Abstract
Background
Delayed gastric emptying (DGE) continues to be a major cause of morbidity following pancreaticoduodenectomy (PD). A change in the method of reconstruction following PD was instituted in an attempt to reduce the incidence DGE.
Methods
Patients undergoing PD from January 2002 to December 2008 were reviewed and outcomes determined. Pylorus-preserving pancreaticoduodenectomy (PPPD) with a retrocolic duodenojejunal anastomosis (n = 79) or a classic PD with a retrocolic gastrojejunostomy (n = 36) was performed prior to January 2008. Thereafter, a classic PD with an antecolic gastrojejunal anastomosis and placement of a retrogastric vascular omental patch was undertaken (n = 36).
Results
A statistically significant decrease in DGE was noted in the antecolic group compared to the entire retrocolic group (14% vs 40%; p = 0.004) and compared to patients treated by classic PD with a retrocolic anastomosis alone (14% vs 39%; p = 0.016). On multivariate analysis, the only modifiable factor associated with reduced DGE was the antecolic technique with an omental patch, odds ratio (OR) 0.3 (confidence interval (CI) 0.1–0.8) p = 0.022. Male gender was associated with an increased risk of DGE with OR 2.3 (CI 1.1–4.8) p = 0.026.
Conclusion
A classic PD combined with an antecolic anastomosis and retrogastric vascular omental patch results in a significant reduction in DGE.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Despite substantial reductions in mortality associated with pancreaticoduodenectomy (PD), the morbidity associated with this procedure remains significant.1,2 In high-volume centers, the morbidity associated with PD continues to range from 30% to 60%, even with improvements in intensive care management and overall perioperative care.3–7 Delayed gastric emptying (DGE) and pancreatic fistula are the two most common complications associated with PD.
The reported incidence of DGE varies according to the definition used. It is only recently that a consensus definition for DGE has been suggested.8 As per the International Study Group of Pancreatic Surgery (ISGPS), DGE has been defined as an inability to return to standard diet by the end of the first post-operative week following pancreatic resection. DGE occurs in approximately 19% to 57% of patients undergoing pancreaticoduodenal resection, with various theories regarding its etiology.9–15
The cause of DGE following PD is probably multifactorial.9,13,16–19 Changes in neuro-hormonal pathways related to duodenal and jejunal resection and regional ileus due to subclinical sepsis are two of several theories concerning the pathogenesis of DGE.20 In all cases of DGE, gastric coordination eventually improves and symptoms resolve. Numerous attempts have been made to prevent DGE without convincing evidence of improved outcomes. In a review of all randomized trials, it was concluded that, due to a lack of homogeneity in the definition DGE and design of studies, definite opinions regarding DGE and variables that influence it could not be derived.21
Based on the various theories concerning DGE, a change of technique in gastric reconstruction following pancreaticoduodenal resection was undertaken to reduce the incidence of DGE. Patients that had reconstruction with the new technique were compared to the preceding cases and factors influencing DGE were determined. Recent consensus definitions were used to define DGE.
Patients and Methods
Patient Population
All patients undergoing PD on the liver, pancreas, and foregut unit at Penn State Milton S. Hershey Medical Center from January 2002 to December 2008 were included in this study. Patients were identified from a prospective operative registry. Patient review and assessment was performed with institutional review board (IRB) approval.
Preoperative Assessment
Demographic data and indications for surgery were recorded for all patients.
Operative Procedures
Operative intervention and complications were identified. The extent of resection and the type of reconstruction was recorded. All surgical resections were performed using standard techniques. Pancreatic reconstruction was performed by two-layer duct-to-mucosa anastomosis and the bile duct reconstruction by single-layer interrupted sutures. In all cases, the jejunum was brought up to these anastomoses in a retrocolic manner through a defect created in the colon mesentery. Prophylactic jejunostomy tubes were utilized only in severely malnourished patients, when extra nutritional requirements were anticipated.
Between January 2002 and January 2008, a pylorus-preserving pancreaticoduodenectomy (PPPD) was the procedure of choice. Classic PD was performed with gastric antral excision when there was tumor infiltration into the proximal duodenum or inflammatory changes in this region. Anastomoses to the stomach or duodenum were constructed in a retrocolic, two-layer, hand-sewn fashion. Drains were placed posterior to the biliary and pancreatic anastomoses. A nasogastric tube was positioned during the case.
