Abstract
Background
Symptomatic walled-off pancreatic necrosis (WOPN) treated with dual modality endoscopic and percutaneous drainage (DMD) has been shown to decrease length of hospitalization (LOH) and use of radiological resources in comparison to standard percutaneous drainage (SPD).
Aim
The aim of this study is to demonstrate that as the cohort of DMD and SPD patients expand, the original conclusions are durable.
Methods
The database of patients receiving treatment for WOPN between January 2006 and April 2011 was analyzed retrospectively.
Patients
One hundred two patients with symptomatic WOPN who had no previous drainage procedures were evaluated: 49 with DMD and 46 with SPD; 7 were excluded due to a salvage procedure.
Results
Patient characteristics including age, sex, etiology of pancreatitis, and severity of disease based on computed tomographic severity index were indistinguishable between the two cohorts. The DMD cohort had shorter LOH, time until removal of percutaneous drains, fewer CT scans, drain studies, and endoscopic retrograde cholangiopancreatography (ERCPs; p < 0.05 for all). There were 12 identifiable complications during DMD, which were successfully treated without the need for surgery. The 30-day mortality in DMD was 4% (one multi-system organ failure and one out of the hospital with congestive heart failure). Three patients receiving SPD had surgery, and three (7%) died in the hospital.
Conclusion
DMD for symptomatic WOPN reduces LOH, radiological procedures, and number of ERCPs compared to SPD.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
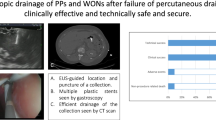
Walled-off pancreatic necrosis (WOPN) emerges during the second to third week after the onset of severe acute pancreatitis (SAP) and frequently is associated with a disrupted main pancreatic duct. WOPN appears as a mixed-density solid and liquid peripancreatic inflammatory mass surrounded by fibrous tissue on contrast-enhanced computed tomograms (Fig. 1).1–3 If asymptomatic, therapy is conservative, avoiding invasive treatment while allowing the pancreatic bed to clear and the patient to regain his previous clinical status. However, if WOPN obstructs or fistulizes to adjacent structures, erodes into blood vessels, becomes infected, or increases in size, then more aggressive treatment is advocated.4–7 Surgical debridement and drainage has been the standard treatment modality for WOPN until the recently published, randomized, controlled trial from the Dutch Pancreatitis Study Group. The Dutch Study Group demonstrated that a “step-up” approach in patients (initial management using minimally invasive techniques) had improved outcomes over patients treated with initial surgical debridement. Patients in the “step-up” cohort had decreased morbidity, fewer subsequent surgeries, lower rates of diabetes, and fewer incisional hernias.8 Less aggressive alternatives to open necrosectomies have developed over the past two decades and now include minimally invasive retroperitoneal drainage,9–13 percutaneous drainage,14–18 and transgastric endoscopic debridement of necromas.19–22 Each of the techniques can result in pancreatic cutaneous fistulae (PCF)21 and require frequent radiographic studies to monitor resolution of WOPN. The radiological studies expose patients to substantial quantities of ionizing radiation.23 Patients who are treated for WOPN often undergo lengthy hospitalizations, multiple endoscopic procedures, numerous consultations, and incur substantial medical costs.24,25
The authors of the current study noted that patients who spontaneously developed a fistula from WOPN to the duodenum during the course of percutaneous drainage of WOPN, the treatment modality historically used at Virginia Mason, had more rapid improvement in their clinical course. We formulated a technique that combined percutaneous drainage with endoscopic transenteric fistula formation at the onset of care that we named dual modality drainage (DMD).Footnote 1 The initial study of 15 patients demonstrated that no patient developed a PCF.26 A subsequent study comparing the first 23 patients to a cohort treated with standard percutaneous drainage (SPD) supported the initial conclusions as well as noted statistically significant reductions in length of hospitalizations and radiologic procedures.27 The current study addresses the durability of the earlier findings now that the cohort of DMD patients has doubled.
Methods
All patients with symptomatic WOPN admitted to our institution between January 2006 and March 2011 who had no drainage procedure beforehand were retrospectively identified from a database. Symptomatic WOPN was defined as follows:
-
1.
Infection of the “necromas” unresponsive to parenteral antibiotics evidenced by persistent fever, leucocytosis, and/or sepsis syndrome;
-
2.
