Abstract
Background
Although remnant gastric cancer (RGC) following distal gastrectomy is located in the proximal stomach, little is known about the differences of the lymphatic distribution and surgical outcomes between RGC and primary proximal gastric cancer (PGC).
Methods
Between 1997 and 2008, 1,149 patients underwent gastrectomy for gastric cancer. Of these, 33 (2.9%) RGC patients and 207 (18.5%) PGC patients were treated at our department. We reviewed their hospital records retrospectively.
Results
Compared with the PGC patients, those with RGC had a slightly higher age at onset (p = 0.09), higher incidence of undifferentiated cancer (p = 0.06), higher incidence of vascular invasion (p = 0.09), and higher incidence of T4 (p = 0.07). Gastrectomy for RGC involved greater blood loss (p < 0.005), longer surgical duration (p = 0.01), combined resection, and high incidence of complications. However, the survival rate for RGC patients was similar to that for PGC patients (p = 0.67). 2) Patients with RGC had a different pattern of lymph node metastasis compared with that in PGC. Particularly in advanced RGC with pT2–T4 tumors, RGC frequently demonstrated jejunal mesentery lymph node metastases (RGC vs. PGC, 35% vs. 0%) and splenic hilar lymph node metastases (RGC vs. PGC, 17% vs. 10%). The jejunal mesentery lymph node metastases were detected only following Billroth II reconstruction (Billroth I vs. Billroth II, 0% vs. 67%).
Conclusion
Although the clinical behaviors of the two gastric cancers were different, the survival rates were similar. The pattern of metastasis indicates that the jejunal mesentery and splenic hilar lymph nodes should be specifically targeted for en bloc resection during complete gastrectomy in RGC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gastric cancer is the second leading cause of cancer-related death in the world1. However, recent advances in diagnostic methods, less invasive treatment techniques, and perioperative management have increased the early detection of gastric cancer and decreased the mortality and morbidity rates.2–4 Consequently, the number of successfully treated patients has been increasing, and some of these patients are at risk of developing second primary cancer in the remnant stomach. This implies that more cases of remnant gastric cancer (RGC) will be encountered in the future.
In previous studies, RGC was commonly found at an advanced stage, resulting in low rates of curative resection (38–40%) and a consequent poor prognosis.5,6 However, recently, the prognosis of RGC following distal gastrectomy has been improving due to diagnostic and technological advances. Indeed, at our institute, more than half of the RGC patients were treated for T1 or T2, node-negative, and early stage cancer and almost 80% of patients with RGC underwent curative resection. Therefore, it is necessary to characterize the clinical features of RGC to develop optimal surgical and treatment planning. However, there is limited information available to help guide the treatment of patients with RGC. RGC after distal gastrectomy is located in the same proximal stomach as a primary proximal gastric cancer (PGC). This study was designed to clarify the differences of the lymphatic distribution and surgical outcomes between RGC and PGC.
Patients and Methods
Patients
Between 1997 and 2008, 1,149 patients underwent gastrectomy for gastric cancer. Of these, 33 consecutive patients with primary RGC (2.9%) and 207 patients with PGC (18.5%) were treated in the Department of Digestive Surgery, Kyoto Prefectural University of Medicine. The follow-up program after gastrectomy consisted of regular physical examinations and laboratory blood tests, chest X-rays, an upper gastrointestinal series or endoscopy, and ultrasonography or computer tomography for the first 5 years and yearly endoscopy thereafter if possible. All patients underwent gastrectomy with lymphadenectomy for RGC. The clinicopathological findings of these patients were determined retrospectively on the basis of their hospital records. Macroscopic and microscopic classifications of gastric cancers were based on the Japanese Classification of Gastric Carcinomas.7 Consistent with the TNM staging system,8 patients with lymph node metastases were reclassified into three groups based on the total number of positive nodes. Histological types were classified as differentiated (papillary, moderately, or well-differentiated adenocarcinoma) and undifferentiated (poorly or undifferentiated adenocarcinoma, signet-ring cell carcinoma, and mucinous adenocarcinoma).