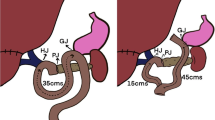
A change in technique was instituted after January 2008 due to concerns of consistently high DGE rates. The technique employed was based on theoretical concepts considered to reduce DGE and results of previously published clinical studies. In 36 consecutive cases, a classic PD was undertaken regardless of the pathology. While the pancreatic and bile duct anastomoses were constructed in a retrocolic fashion as before, the gastrojejunal anastomosis was now completed in an antecolic fashion by standard two-layer, hand-sewn techniques (Fig. 1). In all cases, a tongue of vascularized omentum was fashioned from the greater curve of the stomach to lie behind the gastrojejunal anastomosis to further separate the stomach from the underlying pancreaticojejunal anastomosis.
Post-operatively, all patients were managed in a surgical intensive care unit (SICU) setting for only the first 12 to 24 h, unless further monitoring was required. Nasogastric tubes were routinely removed day 1 post-operatively. A liquid diet was commenced day 2 post-operatively, with progression to soft diet as tolerated. The right and left drains were checked for amylase and bilirubin after day 4 and were removed sequentially over 2 days if there was no evidence of any pancreatic or biliary leakage. Patients were discharged home on day 6 or 7 unless there was an indication for more prolonged hospital stay. In all cases, erythromycin was given intravenously at 200 mg every 8 h until the time of discharge starting on day 2 post-operatively. A proton pump inhibitor was administered intravenously following surgery and converted to an oral dosage once a diet was tolerated. Pancreatic enzyme supplements were prescribed once a soft diet was commenced. Tight serum glucose control was maintained post-operatively by use of an insulin sliding scale.
Complications
Length of intensive care stay and hospital stay were recorded for all patients. Perioperative mortality was defined as death within 30 days of surgery. Complications were defined according to internationally accepted criteria.22 DGE was defined according to the ISGPS as the inability to return to a standard diet by day 7 post-operatively or reinsertion of a nasogastric tube prior to this period.8 Pancreatic fistula was also defined, according to ISGPS criteria, as any measurable amount of fluid after post-operative day 3 with an amylase level three or greater times the serum amylase.23 Patients in whom intra-abdominal collections required drainage in the perioperative period were considered to have high-impact pancreatic fistula, unless another explanation was clearly available.
All patients not tolerating a diet by day 7 post-operatively were defined as having DGE. Total parentral nutrition (TPN) was instituted in the majority of the cases and hospital discharge initiated in those patients that were otherwise well. The severity of DGE was not graded. Once TPN was instituted, there was generally no attempt at early reintroduction of a solid diet.
Statistical Analysis
Results were expressed as median (range) unless otherwise stated. Comparisons between categorical variables were determined by χ2 and Fisher’s exact test as appropriate. Non-categorical variables were assessed by the Mann–Whitney U test. To test the independence of risk factors for DGE, significant variables (p < 0.150) in univariate analysis were entered into a multivariable logistic regression model with likelihood ratio forward selection. A statistical software package (SPSS version 11.5, Chicago, IL, USA) was used for statistical analysis, with p < 0.05 considered as statistically significant.
Results
Patient Characteristics
There were 151 consecutive patients undergoing PD during the study period, with the last 36 performed by classic non-pylorus-preserving resection with an antecolic gastrojejunal anastomosis and retrogastric omental patch. The characteristics of the two groups of patients are shown in Table 1. There were significantly more American Society of Anesthesiologists (ASA) class IV patients in the antecolic group than in the retrocolic group (p < 0.001). There was also a trend toward a higher rate of pre-existing diabetes in the antecolic group compared to the retrocolic group (31% vs 17%; p = 0.066). The operative times in the antecolic group were significantly longer than in the retrocolic group (10 h vs 9 h; p < 0.001). All patients in the antecolic group had a classic PD, compared to 36 of 115 (31%) cases in the retrocolic group.
Complications
Complications
There was no operative mortality in this series. Complications are shown in Table 2. In the retrocolic group, pancreatic fistula occurred in 20 (17%) patients, consisting of 11 (55%) grade A, four (20%) grade B, and five (25%) grade C. In the antecolic group, pancreatic fistula occurred in eight (22%) patients, consisting of five (63%) in grade A and three (38%) in grade C classes. There was no difference in pancreatic fistula rate between the groups (p = 0.515). Wound infections were noted in 20 (12%) patients with no significant differences between the retrocolic and antecolic groups (10% vs 23%; p = 0.069).