Gastric outlet obstruction impeding feeding or causing persistent nausea and vomiting;
-
3.
Biliary obstruction resulting from WOPN;
-
4.
Fistulous connection to the colon, bile duct, skin, or other structure that did not improve with conservative management; and
-
5.
Clinical deterioration in the face of maximal medical therapy short of drainage.
Patient data were entered into a database approved by the Virginia Mason Medical Center Institutional Review Board.
Drainage Techniques
SPD Cohort
Symptomatic SAP patients had percutaneous drainage catheters placed into areas of WOPN as described by Freeny et al.14 Aspirated fluid from WOPN was sent for amylase and culture. Catheters were placed trying to avoid pulmonary, hepatic, colonic, and vascular structures. After placement and aspiration of as much fluid as possible, 12F drains were left to gravity and irrigated with 10 to 20 mL of sterile saline three times daily. Percutaneous catheters were sequentially upsized to a maximum of 28F as patients demonstrated signs of tube occlusion or lack of drainage. Catheters were removed when WOPN resolved and daily fluid output was less than 20 mL.
DMD Cohort
CT-guided percutaneous drains were placed as in the SPD cohort, but only 10 mL of fluid was aspirated for culture and amylase (Video 1). The patient was then rapidly transferred to a fluoroscopically equipped endoscopy suite at which time the WOPN was accessed either transgastrically or transduodenally (Video 2). Endoscopic ultrasound was used if there was an inconclusive luminal bulge (Video 3).
Endoscopic retrograde cholangiopancreatography (ERCP) was then performed to determine whether a pancreatic duct leak existed, and if identified, an endoprosthesis was placed into the pancreatic duct. If a biliary obstruction was found, a bile duct stent was placed as well. A nasojejunal tube was placed at the same setting for patients needing enteral support.
Regardless of drainage technique, patients received culture-directed antibiotics, and all patients were managed by critical care specialists or hospitalists. Signs of tube dysfunction in either cohort were managed by obtaining a CT scan and tube evaluation and/or exchange. Undrained fluid collections that were not in communication to the percutaneously drained collections often had another drain placed into them, especially if there were signs of infection.
Patient Selection
Hospitalists selected SPD or DMD after consultation with gastroenterologists and interventional radiologists. Most patients during the years 2006 through 2008 had SPD since that was the accepted treatment modality for 20 years at our institution. DMD was introduced in November 2007. By 2009, DMD was selected more frequently by hospitalists who had noted good outcomes. Qualifying criteria for DMD were that the “necromas” had to be contiguous with either the stomach or duodenum (Fig. 1).
Patients in both groups were discharged from the hospital when they were clinically stable and could be followed as outpatients. Removal of percutaneous drains occurred when they no longer produced fluid, the CT scan revealed no WOPN (Fig. 2), and patients were asymptomatic. Endoscopic transenteric stents were removed if the pancreatic duct was intact; otherwise, the stents were left indefinitely.
Completion of dual modality drainage (DMD), 54 days after placement of stents in Fig. 1. A Axial view: transgastric stents in resolved WOPN (red arrows) and percutaneous drain (purple arrow). B Coronal view. Note transgastric stents (red arrow) and stomach (green arrow)
Statistical Analysis
Comparative testing between the SPD and DMD groups was done using the unpaired two-tailed Student’s t test (for continuous variables) or chi-squared test (for categorical variables). P values <0.05 were considered significant.
Results
Between January 2006 and March 2011, 102 patients were identified and treated for symptomatic WOPN of whom 85% were transferred from surrounding hospitals and 15% admitted through the emergency department or from outpatient clinics. All patients were managed by hospitalists, critical care specialists, and/or medical residents who consulted with gastroenterologists, interventional radiologists, and surgeons. Fifty patients had planned DMD, but one did not have an adherent, vascular free endoscopic window and therefore was managed solely by SPD. Fifty-two patients underwent SPD from onset of therapy, but seven underwent a salvage dual drainage procedure during the course of therapy and were excluded from analysis (Fig. 3).
