, the median overall survival was 29.9 (19.0–40.3) months in patients who completed stage 2 compared to 13.8 months in patients who did not (p < 0.001). Disease recurred in 60% in the TSH/PVL/PVE cohort and in 87.5% in the ALPPS cohort (p = 0.777). The median recurrence-free survival was 5.9 (1.7–18.6) months after TSH/PVL/PVE and 3 (1.6–14.8) months after ALPPS (p = 0.680).
Conclusion
The treatment of advanced bilateral CRLM remains a surgical and oncological challenge. A tailored approach to bilateral CRLM uses TSH/PVL/PVE as first and ALPPS as second rescue treatment in order to achieve resectability in patients with extensive tumor burden not amenable to one-stage resection. ALPPS should be reserved for patients with no other surgical options.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hepatic resection is the current standard of care in the treatment of colorectal liver metastases (CRLM). Depending on patient selection, 5-year survival rates of up to 50–60% can be reached. At the time of diagnosis, however, only 15–30% of patients are candidates for upfront liver surgery. The majority of patients have either oncological or functional contraindications to liver resection due to a too extensive, often bilateral hepatic tumor burden or a too small liver remnant (future liver remnant (FLR)).1,2,3,4,5,6 To overcome this high rate of primary hepatic irresectability, advances in modern chemotherapy of CRLM including targeted therapy have been able to downsize hepatic lesions offering a chance of secondary resectability in a substantial number of patients. On the other hand, new surgical strategies have been proposed to address the problem of the small (FLR).
Two-stage hepatectomy (TSH) has been implemented with or without portal vein ligation or intermittent portal vein embolization to induce hypertrophy of the remnant liver. TSH has been shown to be safe but bears a substantial risk of dropout during the long interval between stages 1 and 2 due to tumor progression or insufficient hypertrophy of the FLR.7,8,9,10,11,12,13,14,15 The concept of associated liver partition and portal vein ligation for staged hepatectomy (ALPPS) was found to induce a much more efficient and rapid hepatic hypertrophy allowing R0 resections in almost all patients, however, at the expense of a high rate of early recurrences.16,17,18,19
Pushing the limits with these complex surgical strategies in patients with extensively pretreated livers, the role of each strategy in the management of CRLM is yet ill defined. The aim of this analysis is to share our center experience with conventional TSH with or without portal vein ligation (PVL)/portal vein embolization (PVE) and ALPPS in patients with advanced bilateral CRLM.
Patients and Methods
For this analysis, we selected all patients with bilateral CRLM who underwent a staged hepatectomy at our center between January 2008 and December 2017. Data were extracted from a prospectively collected institutional CRLM surgery database. Follow-up was concluded in June 2018.
Institutional Approach to Bilateral Liver Metastases
Bilateral CRLM were resected in a single session (one-stage hepatectomy (OSH)) whenever both volume and function of the FLR were considered sufficient. When FLR function was deemed critical (mostly due to prior extensive chemotherapy), conventional TSH was planned. In cases with a FLR/BW ratio below 0.5, TSH with intermittent PVE or PVL was planned. ALPPS was considered a last resort either for patients with an expected inadequate hypertrophy of the FLR following PVL/PVE (assumed necessary hypertrophy rate > 60%) or in cases presenting with technically critical circumstances such as close proximity of the CRLM to vital vascular or biliary structures of the FLR (in particular when partitioning of the liver at first-stage hepatectomy seemed to be advisable to reduce the risk of tumor invasion into the FLR.)
In either conventional TSH or ALPPS, the FLR was freed from all visible metastases.
Volumetric Analyses
Volumetric analyses were performed using Aquarius © Terarecon. Inc. Intuition Edition Version 4.4.12. Volume increases after stage 1 or after PVE were evaluated 4–6 weeks after the procedure/intervention. The second stage was scheduled as soon as the observed hepatic hypertrophy had led to an FLR/BW ratio > 0.5.