The only statistically significant difference in complication was a decrease in DGE in the antecolic group (14% vs 40%; p = 0.004). Five patients in the antecolic group developed DGE. Two of these patients had manipulation or repair of large paraesophageal hernia during the PD. Another patient in the antecolic group had symptoms of a small bowel obstruction 1 week post-operatively requiring nasogastric tube reinsertion, which resolved after removal of her abdominal drain tube. She was classified as having DGE based on the strict definition set by the ISGPS.
Readmissions
There were 41 (27%) readmissions overall related to one or more complications in this series. The major reason for readmission was DGE (20 (13%)), followed by infective complications (19 (13%)). DGE was treated by intravenous rehydration and initiation of TPN in cases of readmission. Infective complications were mainly in the form of collections caused by pancreatic leaks, requiring drainage. In these cases, patients were generally admitted to hospital for 12 to 24 h of observation following percutaneous interventions. The overall readmission rates were similar in the antecolic and retrocolic groups (20% vs 30% p = 0.233). There was, however, a significant reduction in readmissions related to DGE in the antecolic group (0% vs 17%; p = 0.004). Readmissions due to infective complication were similar between the antecolic and retrocolic groups (14% vs 12% p = 0.787).
Classic Pancreaticoduodenectomy
Comparison of 36 patients undergoing classic PD in the retrocolic group to the 36 patients in the antecolic group is shown (Table 3). The patients in the antecolic group had higher ASA IV classification than the retrocolic group (p = 0.047). There was significantly reduced DGE in the antecolic group compared to the retrocolic classic PD group (14% vs 39%; p = 0.016). No other significant differences were noted. Comparison of all 72 patients treated by classic PD compared to PPPD only showed a trend towards reduced DGE (37% vs 63%; p = 0.067). When excluding the patients in the antecolic group, the rate of DGE between PPPD and classic PD with a retrocolic gastrojejunal anastomosis were similar (41% vs 39%; p = 0.870).
Factors Associated with Delayed Gastric Emptying
The overall effects of various factors on DGE based on univariate analysis is shown in Table 4. Classic PD with antecolic gastrojejunal anastomosis and retrogastric omental patch was the only modifiable factor associated with decreased DGE with an odds ratio (OR) of 0.2 (confidence interval (CI) 0.1–0.9) p = 0.004. There was a strong trend towards higher DGE in male patients, OR 2.0 (CI 1.0–4.0) p = 0.052.
Overall, the presence of complications was not associated with increased DGE. Specifically, pancreatic fistula was not associated with increased DGE. When analyzed as separate groups, in patients treated by classic PD or PPPD with a retrocolic anastomosis or those with a classic PD and antecolic gastrojejunal anastomosis and omental patch, there was still no statistically significant association between pancreatic fistula and DGE. Overall, a trend toward decreased DGE was noted in patients with pancreatic cancer pathology (p = 0.137), and in patients treated by classic PD, rather than PPPD (p = 0.067).
On multivariate analysis, two independent factors significantly influenced DGE. An antecolic anastomosis with a retrogastric omental patch significantly reduced DGE, OR 0.3 (CI 0.1–0.8) p = 0.022, whereas male gender was associated with increased DGE, OR 2.3 (CI 1.1–4.8) p = 0.026.
Discussion
Multiple theories regarding the etiology of DGE have been proposed. Disruption of hormone and neuronal homeostasis;20,21,24 diminished hormonal stimulation;17,19,25–29 gastroparesis due to intra-abdominal complications;9,17,30–34 post-operative pancreatitis;35 pyloric, antral, and duodenal ischemia;36,37 denervation of the stomach;17,38 post-operative pylorospasm;39 and torsion and angulation of reconstruction36,40 are all proposed theories concerning the pathogenesis of DGE.
The reported incidence of DGE is highly variable, and ranges from 0% to 57% in randomized controlled trials.21,41–43 This may reflect the variability in the definition of DGE. Some previous studies defined DGE as an inability to tolerate a diet by 10 days post-operatively.21,43 This definition is not applicable to contemporary series, in which median hospital stay following PD is generally between 7 and 10 days. The incidence of DGE in our series prior to the institution of a change in technique was 40% according to strict consensus statement definitions.