Flow chart of new patients with walled-off pancreatic necrosis: intention-to-treat. WOPN walled off pancreatic necrosis, DMD dual modality-therapy, SPD standard percutaneous drainage, MOSF multiple organ system failure, CHF congestive heart failure, PCF pancreatic cutaneous fistula, Panc ca pancreatic cancer, F/U follow-up
Both SPD and DMD cohorts were predominately male, 62% versus 80%, of equivalent ages, 53.5 versus 55.9 years, and with choledocholithiasis as the most frequent etiology, 56% versus 49%, respectively. Other causes of SAP were similar with the exception of hypertriglyceridemia in six (13%) of SPD cohort and one (2%) in the DMD cohort. Greater than 50% of both groups had ICU stays, and more than 50% of both groups had disconnected main pancreatic ducts. All other patient characteristics were indistinguishable except for a statistically significant variance in mean interval from SAP onset to initial drainage of 34.4 days in SPD and 53.3 days in DMD (Table 1; p > 0.02)
Computed tomography severity index (CTSI) was used as the measure of disease severity.28,29 The maximum CTSI from the onset of SAP became the severity score for each patient. Mean CTSI were similar in SPD and DMD, 7.2 versus 7.9, respectively (p < 0.06). The size and complexity of the fluid collections were similar (Table 1). Given that the vast majority of patients were transferred to our institution at different times from the onset of SAP, clinical measures of severity such as Ranson, Glasgow, and Acute Physiology, Age and Chronic Evaluation II could not be consistently and accurately assessed. Surrogate measures of clinical severity including creatinine, albumin, C-reactive protein (CRP), and American Society of Anesthesiologists (ASA) grading score showed no variance between the cohorts (Table 1). Patients who had any form of drainage whether surgical, endoscopic, or percutaneous prior to arrival were excluded from analysis.
The time of initial placement of percutaneous drains, until removal, decreased by more than 50% in the DMD cohort compared to SPD, 79 versus 183 days (p < 0.001). Three SPD patients had persistent PCFs requiring surgery, whereas no DMD patient had surgery for PCF. One patient in the SPD cohort was lost to follow-up, and two currently have external drains awaiting resolution of WOPN and/or PCF. Seven patients who were excluded from the current analysis had salvage DMD due to persistent PCFs. No DMD patient has been lost to subsequent evaluation (Table 2).
Mean LOH from placement of drains to discharge was significantly shorter in DMD, 24 versus 54 days (p < 0.001). Hospital days during any readmissions while a percutaneous drain was in the patient were added to the total. Occluded percutaneous drains, presenting as pain, fever, and leucocytosis, accounted for the vast majority of readmissions in both cohorts. DMD patients required fewer drains than SPD patients, 1.3 versus 1.9 (p < 0.001). Five SPD patients developed splenic artery pseudoaneurysms that bled and were controlled angiographically. To date, no DMD patient has developed a pseudoaneurysm (p < 0.02).
The mean number of CT scans from placement of drains until removal was fewer in DMD than SPD, 7.83 compared to 14.0 (p < 0.001). Correspondingly, fewer drain studies were needed until completion of therapy in DMD than SPD, 6.24 versus 13.0 (p < 0.001). Fewer ERCPs were performed in DMD than SPD, 1.88 versus 2.67 (p < 0.02). During the initial ERCP, both cohorts received equivalent pancreatic endoprostheses, 33% in DMD and 47% (p < 0.2) in SPD (Table 3).
Complications
Twelve total complications not associated with death were encountered in the DMD cohort (Table 3). Five patients developed abscesses in fluid collections that were adjacent to the initially drained WOPN but not in full communication. All resolved with placement of a second drain into the untreated fluid collection. Two percutaneous drains dislodged in the outpatient setting, but could be replaced through the existing fistula without need for another drain site. One patient developed a gastric outlet obstruction from duodenal obstruction that resolved with parenteral fluid support and antibiotics, but no added drains. Two patients had transient occlusion of the endoscopic gastric fistula noted during a drain exchange and were treated by endoscopic balloon dilation of the fistula. One patient developed a colonic fistula during the course of therapy that responded to reducing the size of the percutaneous drain. One patient was on anticoagulants for a thrombus at the site of an intravenous catheter and later developed a spontaneous psoas hematoma that resolved without intervention. Complications in the SPD cohort were not easily determined as tube occlusions, abscess formations in non-communicating fluid collections, and obstructions of adjacent structures were considered part of the natural history of SPD and were treated with exchange or addition of percutaneous drains.