Conventional Two-Stage Hepatectomy
The conventional TSH group comprised all patients with TSH alone, TSH with PVL during stage 1, and TSH with intermittent PVE.
As pointed out before, right portal vein ligation or embolization was added whenever the functional FLR was considered too little. PVL was performed during the first-stage hepatectomy. If necessary, a partial hilar lymphadenectomy was carried out in order to expose the right portal vein which was then divided between clamps and both stumps sutured.
PVE was performed by interventional radiology in a subsequent session after a median of 7 days (5–10 days). The procedure was carried out under local anesthesia by a contralateral approach to the portal vein and antegrade embolization using a mixture of ethiodized oil and N-butyl cyanoacrylate glue.
Associating Liver Partition and Portal Vein Ligation for Staged Hepatectomy
Complete parenchymal transection was performed along the falciforme ligament during the first stage (no partial ALPPS was performed). In four cases, the FLR was freed from metastatic disease (1–3 nodes) by atypical parenchyma-sparing resections. Hilar lymphadenectomy was performed routinely. In order to avoid adhesions, both transection surfaces were covered with hemostatic patches (TachoSil™) at the end of stage 1. After volumetric confirmation of a sufficient hypertrophy of the FLR, stage 2 was scheduled after a median of 7 days after stage 1.
Morbidity and Mortality
Surgical complications were assessed according to the Dindo/Clavien classification20 and documented separately for each stage. Postoperative liver dysfunction was assessed by Vauthey’s criteria which define an increase of serum bilirubin above 7 mg/dL at any time point as liver insufficiency.21
Mortality was determined as either in-hospital or 90-day mortality calculated from the day of the respective stage of surgery.
Statistical Analysis
Prognostic factors for all cohorts were compared by the modified clinical risk score, which is based on N-stage of the primary tumor, size of metastases > 5 cm, and k-ras status.22 Differences between groups were compared by χ2 test and Student’s t test as appropriate.
Recurrence-free and overall survival was defined as the period from the first stage of surgery until the date of recurrence or death. Patients who did not complete the second stage of the two-stage procedure were excluded from the analysis. Patients with macroscopic tumor residues (R2 resection) were excluded from the analysis of recurrence-free survival.
Categorical data were compared using χ2 test and continuous data of normally distributed data by the Fisher exact test. The Mann-Whitney U test was used to compare medians of data without normal distribution. Survival analyses were performed using the Kaplan-Meier method, and the log-rank test was used to compare median survivals between groups. P values < 0.05 were considered significant.
Results
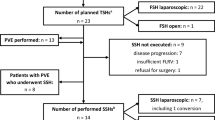
During the study period, a total of 611 patients underwent 790 liver resections for CRLM at our center (Fig. 1). Out of 320 patients with bilateral disease, 63 were scheduled for TSH according to the abovementioned algorithm with 58 of them requiring a right hepatectomy or right trisectionectomy. These 58 patients were included in this analysis (Table 1). The remaining patients underwent one single liver resection for bilobar metastases without previous portal vein manipulations.
Of the 58 patients, 50 underwent conventional TSH (16 with neither PVL nor PVE, 14 with intermittent PVE, and 20 with PVL during stage 1), and 8 patients had an ALPPS procedure. The baseline characteristics of the 58 patients did not differ in between groups (Table 1). Ninety-one percent (53/58) of patients had been treated with chemotherapy with additional targeted therapy (78%) before liver surgery. Reasons for no chemotherapy prior to the first stage were simultaneous resection of the primary cancer and the liver metastases (n = 1), previous local ablative therapy (n = 2), and refusing chemotherapy by patient (n = 2).
Synchronous resection of the CRC primary was performed in seven cases during the first step in the TSH/PVL/PVE group. A liver-first approach was chosen in 2 patients in the TSH/PVL/PVE group. ALPPS was always performed after resection of the CRC primary.