A change in technique of gastric reconstruction was instituted in an attempt to reduce DGE rates. The change undertaken reflected possible theoretic benefits of one or more techniques over another and findings of previously reported studies. Antral resection was performed based on a meta-analysis showing a trend towards reduced DGE with classic PD.44 It was also based on the theory that DGE relates to pylorospasm, duodenal ischemia, and alterations of neurohormonal factors that control antral and pyloric contraction.36,37,39 We acknowledge that there are some reports of long-term advantages of PDDD over standard PD.45 This is, however, controversial, with advocates of both procedures.46 Our primary goal was to significantly reduce DGE by a change in operative technique. Long-term gastro-intestinal function was not examined.
An antecolic gastrojejunal anastomosis was performed to maximally distance this anastomosis from the pancreas, minimize possible jejunal kinking or angulation, and allow greater mobility of the stomach and jejunum. We created a vascularized omental tongue as a patch to further separate the gastrojejunal anastomosis from the pancreaticojejunostomy and any associated pancreatic leaks. In addition, we avoided gastrostomy and feeding jejunostomy tubes to minimize other factors that may slow gastric emptying and intestinal motility. All patients in this series were given erythromycin based on theoretical benefits of improved gastric emptying and positive results of previous randomized controlled trials.19,42,43
A reduction in DGE from 40% to 14% was noted with institution of a change in technique, despite inclusion of sicker patients according to ASA classifications and a trend towards a higher number of diabetics in the antecolic group. We expect to be criticized for a high rate of DGE in the retrocolic group. This, however, reflects strict use of the ISGPS criteria to define DGE. A change in our technique virtually eliminated hospital readmissions due to DGE. The reduced DGE rate noted is unlikely to be related to changes in peri-operative care during the different time periods examined. We specifically confined our study to patients treated after 2002, during a period when all patients had similar peri-operative management. Increased referral of complex patients to our institution with significant co-morbidities may explain the differences in ASA classification and longer operating times seen in the latter antecolic group.
Warshaw was the first to define the concept of DGE and associated it with pylorus-preserving pancreaticoduodenal resection.47 Several studies have suggested decreased DGE or earlier return of gastric function after standard PD.41,48,49 One randomized trial of 33 patients had zero cases of DGE after standard PD resection compared to 43% after PPPD (p < 0.05).41 This study, however, was conducted over an 8-year period with small number of patients. The reverse was shown in a randomized trial of classic PD, including extended lymph node dissection compared to PPPD, with 16% DGE compared to 6% (p = 0.006).50 Most series indicate no difference in DGE between classic PD and PDDD.51 In our own series, there was no only a trend towards reduced DGE in the 72 patients treated by classic PD compared to the 79 patients undergoing PPPD. The trend was lost with exclusion of the antecolic classic PD patients.
The effect of an antecolic anastomoses in reducing DGE is supported by several publications.37,52–55 Theoretically, antecolic anastomosis avoids any mechanical problems by allowing increased mobility of the duodenojejunal or gastrojejunal anastomosis and avoiding torsion that may negatively affect gastric emptying.32,40,56 There are also arguments that decreased blood flow may occur due to venous congestion following retromesenteric passage of the afferent limb.57 In addition, such an anastomosis provides an anatomical barrier from the pancreas, minimizing possible negative effects of a pancreatic leak. In a recent trial of 40 patients undergoing PPPD randomized to either antecolic or retrocolic anastomosis, the rate of DGE in the antecolic group was 5% compared to 50% in the retrocolic group.53 Similar results were shown in a prospective study of 100 patients with retrocolic duodenojejunal anastomosis undergoing PPPD compared to 100 patients with an antecolic duodenojejunal anastomosis.52 The DGE rate was 5% in the antecolic group compared to 24% in the retrocolic cases. However, patients in the retrocolic group had greater operative blood loss and had a higher rate of medical complications than the antecolic group. In a recent study consisting of a small number of patients undergoing standard PD, an antecolic gastric anastomosis and undivided Roux-en-Y with a Braun enteroenterostomy resulted in less DGE that a standard reconstruction.58 It is possible that an antecolic method of reconstruction rather than creation of an enteroenterostomy was the cause of reduced DGE. In our study, the only modifiable factor resulting in reduced DGE on multivariate analysis was our change of technique, performing a classic PD with an antecolic anastomosis and retrogastric omental patch. Our patients appeared well-matched, with the only differences being higher ASA IV classification and longer operating times in the antecolic group. There was also a trend towards more patients with diabetes in the antecolic group. Intuitively, these differences would be considered to be more likely to increase DGE rates than to decrease them. We also noted that male gender was associated with higher risk of DGE. Although the pathophysiologic basis of this is undermined, this is in keeping with the findings of other studies.52,59
Post-operative complications were shown in several studies to be associated with DGE.9,32,33,59,60. In a study of 51 patients undergoing PPPD, DGE did not occur when there were no other complications, whereas 43% of patients with severe complications also had DGE.32 Pancreatic fistula is the most common complication associated with DGE based on several large series.61,62 Although not demonstrated in our study, it is possible that an antecolic anastomosis with the addition of a retroanastomotic omental patch reduces the effects of a clinical or subclinical pancreatic leak on gastric, intestinal, and anstomotic functioning. In our series, overall complications and pancreatic fistula rates were similar in the retrocolic and antecolic treatment groups and were not associated with increased DGE.