Two SPD patients died of respiratory failure and one of multi-organ system failure (MOSF; 7%), all with percutaneous drains in place. In the DMD cohort, the 30-day mortality (4%) included one patient with severe congestive heart failure who died under hospice care as an outpatient. He had a drain in place. The other DMD death occurred in the hospital from MOSF. At autopsy, he was found to have an occult pancreatic cancer (Table 4).
Discussion
There are greater than 200,000 admissions to US hospitals yearly for acute pancreatitis of whom 20% have SAP and of those, one quarter have necrotizing pancreatitis.2 Deaths occur in patients with necrotizing pancreatitis in the early stage due to MOSF caused by a severe systemic inflammatory response. Those patients surviving the early stages of pancreatic necrosis may develop WOPN that can become infected or result in complications of sterile necrosis that include obstruction, fistulization, and persistent deterioration in health status.1,2,4 Patients with WOPN remain in hospital for weeks to months. They require multiple radiological and endoscopic procedures, and some patients develop diabetes, pancreatic insufficiency, pancreatic cutaneous fistulae, and if they have undergone surgery, incisional hernias.8 They generate substantial costs during their extended hospitalizations.24,25
Treatment for symptomatic WOPN has changed from open surgical debridement30–33 to favor less invasive techniques such as minimally invasive retroperitoneal drainage, percutaneous drainage, endoscopic necrosectomy, and combined modalities. The transition to less invasive management techniques was hastened by Van Santvoort’s multicenter, randomized study showing benefit in a “step-up” approach, which reduced the need for subsequent surgeries and decreased major complications.8 The authors of the current study previously have demonstrated that combining percutaneous drainage with immediate transenteric stenting eliminated PCFs26 and decreased both LOH as well as use of radiological resources.27 The expansion of our initial cohorts reveals that our previous findings are durable and also result in fewer ERCPs.
At our institution, percutaneous drainage has been the standard of care for nearly two decades.14–16 In spite of familiarity with the technique and constantly improving post-procedure management, patients undergoing SPD had very long LOH, were exposed to large quantities of ionizing radiation, and developed PCFs.34 By creating a controlled fistula at the onset of percutaneous drainage, LOH, monitoring CTs, and drain tube exchanges have decreased by 50%. Although it is difficult to compare varying management techniques between institutions, the decrease in LOH for DMD was comparative to the Dutch study where LOH was decreased by 10 days in the minimally invasive cohort8 and a US multicenter direct endoscopic necrosectomy study showing that patients remained in the hospital for 12 days after starting therapy.19 Compared to SPD, patients in DMD had drains removed after 79 rather than 183 days. The reduction in LOH, procedures, and duration of outpatient management of drains suggests that there was a substantial reduction in incurred costs to the patient and insurers although not computed for this study.
Treatment of WOPN is not without complications. Five abscesses occurred during treatment of DMD, all in patients who had non-communicating fluid collections adjacent to the one that was drained. Two patients dislodged drains as outpatients. Two other patients occluded their endoscopically created fistula. One patient developed transient gastric outlet obstruction, another had a spontaneous psoas hematoma, and one patient developed a pancreatic–colonic fistula. None of the complications resulted in surgery. All were successfully treated with a new drain, replacement of a drain, endoscopic balloon dilation, or conservative management. The noted complications and course of DMD patients compared favorably to reports of nonsurgical treatments for WOPN at other institutions that have included air emboli, severe bleeding, and procedure-related deaths.17–20 The 30-day mortality for the DMD patients is encouraging: one death from MOSF and occult pancreatic carcinoma and one of underlying severe cardiac disease outside of the hospital. There were no procedure-related deaths. Considering these deaths, one might argue that two PCFs had not resolved during the course of DMD. Comparatively, nine SPD patients had PCFs: three deaths, three surgeries, one lost to follow-up, and two awaiting PCF closure at the time of this manuscript. Furthermore, seven patients in the SPD cohort had salvage dual drainage to avoid chronic PCFs. Assuming that the salvage SPD patients would not close the PCFs, then 35% of SPD patients had PCFs while only 4% had them in the DMD cohort.