In the TSH/PVL/PVE group, a median of three metastases (range 1–7) was removed from the FLR during stage 1 compared to a median of one metastasis (range 0–3) in the ALPPS group (p = 0.005).
In the TSH/PVL/PVE group, there were 7 patients with extrahepatic tumor at the time of liver resection (lung metastases in five and primary tumor in two cases) while no ALPPS patient had extrahepatic disease (p = 0.358).
The majority of patients had a clinical risk score ≥ 2. The groups did not differ regarding k-ras status or clinical risk scores (Table 1).
Portal Vein Interventions
PVE was performed by interventional radiology without any complication. There was no observed morbidity from PVL during stage 1, either. The time intervals between PVL or PVE and stage 2 did not differ significantly (58 days after PVE vs 70 days after PVL, p = 0.210).
Stage-Two Procedures
Of the entire cohort of patients, 47 (81%) proceeded to stage 2 and 44 completed the staged hepatectomy.
In the TSH/PVL/PVE group, 39 (78%) out of 50 patients went into stage 2. Reasons for not proceeding to stage 2 in this group (n = 11) were tumor progress (n = 8; TSH/PVL/PVE 4/3/1), poor general condition (TSH n = 1), cardiac decompensation/arrest (TSH n = 1), and liver failure (PVL n = 1). Out of these 39 patients, hepatectomy could be completed in 36 patients (72%) as a right hepatectomy (n = 25) or right trisectionectomy (n = 11). Reasons for not completing stage 2 were the intraoperative diagnosis of multiple paracaval lymph node metastases (n = 1) or newly occurred liver lesions in the FRL (n = 2) which were not detectable in the preoperative CT scan.
In the ALPPS group, stage 2 was performed after a median of 7 days (range 6–11, IQR 1.5) and completed in all patients (100%).
The extent of resection was significantly higher in the ALPPS group (right trisectionectomies only; + segment I (n = 4)) than in the TSH/PVL/PVE group (11/36 right trisectionectomies) (p = 0.007).
In the ALPPS group, all resections were R0 resections while there were 35 R0 resections and one R1 resection in the group of completed TSH (Table 2).
While 14/50 patients (28%) in the TSH/PVE/PVL group did not have complete tumor clearance (R2), none of the ALPSS patients had incomplete tumor clearance (p = 0.09).
Morbidity and Mortality
The perioperative outcome of the first and second stages is listed in Table 3 (only complications grade III or higher are reported). The in-hospital mortality was 3.4% (2/58; 1/50 after TSH and 1/8 ALPPS each), and the 90-day perioperative mortality was 5.2% (3/58; 2 after TSH and 1 after ALPPS).
In the conventional TSH group, one patient died after stage 1 from cardiac decompensation and another after PVL on day 90 due to liver failure. There was no mortality after stage 2.
In the ALPPS group, one patient died on postoperative day 53 from iatrogenic duodenal perforation and inferior vena cava injury related to endoscopic retrograde cholangiography for persistent bile leak. Up to this point, the patient had no signs of liver insufficiency but, however, evidence of intrahepatic tumor recurrence.
There was no significant difference in the postoperative complication rate between TSH/PVL/PVE and ALPPS.
Liver Failure
Postoperative liver failure occurred in six patients after TSH (n = 1)/PVL (n = 3)/PVE (n = 2) after the first stage. Two of them failed to reach the second stage. The initial FLR/BW ratios in these two patients had been 0.3 and 0.4. One of these patients (PVL) died on day 90 from persistent liver failure. Liver histology was unremarkable in one patient and showed signs of mild fibrosis in the other.
After the second stage, four patients developed bilirubin serum levels above 7 mg/dL within a median interval of 12 days (range 5–21). These patients fully recovered from liver insufficiency. Histology of the resected specimen revealed mild steatosis in two patients, while parenchyma was normal in the other two.
No postoperative liver insufficiency occurred after ALPPS (p = 0.159).