We can conclude from this study that a classic PD with an antecolic anastomosis and retrogastric omental patch results in significant reductions in DGE and related hospital readmissions. Further randomized studies are required to fully confirm these findings and to determine the role of antecolic anastomosis and vascularized omental patch in the setting of both classic PD and PPPD.
References
Trede M, Saeger HD, Schwall G, et al. Resection of pancreatic cancer—surgical achievements. Langenbecks Arch Surg. 1998;383:121–128.
Cameron JL, Riall TS, Coleman J, et al. One thousand consecutive pancreaticoduodenectomies. Ann Surg. 2006;244:10–15. doi:10.1097/01.sla.0000217673.04165.ea.
Pratt W, Joseph S, Callery MP, et al. POSSUM accurately predicts morbidity for pancreatic resection. Surgery 2008;143:8–19. doi:10.1016/j.surg.2007.07.035.
Lowy AM, Lee JE, Pisters PW, et al. Prospective, randomized trial of octreotide to prevent pancreatic fistula after pancreaticoduodenectomy for malignant disease. Ann Surg. 1997;226:632–641. doi:10.1097/00000658-199711000-00008.
DeOliveira ML, Winter JM, Schafer M, et al. Assessment of complications after pancreatic surgery: A novel grading system applied to 633 patients undergoing pancreaticoduodenectomy. Ann Surg. 2006;244:931–937. doi:10.1097/01.sla.0000246856.03918.9a discussion 937–939.
Grobmyer SR, Pieracci FM, Allen PJ, et al. Defining morbidity after pancreaticoduodenectomy: use of a prospective complication grading system. J Am Coll Surg. 2007;204:356–364. doi:10.1016/j.jamcollsurg.2006.11.017.
Balcom JHT, Rattner DW, Warshaw AL, et al. Ten-year experience with 733 pancreatic resections: changing indications, older patients, and decreasing length of hospitalization. Arch Surg. 2001;136:391–398. doi:10.1001/archsurg.136.4.391.
Wente MN, Bassi C, Dervenis C, et al. Delayed gastric emptying (DGE) after pancreatic surgery: a suggested definition by the International Study Group of Pancreatic Surgery (ISGPS). Surgery 2007;142:761–768. doi:10.1016/j.surg.2007.05.005.
Miedema BW, Sarr MG, van Heerden JA, et al. Complications following pancreaticoduodenectomy. Current management. Arch Surg. 1992;127:945–949. discussion 949–950.
Yeo CJ, Cameron JL, Sohn TA, et al. Six hundred fifty consecutive pancreaticoduodenectomies in the 1990s: pathology, complications, and outcomes. Ann Surg. 1997;226:248–257. doi:10.1097/00000658-199709000-00004. discussion 257–260.
Izbicki JR, Bloechle C, Broering DC, et al. Extended drainage versus resection in surgery for chronic pancreatitis: a prospective randomized trial comparing the longitudinal pancreaticojejunostomy combined with local pancreatic head excision with the pylorus-preserving pancreatoduodenectomy. Ann Surg. 1998;228:771–779. doi:10.1097/00000658-199812000-00008.