Limitations of the Study
The current study is limited by its retrospective nature and its lack of randomization. Bias could have been introduced during the selection of patients and the analysis. Furthermore, there appears to be a significant variance between the cohorts in the interval from onset of SAP to initial drain placement. SPD patients had drains placed after a mean of 34 days in comparison to 53 days in the DMD cohort (p < 0.02). Statistical significance was reached approximately when both cohorts had more than 40 patients. Possible explanations include recognition by all the consultants that delaying definitive procedures until later in the course of WOPN improved outcomes. Other possible explanations include later transfers over the study period from outlying hospitals and formerly stable patient who were weeks to months distant from the inciting episode of SAP who then became symptomatic and were seen in outpatient clinics. Managing patients with WOPN over the past few years with DMD has resulted in bias by both hospitalists and radiologists toward it over SPD. The vast majority of SPD patients were included in the cohort between 2006 and 2008. Since 2009, there have been 26 additions to the DMD cohort and only 9 to the SPD of whom 7 were shifted to salvage therapy. With the change in management in favor of DMD, the authors suspect that later drainage appears to have been favored. Our experience is consistent with surgical practice and endoscopic necrosectomy that advocates later drainage when WOPN is more liquefied.33,35,36
Another limitation of our study is that approximately 45% of both cohorts had infected necrosis, a number lower than some surgical or endoscopic series.8,20 The current study used positive cultures to demonstrate infection. We did not include “surrogates” of infection such as “air in WOPN” or “no other source of sepsis.”
Symptomatic WOPN presenting with obstruction, fistulization, and clinical deterioration short of sepsis were considered legitimate rationales for treatment. These patients, referred to in other series as having sterile necrosis, can linger in the hospital unable to be nourished adequately, unable to embark on physical therapy, and occasionally are jaundiced. The practice pattern of the authors, resulting from many years of managing patients with WOPN, is to provide drainage. At our tertiary pancreatitis referral center, we have instituted early enteral nutrition, improved nursing protocols, intensified physical therapy, and convened a multidisciplinary pancreatitis workgroup. Yet, the modification that most definitively reduced LOH, radiological studies, and endoscopic procedures has been DMD, with results that are more durable as the cohort enlarges. The interventional radiologists who do the vast majority of the procedural care encourage our hospitalists to consider initial DMD in hopes of reducing the number of drain studies, CT scans, and second tube placements. Furthermore, the radiologists have been instrumental in developing new multidisciplinary approaches to drainage (salvage therapy) for patients who have previously had SPD and now are facing PCFs. DMD for WOPN has proven effective in reducing LOH, radiological studies, endoscopic procedures, and nearly eliminated PCFs while maintaining mortality of less than 10% and to date, the elimination of the need for surgical intervention.
Notes
Also referred to as combined modality therapy in previous publications by our group.
References
Banks PA, Freeman ML, Practice Parameters Committee of the American College of Gastroenterology. Practice Guidelines in Acute Pancreatitis. Am J Gastroenterol 2006; 101:2379–2400.
Whitcomb DC. Acute Pancreatitis. New Engl J Med 2006; 354;2142–2150.
Baron TH, Morgan De. Acute necrotizing pancreatitis. New Engl J Med 1999; 340:1412–1418.
AGA Institute. Technical review on acute pancreatitis. Gastroenterology 2007;132:2022–2044.
Ashley SW, Perez A, Pierce EA, Brooks DC, Moore FD, Jr., Whang EE, Banks PA, Zinner MJ. Necrotizing pancreatitis. Contemporary analysis of 99 consecutive cases. Ann Surg 2001;234:572–580.
Rau B, Bothe A, Beger HG. Surgical treatment of necrotizing pancreatitis by necrosectomy and closed lavage: changing patient characteristics and outcome in a 19-year, single-center series. Surgery 2005; 138;28–39.
Howard TJ, Patel JB, Zyromski N, Sandrasegaran K, Yu J, Nakeeb A, Pitt HA, Lillemoe DK. Declining morbidity and mortality rates in the surgical management of pancreatic necrosis. J Gastrointest Surg 2007; 11:43–49.
Van Santvoort HC, Besselink MG, Baker OJ, Hofker HS, Boermeester MA, Dejong CH, van Goor H, Schaapherder AF, van Eijck CH, Bollen TL, van Ramshorst B, Nieuwenhuijs VB, Timmer R, Lameris JS, Kruyt PM, Manusama ER, van der Harst E, van der Schelling GP, Karsten T, Hesselink EJ, van Laarhoven CJ, Rosman C, Bosscha K, de Wit RJ, Houdijk AP, van Leeuwen MS, Buskens E, Gooszen HG, Dutch Pancreatitis Study Group. A step-up approach or open necrosectomy for necrotizing pancreatitis. N Engl J Med 2010; 362:1491–502.