Comparison of associated clinical factors between RGC and PGC patients
Comparison between RGC and PGC was performed because RGC was located in the proximal stomach. To examine treatment targets in particular, several clinicopathological factors such as age, sex, histological types, lymphatic invasion, venous invasion, tumor size, depth of tumor, area and number of lymph node metastases, pathological stage, and surgical factors such as surgical duration, bleeding amount, organs with combined resection, and complications were retrospectively evaluated from the hospital records.
Statistical Analysis
Cause-specific death was recorded when the cause of death was specified as recurrent RGC. Chi-square test and Fisher’s exact probability test were performed for categorical variables, while Student’s t test and Mann–Whitney U test for unpaired data of continuous variables were performed to compare the clinicopathological characteristics between two groups. The cumulative cause-specific overall survival rates were calculated using the Kaplan–Meier method, and log rank test was used for assessment of differences between clinical factors. A p < 0.05 was considered significant.
Results
Clinicopathological characteristics of patients with primary remnant gastric cancer
Table 1 shows the characteristics of the 33 RGC patients. The mean patient age was 68 years, and the male/female ratio was 2.7:1. Ten patients had symptoms and the remaining 23 patients were asymptomatic. Regarding the initial gastric disease, there were 19 patients with benign disease and 14 patients with gastric cancer. Reconstruction during the first surgery was mainly Billroth I or II. More than half of the RGC patients demonstrated T1 or T2, undifferentiated, node-negative, and early stage cancer. In 78.8% (26/33) of the patients, resections were performed with curative intent. En bloc resection of the tumor by total remnant gastrectomy was performed with jejunal mesentery and D2 lymphadenectomy and concomitant organ resection. In addition, splenectomy was performed in 18 patients, distal pancreatectomy in four, partial colon resection in two, and liver resection in two. Reconstruction was performed in 16 patients by Billroth I, in 16 patients by Billroth II, and in one by Roux-en Y procedure for all resected RGC tumors. Tumors were located at the anastomotic site in 16 (61%) patients, corpus and/or cardia in nine (34%), and throughout the entire remnant in one (4%). The median interval between the first and second surgery was 20 years. Of the 33 RGC patients, RGC was detected in 19 (58%) by routine screening in whom the follow-up periods were short (0.5–2 year). On the other hand, RGC was detected incidentally in 14 (42%) patients in whom the follow-up periods were more than 5 years. Patients with early stage RGC such as stages I and II tended to have been diagnosed every second year (data not shown). Therefore, surveillance endoscopic screening following distal gastrectomy should be performed every second year for at least 20 years.
Primary Remnant Gastric Cancer and Upper One Third Gastric Cancers
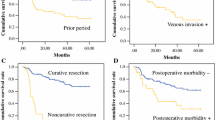
Table 2 shows a comparison of clinicopathological factors between the 33 RGC and 207 primary PGC. RGC patients had a slightly higher age at onset (p = 0.09), higher incidence of undifferentiated cancer (p = 0.06), higher incidence of vascular invasion (p = 0.09), and higher incidence of T4 (p = 0.07) than those with PGC. Gastrectomy for RGC involved greater blood loss (p < 0.005), longer surgical duration (p = 0.01), combined resection, and high incidence of complications. As shown in Fig. 1, the survival curves for the two groups were similar. Figure 2 shows the metastatic region and extent of lymph node involvement between RGC and PGC. Particularly in advanced RGC with pT2–T4 tumors, compared with PGC, RGC more frequently demonstrated jejunal mesentery lymph node metastasis (RGC vs. PGC, 35% vs. 0%) and splenic hilar lymph node metastasis (RGC vs. PGC, 17% vs. 10%) because RGC had a different pattern of lymphatic flow after initial distal gastrectomy. The jejunal mesentery lymph node metastases were detected only following Billroth II reconstruction (Billroth I vs. Billroth II, 0% vs. 67%).