Overall Survival
At the time of this analysis, 23 patients (46%) are alive after TSH with or without PVE/PVL and 2 after ALPPS (25%). The median follow-up was 15.8 months (range 0.9–111.9) and no patient was lost to follow-up.
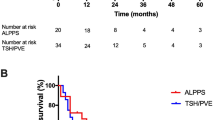
On an intention-to-treat basis, the median overall survival after TSH/PVE/PVL was 26.7 months [CI 21.8–35.1 months] and did not differ significantly from the median overall survival of patients who underwent an ALPPS procedure with 36.2 months (CI 11.3–61.2 months) (p = 0.809) (Fig. 2).
On a per protocol basis, overall survival of patients who completed the two-stage concept (n = 36) was significantly longer (29.9 months, range 19.0–40.3) compared to patients who had only stage 1 (n = 11) or an incomplete second stage (n = 3) (13.8 months, p < 0.001) (Figs. 3 and 4).
Recurrence
Tumor recurred in a total of 78.7%. The recurrence rate was higher after ALPPS than after completed TSH with or without PVE/PVL (87.5% vs 60%, p = 0.777).
The median recurrence-free survival of the TSH/PVL/PVE cohort was 5.9 months (range 1.7–18.6), while it was 3 months (range 1.6–14.8) for the ALPPS group (p = 0.680) (Fig. 5).
Discussion
TSH ± PVL/PVE and ALPPS are accepted surgical procedures in the treatment of bilateral CRLM. In order to find their optimal role and field of application, one has to reflect the advantages and disadvantages of each technique in relation to the patient’s performance status and his individual oncologic prognosis. Considering the success of modern chemotherapy and these pros and cons of two-stage procedures, we have established a policy at our institution, which is mainly based on patient safety. Our aim is to resect all metastases during one procedure by accepting narrow resection margins. If the volume and estimated functional capacity of the future liver remnant are too small, a classical two-stage procedure is initiated. Depending on the estimated tumor biology, these procedures are embedded into chemotherapy concepts. Only, if TSH/PVE/PVL fail or are assumed not to be efficient enough to achieve the required increase in liver volume, an ALPPS procedure is performed.
A major problem of TSH ± PVL/PVE is the risk of dropout between stages 1 and 2. In our cohort, 22% of patients failed to proceed to the second stage. This result is slightly better than reported data from other series with dropouts ranging from 25 to 38%.23 Reasons for dropout in our series were progressive disease and persistent post stage 1 liver dysfunction. We did not observe any case of insufficient hypertrophy of the FLR which is one of the main reasons for failure of TSH concepts in the literature.24,25 In our series, the ALPPS group had a higher completion rate than the TSH group which is concordant with published data. The high completion rate in ALPPS is important as patients not proceeding to stage 2 resection are known to have a poor prognosis with survival rates even lower than in patients treated with chemotherapy alone. Thus, the completion of stage 2 resection is of major importance in either two-stage concept (Fig. 6).8,23,24,26,27,28
Similarly to other studies, we also observed a slightly but not significant higher complication rate in our ALPPS cohort. In contrast, five patients in the TSH± group experienced liver insufficiency which was fatal in two cases. Interestingly, we observed liver insufficiency twice after the first stage of the TSH± concept, which is an unusual finding.15,27 The reasons for post stage 1 liver insufficiency remain unclear since we could not identify any differences regarding the perioperative, histopathological, or oncological characteristics of the affected patients in comparison to the entire patient cohort. In contrast, no postoperative liver failure occurred after ALPPS. A recently published meta-analysis by Moris et al. still accounted a higher morbidity and mortality in ALPPS than in conventional TSH.29 Also, in our series, the mortality also appeared higher after ALPPS (1/8 vs 2/50), but we could not test for significance due to the low number of patients. Noteworthy, in our series, the extent of liver resection differed significantly between both cohorts. In the ALPPS group, we only performed right trisectionectomies with an additional resection of segment I and excision of 1–3 metastases out of the FLR in four cases, each. In contrast, in the TSH±, only 11 patients (28%) out of 39 received a right trisectionectomy.