Parekh D. Laparoscopic-assisted pancreatic necrosectomy: a new surgical option for treatment of severe necrotizing pancreatitis. Arc Surg 2006; 141-895-903.
Carter CR, McKay CJ, Imrie CW. Percutaneous necrosectomy and sinus tract endoscopy in the management of infected pancreatic necrosis: an initial experience. Ann Surg 2000; 232:175–180.
Ammori BJ. Laparoscopic transgastric pancreatic necrosectomy for infected pancreatic necrosis. Surg Endosc 2002; 16:1362.
Connor S, Ghaneh P, Raraty M, Sutton R, Rosso E, Garvey CJ, Hughes ML, Evans JC, Rowlands P, Neoptolemos JP. Minimally invasive retroperitoneal pancreatic necrosectomy. Dig Surg 2003; 20:270–277.
Horvath K, Freeny P, Escallon J, Heagerty P, Comstock B, Glickerman DJ, Bulger E, Sinanan M, Langdale L, Kolokythas O, Andrews RT. Safety and efficacy of video-assisted retroperitoneal debridement for infected pancreatic collections. A multi-center, prospective, single-arm phase 2 study. Arch Surg 2010;145:817–825.
Freeny PC, Hauptmann E, Althaus SJ, Traverso LW, Sinanan M. Percutaneous CT-guided catheter drainage of infected acute necrotizing pancreatitis: techniques and results. AJR Am J Roentgenol 1998; 170;969–975.
Fotoohi M, Traverso LW. Management of severe pancreatic necrosis. Curr Treat Options Gastroenterol 2007; 10:341–346.
Fotoohi M, Traverso LW. Pancreatic necrosis: paradigm of a multidisciplinary team. Adv Surg 2006; 40:107–118.
Endlicher E, Volk M, Feuerbach S, Scholmerich J, Schuffler A, Messmann H. Long-term follow-up of patients with necrotizing pancreatitis treated by percutaneous necrosectomy. Hepato-Gastroenterol 2003; 50:2225–2228.
VanSonnenberg E, Wittich GR, Chon KS, D’Agostino HB, Casola G, Easter D, Morgan RG, Walser EM, Nealon WH, Goodacre B, Stabile BE. Percutaneous radiologic drainage of pancreatic abscesses. AJR 1997; 168:979–984.
Papachristou GI, Takahashi N, Chahal P, Sarr MG, Baron TD. Peroral endoscopic drainage/debridement of walled-off pancreatic necrosis. Ann Surg 2007; 245:843–951.
Gardner TB, Chahal P, Papachristou GI, Vege SS, Petersen BT, Gostout CJ, Topazian MD, Takahashi N, Sarr MG, Baron TH. A comparison of direct endoscopic necrosectomy with transmural endoscopic drainage for the treatment of walled-off pancreatic necrosis. Gastrointest Endosc 2009; 69:1085–1094.
Gardner TB, Coelho-Prabhu N, Gordon Sr, Gelrud A, Maple JT, Papachristou GI, Freeman ML, Topazian MD, Attam R, Mackenzie TA, Baron TH. Direct endoscopic necrosectomy for the treatment of walled-off pancreatic necrosis; results from a multicenter U.S. series. Gastrointest Endosc 2011; 73:718–726.
Seifert H, Biermer M, Schmitt W, Jurgensen C, Will U, Gerlach R, Kreitmair C, Meining A, Wehrmann T, Rosch T. Transluminal endoscopic necrosectomy after acute pancreatitis: a multicentre study with long-term follow-up (the GEPARD Study). Gut 2009; 58:1260–1266.
Morgan DE, Ragheb CM, Lockhart ME, Cary B, Fineberg NS, Berland LL. Acute pancreatitis: computed tomography utilization and radiation exposure are related to severity but not patient age. Clin Gastroenterol Hepatol 2010; 8:303–308.
Fagenholz PJ, Fernandez-del Castillo C, Harris NS, Pelletier AJ, Camargo CA, Jr. Direct medical costs of acute pancreatitis hospitalizations in the United States. Pancreas 2007; 35:302–307.