Comparison of the metastatic region and extent of lymph node metastasis between RGC and PGC. Compared with that in PGC, RGC frequently showed jejunal mesentery lymph node metastasis and splenic hilar lymph node metastasis because RGC has a different pattern of lymphatic flow after the initial distal gastrectomy
Discussion
RGC following distal gastrectomy has been reported to account for 1–2% of all gastric cancers in Japan.9,10 Previously, RGC was reported to be caused by multiple factors, and the incidence, pathological features, and potential mechanisms have been extensively investigated.11–13 RGC is commonly found at an advanced stage, resulting in low rates of curative resection (38–40%) and a consequently poor prognosis.5,6 However, recently, the incidence and etiology of RGC have been changing14 because of the long latency periods, decreasing prevalence of gastrectomy for benign disease,5,15 early detection, and improved outcomes in patients with gastric cancers.16,17 Moreover, recent advances in diagnostic and treatment techniques have led to a higher detection rate of early RGC following distal gastrectomy.18 Consequently, endoscopic therapy such as EMR or ESD is performed for treatment of early stage RGC19,20. Therefore, it is important to re-evaluate the clinical features of RGC in order to develop optimal surgical and treatment planning.
In comparison between RGC and primary PGC, the two survival rates were similar (Fig. 1) even though RGC showed a slightly higher involvement of jejunal mesenteric or splenic hilar nodes and a higher incidence of undifferentiated cancer, vascular invasion, and T4 cancers, and gastrectomy for RGC involved more blood loss, longer surgical duration, combined resection, and a higher incidence of complications than PGC (Table 2). These results are consistent with several recent reports;6,21,22 however, the reason for this similarity remains unclear. One of reasons might be that the incidence and etiology of RGC following distal gastrectomy have been changing owing to diagnostic and technological advances, although previously RGC was commonly found at more advanced stage, resulting in low rates of curative resection (38–40%) and a consequent poor prognosis. Indeed, at our department, more than half of the RGC patients were treated for T1 or T2, node-negative, and early stage cancer, in contrast to that in previous series (Table 1) and almost 80% of patients underwent curatively resection with extensive lymphadenectomy. On the contrary, in recent years, the incidence of PGC has been increasing, whereas the prognosis of PGC has not been improved in comparison with middle and lower gastric cancers (data not shown). Therefore, the prognoses of RGC and PGC might be similar at this point in time. Indeed, RGC is not always advanced at diagnosis, and if it is, extensive surgery for RGC is not necessarily associated with poor prognosis in comparison to that for primary gastric cancer.
Concerning lymph node metastasis from RGC, the main lymphatic flow from a tumor located in the upper one third of the stomach drains into lymph nodes along the celiac artery through the lymph nodes at the lesser curvature, the right side of cardia, and the left gastric artery. In the remnant stomach, these lymphatic pathways have been transected during the initial surgery, thus altering the lymphatic flow at the greater curvature, splenic artery, and splenic hilum.5,23 Indeed, patients with RGC have a different pattern of lymph node metastasis compared with that in PGC (Fig. 2). Regarding the ligation of the left gastric artery, of the 33 RGC patients analyzed, 14 (42%) patients underwent the initial gastrectomy for gastric cancer and all left gastric arteries were ligated. On the other hand, there were no patients who underwent ligation of the left gastric artery for the initial benign disease. As a result, four (12%) patients exhibited splenic hilar lymph node metastasis out of the 33 RGC patients: Two patients had initial gastric cancer, and the remaining two patients had initial benign disease. Thus, the ligation of the left gastric artery was not the main reason for the metastases to the splenic hilar lymph node. Indeed, other interruption of lymphatic flows might also influence lymphatic flow from a tumor. In our hospital, the incidence of splenic hilar lymph node metastasis from RGC was higher than that from PGC [RGC vs. PCG, 12% (4/33) vs. 7% (14/207)]. Therefore, the interruption of lymphatic flow at the initial surgery might alter lymphatic flow from a tumor. However, the detailed mechanisms of lymphatic flow remain unclear.