Next to two-stage procedures, parenchymal-sparing liver resections in one-stage hepatectomy (OSH) are an accepted alternative to extended hepatectomies. In this concept, major anatomical resections are avoided if possible, and CRLM are mainly removed by atypical resections. This approach minimizes parenchymal loss but at a higher risk of tumor-positive resection margins. However, although at first glance this is in contradiction to basic rules of oncologic surgery, in recent years, R1 resection has become more and more accepted in the treatment of CRLM, in particular in multifocal CRLM with good response to chemotherapy. Even more so, the detachment of metastases from major vascular structures (R1 vascular margin) has been shown to be almost equivalent to R0 resection.30,31,32,33 We favor OSH and parenchymal-sparing resections when possible and perform TSH± and ALPPS only in patients with high-risk features for postoperative liver dysfunction. This is reflected by the fact that we chose a two-stage approach in only about 20% of patients with bilateral CRLM.8,11,27,34,35,36
When comparing our results after conventional TSH± PVL/PVE and ALPPS with published data, we have to emphasize again that in our institutional approach, TSH and especially ALPPS are considered a last resort which implies a negative oncological selection. Accordingly, 48% of two-stage and 63% of ALPPS patients had high CRS (2–3), but did not differ from each other. We perform any two-stage procedure very selectively and ALPPS only when there is no surgical alternative. This explains the 100% trisectionectomy rate in our cohort. This is a most striking difference in comparison to data of the first randomized controlled trial comparing ALPPS vs TSH/PVE, in which ALPPS was significantly superior with regard to resection rate, but no difference in the perioperative complication and 90-day mortality rate was found. Oncologic long-term data were not given.37 But in this trial, the majority of TSH and also of ALPPS were right hepatectomies only. Our patients all had extensive chemotherapy which is another important difference to most other data from the literature with a more liberal indication of ALPPS.38 In the light of this and considering that ALPPS is a last resort in our institutional approach, the oncological outcome of ALPPS for CRLM needs to be compared to the outcome after palliative chemotherapy. And here, our median survival of 36.2 months for the ALPPS group seems to be considerably better than the median overall survival after palliative treatment (currently about 25 months) and even more so, if not the date of surgery but the start of preoperative chemotherapy is considered which adds another 6 to 7 months resulting in a median survival after ALPPS of about three and a half years.
Conclusion
A tailored approach to bilateral CRLM, as proposed by our series, uses TSH± as first and ALPPS as second rescue treatment in order to achieve resectability in patients with extensive bilateral tumor not amenable to one-stage resection. In our thinking, ALPPS does not replace other surgical strategies but adds up to the armamentarium of the experienced liver surgeon. As long as robust data regarding oncological outcome are missing, standard TSH with or without PVL/PVE and ALPPS might be complementary strategies for resection of CRLM. ALPPS should be reserved for patients with no other surgical option, i.e., after failed PVE or those with an extremely small FLR. In these individual situations and always embedded into a multimodal treatment setting, ALPPS may still offer a chance of complete tumor removal, prolonged survival, and even a chance for cure.
References
Neumann, U.P., D. Seehofer, and P. Neuhaus, The surgical treatment of hepatic metastases in colorectal carcinoma. Dtsch Arztebl Int, 2010. 107(19): p. 335–42.
Adam, R., et al., Patients with initially unresectable colorectal liver metastases: is there a possibility of cure? J Clin Oncol, 2009. 27(11): p. 1829–35.
Wicherts, D.A., R.J. de Haas, and R. Adam, Bringing unresectable liver disease to resection with curative intent. Eur J Surg Oncol, 2007. 33 Suppl 2: p. S42–51.
Clavien, P.A., et al., Strategies for safer liver surgery and partial liver transplantation. N Engl J Med, 2007. 356(15): p. 1545–59.