Yamaguchi K, Tanaka M, Chijiiwa K, et al. Early and late complications of pylorus-preserving pancreatoduodenectomy in Japan 1998. J Hepatobiliary Pancreat Surg. 1999;6:303–311. doi:10.1007/s005340050122.
Martignoni ME, Friess H, Sell F, et al. Enteral nutrition prolongs delayed gastric emptying in patients after Whipple resection. Am J Surg. 2000;180:18–23. doi:10.1016/S0002-9610(00)00418-9.
Wente MN, Shrikhande SV, Kleeff J, et al. Management of early hemorrhage from pancreatic anastomoses after pancreaticoduodenectomy. Dig Surg. 2006;23:203–208. doi:10.1159/000094750.
Richter A, Niedergethmann M, Sturm JW, et al. Long-term results of partial pancreaticoduodenectomy for ductal adenocarcinoma of the pancreatic head: 25-year experience. World J Surg. 2003;27:324–329. doi:10.1007/s00268-002-6659-z.
Hocking MP, Harrison WD, Sninsky CA. Gastric dysrhythmias following pylorus-preserving pancreaticoduodenectomy. Possible mechanism for early delayed gastric emptying. Dig Dis Sci. 1990;35:1226–1230. doi:10.1007/BF01536411.
Braasch JW, Deziel DJ, Rossi RL, et al. Pyloric and gastric preserving pancreatic resection. Experience with 87 patients. Ann Surg. 1986;204:411–418. doi:10.1097/00000658-198610000-00009.
Liberski SM, Koch KL, Atnip RG, et al. Ischemic gastroparesis: resolution after revascularization. Gastroenterology 1990;99:252–257.
Tanaka M, Sarr MG. Role of the duodenum in the control of canine gastrointestinal motility. Gastroenterology 1988;94:622–629.
Lytras D, Paraskevas KI, Avgerinos C, et al. Therapeutic strategies for the management of delayed gastric emptying after pancreatic resection. Langenbecks Arch Surg. 2007;392:1–12. doi:10.1007/s00423-006-0096-7.
Traverso LW, Hashimoto Y. Delayed gastric emptying: the state of the highest level of evidence. J Hepatobiliary Pancreat Surg. 2008;15:262–269. doi:10.1007/s00534-007-1304-8.
Pratt WB, Maithel SK, Vanounou T, et al. Clinical and economic validation of the International Study Group of Pancreatic Fistula (ISGPF) classification scheme. Ann Surg. 2007;245:443–451. doi:10.1097/01.sla.0000251708.70219.d2.
Bassi C, Falconi M, Molinari E, et al. Reconstruction by pancreaticojejunostomy versus pancreaticogastrostomy following pancreatectomy: results of a comparative study. Ann Surg. 2005;242:767–771. doi:10.1097/01.sla.0000189124.47589.6d discussion 771–763.
Meyer BM, Werth BA, Beglinger C, et al. Role of cholecystokinin in regulation of gastrointestinal motor functions. Lancet. 1989;2:12–15. doi:10.1016/S0140-6736(89)90255-9.
Fox JE, Daniel EE, Jury J, et al. The mechanism of motilin excitation of the canine small intestine. Life Sci. 1984;34:1001–1006. doi:10.1016/0024-3205(84)90305-9.
Naritomi G, Tanaka M, Matsunaga H, et al. Pancreatic head resection with and without preservation of the duodenum: different postoperative gastric motility. Surgery 1996;120:831–837. doi:10.1016/S0039-6060(96)80091-2.
Tait IS. Whipple's resection—proximal pancreaticoduodenectomy (PD). J R Coll Surg Edinb. 2002;47:528–540.
Tanaka M, Sarr MG. Total duodenectomy: effect on canine gastrointestinal motility. J Surg Res. 1987;42:483–493. doi:10.1016/0022-4804(87)90022-9.
Kim HC, Suzuki T, Kajiwara T, et al. Exocrine and endocrine stomach after gastrobulbar preserving pancreatoduodenectomy. Ann Surg. 1987;206:717–727. doi:10.1097/00000658-198712000-00006.
Haddad O, Porcu-Buisson G, Sakr R, et al. Diagnosis and management of adenocarcinoma of the ampulla of Vater during pregnancy. Eur J Obstet Gynecol Reprod Biol. 2005;119:246–249. doi:10.1016/j.ejogrb.2004.01.048.