Shaheen NJ, Hansen RA, Morgan DR, Gangarosa LM, Ringel Y, Thiny MT, Russo MW, Sandler RS. The burden of gastrointestinal and liver diseases, 2006. Am J Gastroenterol 2006; 101:2128–2138.
Ross A, Gluck M, Irani S, Hauptmann E, Fotoohi M, Siegal J, Robinson D, Crane R, Kozarek R. Combined endoscopic and percutaneous drainage of organized pancreatic necrosis. Gastrointest Endosc 2009; 71:79–84.
Gluck M, Ross A, Irani S, Lin O, Hauptmann E, Siegal J, Fotoohi M, Crane R, Robinson D, Kozarek RA. Endoscopic and percutaneous drainage of symptomatic walled-off pancreatic necrosis reduces hospital stay and radiographic resources. Clin Gastroenterol Hepatol 2010; 8:1083–1088.
Balthazar EJ, Freeny PC, VanSonnenberg E. Imaging and intervention in acute pancreatitis. Radiology 1994; 193:297–306.
Balthazar EJ. Acute pancreatitis: assessment of severity with clinical and CT evaluation. Radiology 2002; 223:603–613.
Beger HG, Buchler M, Bittner R, Oettinger W, Block S, Nevalainen T. Necrosectomy and postoperative local lavage in patients with necrotizing pancreatitis: results of a prospective clinical trial. World J Surg 1988; 12:255–262.
Traverso L W, Kozarek RA. Pancreatic necrosectomy: definitions and technique. J Gastrointest Surg 2005; 9:435–439.
Fernandez-del Castillo C, Rattner DW, Makary MA, Mostafavi A, McGrath D, Warshaw AL. Debridement and closed packing for the treatment of necrotizing pancreatitis. Ann Surg 1998; 228:676–684.
Connor S, Raraty MGT, Neoptolemos JP, Layer P, Runzi M. Does infected pancreatic necrosis require immediate of emergency debridement? Pancreas 2006; 33:128–134.
Fotoohi M, D’Agostino HB, Wollman B, Chon K, Shahrokni S, VanSonnenberg E. Persistent pancreatocutaneous fistula after percutaneous drainage of pancreatic fluid collections: role of cause and severity of pancreatitis. Radiology 1999; 213:373–578.
Mier J, Luque-de Leon E, Castillo A, Robledo F, Blanco R. Early versus late necrosectomy in severe necrotizing pancreatitis. Am j Surg 1997; 173:71–75.
Besselink MGH, Verwer TJ, Schoenmaeckers EJP, Buskens E, Ridwan BU, Visser MR, Nieuwenhuijs VB, Gooszen HG. Timing of surgical intervention of necrotizing pancreatitis. Arch Surg 2007; 142:1194–1201.
Author information
Authors and Affiliations
Corresponding author
Additional information
Discussant
Dr. Todd Baron (Rochester, MN): Use of irrigation in the management of pancreatic necrosis was initially described when specific open necrosectomy approaches were used. Irrigation became vital in the percutaneous approach as established by the Virginia Mason group. The initial series of successful endoscopic drainage of pancreatic necrosis employed aggressive irrigation through a single transmural tract using a nasobiliary tube inserted alongside internally placed stents. Subsequently, some authors reported using irrigation provided through placement of a jejunal extension tube through a PEG tube that was then passed through the transmural site into the collection; this also avoids external fistula.
The present study by Dr. Gluck and colleagues uses less intensive endoscopic and percutaneous approaches than when either is used alone. I believe this dual modality drainage (DMD) therapy could potentially be reproducible and particularly useful in community centers where endoscopic techniques for transmural placement of stents as for pseudocyst drainage and percutaneous techniques as for abscess drainage are now commonly available.
Questions:
1.What was the volume and type of irrigant used in the DMD group?
2.Your patients had primarily central pancreatic necrosis. Can you comment on how many patients had large paracolic gutter extensions (unilateral and bilateral)? These are patients traditionally difficult to treat using endoscopic transmural techniques alone.
3.What was the outcome of those in disconnected ducts in whom transmural stents remained long term? How long did those stents remain in place?
4.Can you comment as to why you think the pseudoaneurysm rate was much lower in the DMD group?
Closing Discussant
Dr. Michael Gluck:
1. What was the volume and type of irrigant used in the DMD group?
We used 20 mL of sterile normal saline, infused three times daily through each percutaneous drain. This was the same quantity and type of fluid as used in the SPD group. As the cavities closed, we decreased the volume until the drains were removed.