In early RGC, no lymph node metastasis was detected although a low incidence of peri-gastric lymph node metastasis was noted in PGC. Namely, differences of the metastatic region and extent of lymph node involvement between RGC and PGC were small. On the other hand, in advanced RGC, the incidences of splenic hilar lymph node metastasis (RGC vs. PGC, 17% vs. 10%) and jejunal mesentery lymph node metastasis (RGC vs. PGC, 35% vs. 0%) were higher because RGC has a different pattern of lymphatic flow after initial distal gastrectomy. Concerning the initial surgery, the splenic hilar lymph node metastases occurred following every type of reconstruction. In contrast, the jejunal mesentery lymph node metastases occurred only following Billroth II reconstruction (Billroth I vs. Billroth II, 0% vs. 67%). This incidence of metastasis was higher than the previously reported incidences of 9–26%.23–25 Therefore, the splenic hilar lymph node dissection is essential for curative gastrectomy in all RCG patients. Additionally, the jejunal mesentery lymph node dissection should be performed in patients with RGC following Billroth II reconstruction.
Owing to recent advances in diagnostic and treatment techniques, RGC is not always advanced at diagnosis and, if it is, extensive surgery for RGC does not necessarily lead to poor prognosis in comparison to that for primary PGC. Patients with RGC have a different pattern of lymph node metastasis compared with that in PGC. Therefore, in decision making regarding the area of lymphadenectomy, the jejunal mesentery and splenic hilar lymph nodes should be specifically targeted for en bloc resection during complete gastrectomy in RGC.
References
Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin 2005; 55: 74–108.
Gotoda T. Endoscopic resection of early gastric cancer. Gastric Cancer. 2007; 10:1–11.
Degiuli M, Sasako M, Ponti A; Italian Gastric Cancer Study Group. Morbidity and mortality in the Italian Gastric Cancer Study Group randomized clinical trial of D1 versus D2 resection for gastric cancer. Br J Surg. 2010; 97:643–649.
Katai H, Sasako M, Fukuda H, Nakamura K, Hiki N, Saka M, et al. JCOG Gastric Cancer Surgical Study Group. Safety and feasibility of laparoscopy-assisted distal gastrectomy with suprapancreatic nodal dissection for clinical stage I gastric cancer: a multicenter phase II trial (JCOG 0703). Gastric Cancer. 2010; 13:238–244.
Sasako M, Maruyama K, Kinoshita T, Okabayashi K. Surgical treatment of carcinoma of the gastric stump. Br J Surg. 1991; 78:822–824.
Newman E, Brennan MF, Hochwald SN, Harrison LE, Jr. Karpeh MS. Gastric remnant carcinoma: just another proximal gastric cancer or a unique entity? Am J Surg 1997; 173:292–297.
Japanese Gastric Cancer Association. Japanese classification of gastric carcinoma, Fourteenth Edition. (in Japanese). Tokyo: Kanehara, 2010.
Sobin L, Gospodarowicz M, Wittekind C, editors. International union against cancer. TNM classification of malignant tumours, Seventh Edition. New York: Wiley-Blackwell; 2010.
Ohashi M, Katai H, Fukagawa T, et al. Cancer of the gastric stump following distal gastrectomy for cancer. Br J Surg 2007; 94:92–95.
Kaneko K, Kondo H, Saito D, Shirao K, Yamaguchi H, Yokota T, et al. Early gastric stump cancer following distal gastrectomy. Gut 1998; 43:342–344.
Kaminishi M, Shimizu N, Shiomoyama S, Yamaguchi H, Ogawa T, Sakai S, et al. Etiology of gastric remnant cancer with special reference to the effects of denervation of the gastric mucosa. Cancer 1995; 75:1490–1496.