Horstmann O, Becker H, Post S, et al. Is delayed gastric emptying following pancreaticoduodenectomy related to pylorus preservation? Langenbecks Arch Surg. 1999;384:354–359. doi:10.1007/s004230050213.
Horstmann O, Markus PM, Ghadimi MB, et al. Pylorus preservation has no impact on delayed gastric emptying after pancreatic head resection. Pancreas. 2004;28:69–74. doi:10.1097/00006676-200401000-00011.
Riediger H, Makowiec F, Schareck WD, et al. Delayed gastric emptying after pylorus—preserving pancreatoduodenectomy is strongly related to other postoperative complications. J Gastrointest Surg. 2003;7:758–765. doi:10.1016/S1091-255X(03)00109-4.
Hunt DR, McLean R. Pylorus-preserving pancreatectomy: functional results. Br J Surg. 1989;76:173–176. doi:10.1002/bjs.1800760223.
Lin PW, Lin YJ. Prospective randomized comparison between pylorus-preserving and standard pancreaticoduodenectomy. Br J Surg. 1999;86:603–607. doi:10.1046/j.1365-2168.1999.01074.x.
Itani KM, Coleman RE, Meyers WC, et al. Pylorus-preserving pancreatoduodenectomy. A clinical and physiologic appraisal. Ann Surg. 1986;204:655–664. doi:10.1097/00000658-198612000-00007.
Kurosaki I, Hatakeyama K. Preservation of the left gastric vein in delayed gastric emptying after pylorus-preserving pancreaticoduodenectomy. J Gastrointest Surg. 2005;9:846–852. doi:10.1016/j.gassur.2005.02.009.
Tanaka A, Ueno T, Oka M, et al. Effect of denervation of the pylorus and transection of the duodenum on acetaminophen absorption in rats; possible mechanism for early delayed gastric emptying after pylorus preserving pancreatoduodenectomy. Tohoku J Exp Med. 2000;192:239–247. doi:10.1620/tjem.192.239.
Kim DK, Hindenburg AA, Sharma SK, et al. Is pylorospasm a cause of delayed gastric emptying after pylorus-preserving pancreaticoduodenectomy? Ann Surg Oncol. 2005;12:222–227. doi:10.1245/ASO.2005.03.078.
Ueno T, Tanaka A, Hamanaka Y, et al. A proposal mechanism of early delayed gastric emptying after pylorus preserving pancreatoduodenectomy. Hepatogastroenterology 1995;42:269–274.
Lin PW, Shan YS, Lin YJ, et al. Pancreaticoduodenectomy for pancreatic head cancer: PPPD versus Whipple procedure. Hepatogastroenterology 2005;52:1601–1604.
Ohwada S, Satoh Y, Kawate S, et al. Low-dose erythromycin reduces delayed gastric emptying and improves gastric motility after Billroth I pylorus-preserving pancreaticoduodenectomy. Ann Surg. 2001;234:668–674. doi:10.1097/00000658-200111000-00013.
Yeo CJ, Barry MK, Sauter PK, et al. Erythromycin accelerates gastric emptying after pancreaticoduodenectomy. A prospective, randomized, placebo-controlled trial. Ann Surg. 1993;218:229–237. doi:10.1097/00000658-199309000-00002 discussion 237–228.
Diener MK, Knaebel HP, Heukaufer C, et al. A systematic review and meta-analysis of pylorus-preserving versus classical pancreaticoduodenectomy for surgical treatment of periampullary and pancreatic carcinoma. Ann Surg. 2007;245:187–200. doi:10.1097/01.sla.0000242711.74502.a9.
Pitt HA, Grace PA. Cancer of the pancreas. Pylorus-preserving resection of the pancreas. Baillieres Clin Gastroenterol. 1990;4:917–930. doi:10.1016/0950-3528(90)90026-D.
Han SS, Kim SW, Jang JY, et al. A comparison of the long-term functional outcomes of standard pancreatoduodenectomy and pylorus-preserving pancreatoduodenectomy. Hepatogastroenterology 2007;54:1831–1835.