Lang, H., J. Baumgart, and J. Mittler, Associating Liver Partition and Portal Vein Ligation for Staged Hepatectomy in the Treatment of Colorectal Liver Metastases: Current Scenario. Dig Surg, 2018. 35(4): p. 294–302.
Folprecht, G., et al., Neoadjuvant treatment of unresectable colorectal liver metastases: correlation between tumour response and resection rates. Ann Oncol, 2005. 16(8): p. 1311–9.
Adam, R., et al., Two-stage hepatectomy: A planned strategy to treat irresectable liver tumors. Ann Surg, 2000. 232(6): p. 777–85.
Brouquet, A., et al., High survival rate after two-stage resection of advanced colorectal liver metastases: response-based selection and complete resection define outcome. J Clin Oncol, 2011. 29(8): p. 1083–90.
Cardona, K., et al., Treatment of extensive metastatic colorectal cancer to the liver with systemic and hepatic arterial infusion chemotherapy and two-stage hepatic resection: the role of salvage therapy for recurrent disease. Ann Surg Oncol, 2014. 21(3): p. 815–21.
Jaeck, D., et al., A two-stage hepatectomy procedure combined with portal vein embolization to achieve curative resection for initially unresectable multiple and bilobar colorectal liver metastases. Ann Surg, 2004. 240(6): p. 1037–49; discussion 1049-51.
Wicherts, D.A., et al., Long-term results of two-stage hepatectomy for irresectable colorectal cancer liver metastases. Ann Surg, 2008. 248(6): p. 994–1005.
Heinrich, S. and H. Lang, Liver metastases from colorectal cancer: technique of liver resection. J Surg Oncol, 2013. 107(6): p. 579–84.
Ratti, F., et al., Strategies to Increase the Resectability of Patients with Colorectal Liver Metastases: A Multi-center Case-Match Analysis of ALPPS and Conventional Two-Stage Hepatectomy. Ann Surg Oncol, 2015. 22(6): p. 1933–42.
Passot, G., et al., Predictors of Safety and Efficacy of 2-Stage Hepatectomy for Bilateral Colorectal Liver Metastases. J Am Coll Surg, 2016. 223(1): p. 99–108.
Vigano, L., et al., Drop-out between the two liver resections of two-stage hepatectomy. Patient selection or loss of chance? Eur J Surg Oncol, 2016. 42(9): p. 1385–93.
Schnitzbauer, A.A., et al., Right portal vein ligation combined with in situ splitting induces rapid left lateral liver lobe hypertrophy enabling 2-staged extended right hepatic resection in small-for-size settings. Ann Surg, 2012. 255(3): p. 405–14.
Lang, H., ALPPS for Colorectal Liver Metastases. J Gastrointest Surg, 2017. 21(1): p. 190–192.
Oldhafer, K.J., et al., ALPPS for patients with colorectal liver metastases: effective liver hypertrophy, but early tumor recurrence. World J Surg, 2014. 38(6): p. 1504–9.
Hernandez-Alejandro, R., et al., Can we improve the morbidity and mortality associated with the associating liver partition with portal vein ligation for staged hepatectomy (ALPPS) procedure in the management of colorectal liver metastases? Surgery, 2015. 157(2): p. 194–201.
Dindo, D., N. Demartines, and P.A. Clavien, Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg, 2004. 240(2): p. 205–13.
Mullen, J.T., et al., Hepatic insufficiency and mortality in 1,059 noncirrhotic patients undergoing major hepatectomy. J Am Coll Surg, 2007. 204(5): p. 854–62; discussion 862-4.
Brudvik, K.W., et al., RAS Mutation Clinical Risk Score to Predict Survival After Resection of Colorectal Liver Metastases. Ann Surg, 2017.
Adam, R., et al., Outcome after associating liver partition and portal vein ligation for staged hepatectomy and conventional two-stage hepatectomy for colorectal liver metastases. Br J Surg, 2016. 103(11): p. 1521–9.