Tersmette AC, Offerhaus GJ, Tersmette KW, Giardiello FM, Moore GW, Tytgat GN, et al. Meta-analysis of the risk of gastric stump cancer: detection of high risk patient subsets for stomach cancer after remote partial gastrectomy for benign conditions. Cancer Res 1990; 50:6486–6489.
Ahn HS, Kim JW, Yoo MW, Park do J, Lee HJ, Lee KU, et al. Clinicopathological features and surgical outcomes of patients with remnant gastric cancer after a distal gastrectomy. Ann Surg Oncol 2008; 15:1632–1639.
Tanigawa N, Nomura E, Lee SW, Kaminishi M, Sugiyama M, Aikou T, et al. Society for the Study of Postoperative Morbidity after Gastrectomy. Current state of gastric stump carcinoma in Japan: based on the results of a nationwide survey. World J Surg 2010; 34:1540–1547.
Kodera Y, Yamamura Y, Torii A, Uesaka K, Hirai T, Yasui K, et al. Gastric stump carcinoma after partial gastrectomy for benign gastric lesion: what is feasible as standard surgical treatment? J Surg Oncol 1996; 63:119–124.
Maruyama K, Kaminishi M, Hayashi K, Isobe Y, Honda I, Katai H, et al. Gastric cancer treated in 1991 in Japan: data analysis of nationwide registry. Gastric Cancer 2006; 9:51–66
Kitano S, Shiraishi N, Uyama I, Sugihara K, Tanigawa N. Japanese Laparoscopic Surgery Study Group. A multicenter study on oncologic outcome of laparoscopic gastrectomy for early cancer in Japan. Ann Surg 2007; 245:68–72.
Nakayoshi T, Tajiri H, Matsuda K, Kaise M, Ikegami M, Sasaki H. Magnifying endoscopy combined with narrow band imaging system for early gastric cancer: correlation of vascular pattern with histopathology (including video). Endoscopy 2004; 36:1080–1084.
Takenaka R, Kawahara Y, Okada H, Tsuzuki T, Yagi S, Kato J, et al. Endoscopic submucosal dissection for cancers of the remnant stomach after distal gastrectomy. Gastrointest Endosc 2008; 67:359–363.
Hirasaki S, Kanzaki H, Matsubara M, Fujita K, Matsumura S, Suzuki S. Treatment of gastric remnant cancer post distal gastrectomy by endoscopic submucosal dissection using an insulation-tipped diathermic knife. World J Gastroenterol 2008; 14:2550–2555.
An JY, Choi MG, Noh JH, Sohn TS, Kim S. The outcome of patients with remnant primary gastric cancer compared with those having upper one-third gastric cancer. Am J Surg 2007; 194:143–147.
Schaefer N, Sinning C, Standop J, Overhaus M, Hirner A, Wolff M. Treatment and prognosis of gastric stump carcinoma in comparison with primary proximal gastric cancer. Am J Surg 2007; 194:63–67
Han SL, Hua YW, Wang CH, Ji SQ, Zhuang J. Metastatic pattern of lymph node and surgery for gastric stump cancer. J Surg Oncol 2003; 82:241–246.
Kunisaki C, Shimada H, Nomura M, Hosaka N, Akiyama H, Ookubo K, et al. Lymph node dissection in surgical treatment for remnant stomach cancer. Hepatogastroenterology 2002; 49:580–584.
Yonemura Y, Ninomiya I, Tsugawa K, Masumoto H, Takamura H, Fushida S, et al. Lymph node metastases from carcinoma of the gastric stump. Hepatogastroenterology 1994; 41:248–252.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Komatsu, S., Ichikawa, D., Okamoto, K. et al. Differences of the Lymphatic Distribution and Surgical Outcomes Between Remnant Gastric Cancers and Primary Proximal Gastric Cancers. J Gastrointest Surg 16, 503–508 (2012). https://doi.org/10.1007/s11605-011-1804-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-011-1804-3