Warshaw AL, Torchiana DL. Delayed gastric emptying after pylorus-preserving pancreaticoduodenectomy. Surg Gynecol Obstet. 1985;160:1–4.
van Berge Henegouwen MI, van Gulik TM, DeWit LT, et al. Delayed gastric emptying after standard pancreaticoduodenectomy versus pylorus-preserving pancreaticoduodenectomy: an analysis of 200 consecutive patients. J Am Coll Surg. 1997;185:373–379. doi:10.1016/S1072-7515(97)00078-1.
Patel AG, Toyama MT, Kusske AM, et al. Pylorus-preserving Whipple resection for pancreatic cancer. Is it any better? Arch Surg 1995;130:838–842. discussion 842–833.
Yeo CJ, Cameron JL, Lillemoe KD, et al. Pancreaticoduodenectomy with or without distal gastrectomy and extended retroperitoneal lymphadenectomy for periampullary adenocarcinoma, part 2: randomized controlled trial evaluating survival, morbidity, and mortality. Ann Surg. 2002;236:355–366. doi:10.1097/00000658-200209000-00012 discussion 366–358.
Paraskevas KI, Avgerinos C, Manes C, et al. Delayed gastric emptying is associated with pylorus-preserving but not classical Whipple pancreaticoduodenectomy: a review of the literature and critical reappraisal of the implicated pathomechanism. World J Gastroenterol. 2006;12:5951–5958.
Hartel M, Wente MN, Hinz U, et al. Effect of antecolic reconstruction on delayed gastric emptying after the pylorus-preserving Whipple procedure. Arch Surg. 2005;140:1094–1099. doi:10.1001/archsurg.140.11.1094.
Tani M, Terasawa H, Kawai M, et al. Improvement of delayed gastric emptying in pylorus-preserving pancreaticoduodenectomy: results of a prospective, randomized, controlled trial. Ann Surg. 2006;243:316–320. doi:10.1097/01.sla.0000201479.84934.ca.
Traverso LW, Kozarek RA. Pancreatoduodenectomy for chronic pancreatitis: anatomic selection criteria and subsequent long-term outcome analysis. Ann Surg. 1997;226:429–435. doi:10.1097/00000658-199710000-00004 discussion 435–428.
Sugiyama M, Abe N, Ueki H, et al. A new reconstruction method for preventing delayed gastric emptying after pylorus-preserving pancreatoduodenectomy. Am J Surg. 2004;187:743–746. doi:10.1016/j.amjsurg.2003.10.013.
Kingsnorth AN, Berg JD, Gray MR. A novel reconstructive technique for pylorus-preserving pancreaticoduodenectomy: avoidance of early postoperative gastric stasis. Ann R Coll Surg Engl. 1993;75:38–42.
Park YC, Kim SW, Jang JY, et al. Factors influencing delayed gastric emptying after pylorus-preserving pancreatoduodenectomy. J Am Coll Surg. 2003;196:859–865. doi:10.1016/S1072-7515(03)00127-3.
Wayne MG, Jorge IA, Cooperman AM. Alternative reconstruction after pancreaticoduodenectomy. World J Surg Oncol. 2008;6:9. doi:10.1186/1477-7819-6-9.
Fabre JM, Burgel JS, Navarro F, et al. Delayed gastric emptying after pancreaticoduodenectomy and pancreaticogastrostomy. Eur J Surg. 1999;165:560–565. doi:10.1080/110241599750006460.
Bar-Natan M, Larson GM, Stephens G, et al. Delayed gastric emptying after gastric surgery. Am J Surg. 1996;172:24–28. doi:10.1016/S0002-9610(96)00048-7.
Yeo CJ, Cameron JL, Sohn TA, et al. Pancreaticoduodenectomy with or without extended retroperitoneal lymphadenectomy for periampullary adenocarcinoma: comparison of morbidity and mortality and short-term outcome. Ann Surg. 1999;229:613–622. doi:10.1097/00000658-199905000-00003 discussion 622–614.
Howard JM. Pancreatojejunostomy: leakage is a preventable complication of the Whipple resection. J Am Coll Surg. 1997;184:454–457.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nikfarjam, M., Kimchi, E.T., Gusani, N.J. et al. A Reduction in Delayed Gastric Emptying by Classic Pancreaticoduodenectomy with an Antecolic Gastrojejunal Anastomosis and a Retrogastric Omental Patch. J Gastrointest Surg 13, 1674–1682 (2009). https://doi.org/10.1007/s11605-009-0944-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-009-0944-1