2. Your patients had primarily central pancreatic necrosis. Can you comment on how many patients had large paracolic gutter extensions (unilateral and bilateral)? These are patients traditionally difficult to treat using endoscopic transmural techniques alone.
Radiographs of all patients who had SPD and DMD were reviewed to determine if paracolic gutter extension existed on the day of percutaneous drainage. Sixty percent of SPD and 39% of DMD had evidence of left-sided, right-sided, or bilateral extension (p < 0.03). In the SPD group, 16 (35%) had bilateral extension compared to 10 (21%) in the DMD. Only two patients in the SPD cohort had solely right-sided involvement without left-sided as compared to none in the DMD cohort. As Dr. Baron mentioned, those patients with paracolic extension appear to have longer hospitalizations and duration of percutaneous drain tubes in both SPD and DMD. Comparing those with bilateral extension in both cohorts still demonstrates reduced hospital stays and number of radiological studies needed until discharge in the DMD cohort.
3. What was the outcome of those in disconnected ducts in whom transmural stents remained long term? How long did those stents remain in place?
Of the original 15 patients treated with DMD whose therapy was completed almost 3 years ago, 8 had disconnected ducts. Stents spontaneously passed in two. One developed a fluid collection that resolved with placement of a transpapillary stent. After removal of the transpapillary stent, the fluid collection recurred, but the patient is asymptomatic and has not required any new procedures. The other patient has no new fluid collection and is asymptomatic. Two fluid collections recurred in patients who had side branch leaks after transgastric stents were removed due to main branch strictures. Both fluid collections resolved with transpapillary stents, but as in the patients with disconnected ducts, the fluid collections recurred after stent removal. Both patients with new fluid collections remain asymptomatic. In all patients with side branch leaks, transgastric stents were removed at completion of therapy. For those eight with disconnected ducts, six patients have the transgastric stents in position currently without symptoms. We are aware of 1 other patient with a disconnected main pancreatic duct, not in the original cohort of 15, who developed a new symptomatic fluid collection adjacent to the ligament of Treitz 6 months after removal of percutaneous drains. The new collection was drained using endoscopic ultrasound. His previously placed transgastric stents are in position but not contiguous with the new fluid collection.
4. Can you comment as to why you think the pseudoaneurysm rate was much lower in the DMD group?
Although speculative, we think that pseudoaneurysms develop due to direct contact of arteries to the erosive effects of pancreatic secretions, inflammatory debris, and the mechanical trauma of percutaneous tube exchanges. The DMD cohort had fewer drains, fewer drain exchanges, smaller maximal drain size (18 French versus 28 French), and significantly shorter time to resolution of WOPN, decreasing exposure of the retroperitoneal vasculature to pancreatic secretions. The secretions preferentially drained into the stomach or duodenum and away from the WOPN.
Dr. Baron suggested that DMD could conceivably be instituted at community hospitals given that skilled endoscopic ultrasonographers place transgastric stents into pseudocysts and interventional radiologists commonly drain abscesses. In theory and with respect to skill level, we agree with him; however, we have cared for two patients who had DMD undertaken at community hospitals who then developed abscesses requiring further intensive management at our tertiary pancreatic center. Management of WOPN requires a team approach where there are an adequate number of committed interventional radiologists who cover one another, gastroenterologists who manage pancreatic diseases regularly, and surgeons who can and will operate on the pancreas if necessary. Until those teams are assembled, we recommend that management of WOPN be initiated and completed in centers with a substantial volume of patients with severe acute pancreatitis, expertise in minimally invasive techniques to treat WOPN, and a coordinated multidisciplinary approach for those patients.
All authors were involved in the design and analysis of study results and assisted in writing this paper.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
(MPG 13745 kb)
(WMV 14442 kb)
(MPG 26848 kb)
Rights and permissions
About this article
Cite this article
Gluck, M., Ross, A., Irani, S. et al. Dual Modality Drainage for Symptomatic Walled-Off Pancreatic Necrosis Reduces Length of Hospitalization, Radiological Procedures, and Number of Endoscopies Compared to Standard Percutaneous Drainage. J Gastrointest Surg 16, 248–257 (2012). https://doi.org/10.1007/s11605-011-1759-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-011-1759-4