Simoneau, E., et al., Portal vein embolization and its effect on tumour progression for colorectal cancer liver metastases. Br J Surg, 2015. 102(10): p. 1240–9.
Giuliante, F., et al., Tumor progression during preoperative chemotherapy predicts failure to complete 2-stage hepatectomy for colorectal liver metastases: results of an Italian multicenter analysis of 130 patients. J Am Coll Surg, 2014. 219(2): p. 285–94.
Abdalla, E.K., et al., Resection of hepatic colorectal metastases involving the caudate lobe: perioperative outcome and survival. J Gastrointest Surg, 2007. 11(1): p. 66–72.
Narita, M., et al., Two-stage hepatectomy for multiple bilobar colorectal liver metastases. Br J Surg, 2011. 98(10): p. 1463–75.
Imai, K., et al., Failure to Achieve a 2-Stage Hepatectomy for Colorectal Liver Metastases: How to Prevent It? Ann Surg, 2015. 262(5): p. 772–8; discussion 778-9.
Moris, D., et al., Operative Results and Oncologic Outcomes of Associating Liver Partition and Portal Vein Ligation for Staged Hepatectomy (ALPPS) Versus Two-Stage Hepatectomy (TSH) in Patients with Unresectable Colorectal Liver Metastases: A Systematic Review and Meta-Analysis. World J Surg, 2018. 42(3): p. 806–815.
Vigano, L., et al., Is Tumor Detachment from Vascular Structures Equivalent to R0 Resection in Surgery for Colorectal Liver Metastases? An Observational Cohort. Ann Surg Oncol, 2016. 23(4): p. 1352–60.
Vigano, L., et al., R1 Resection for Colorectal Liver Metastases: a Survey Questioning Surgeons about Its Incidence, Clinical Impact, and Management. J Gastrointest Surg, 2018.
Hosokawa, I., et al., Long-Term Survival Benefit and Potential for Cure after R1 Resection for Colorectal Liver Metastases. Ann Surg Oncol, 2016. 23(6): p. 1897–905.
Torzilli, G., et al., Is Enhanced One-Stage Hepatectomy a Safe and Feasible Alternative to the Two-Stage Hepatectomy in the Setting of Multiple Bilobar Colorectal Liver Metastases? A Comparative Analysis between Two Pioneering Centers. Dig Surg, 2018. 35(4): p. 323–332.
Karoui, M., et al., Combined first-stage hepatectomy and colorectal resection in a two-stage hepatectomy strategy for bilobar synchronous liver metastases. Br J Surg, 2010. 97(9): p. 1354–62.
Pamecha, V., et al., Prospective evaluation of two-stage hepatectomy combined with selective portal vein embolisation and systemic chemotherapy for patients with unresectable bilobar colorectal liver metastases. Dig Surg, 2008. 25(5): p. 387–93.
Vigano, L., et al., Early recurrence after liver resection for colorectal metastases: risk factors, prognosis, and treatment. A LiverMetSurvey-based study of 6,025 patients. Ann Surg Oncol, 2014. 21(4): p. 1276–86.
Sandstrom, P., et al., ALPPS Improves Resectability Compared With Conventional Two-stage Hepatectomy in Patients With Advanced Colorectal Liver Metastasis: Results From a Scandinavian Multicenter Randomized Controlled Trial (LIGRO Trial). Ann Surg, 2018. 267(5): p. 833–840.
Schnitzbauer, A.A., et al., Indicating ALPPS for Colorectal Liver Metastases: A Critical Analysis of Patients in the International ALPPS Registry. Surgery, 2018.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Baumgart, J., Jungmann, F., Bartsch, F. et al. Two-Stage Hepatectomy and ALPPS for Advanced Bilateral Liver Metastases: a Tailored Approach Balancing Risk and Outcome. J Gastrointest Surg 23, 2391–2400 (2019). https://doi.org/10.1007/s11605-019-04145-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-019-04145-9