Abstract
Introduction
Swallowing sounds can be heard in the lower esophagus by xiphoid auscultation. We hypothesize that the xiphoid sound analysis could provide information concerning the integrity of the esophagogastric junction (EGJ), i.e., superposition of the lower esophageal sphincter (LES) and the diaphragm to assess clinical diagnosis of gastroesophageal reflux disease (GERD) and results of Nissen fundoplication (NF). The aim was to evaluate the changes in sound parameters using our acoustic technique after reorganization of the EGJ after NF.
Methods
For 21 patients with GERD and hiatus hernia, two microphones were placed below the cricoid and on the xiphoid cartilages. The frequency and duration of xiphoid sounds, esophageal transit time were calculated. We defined the xiphoid sound as composed of vibration groups separated by periods >100 ms. The number of vibration groups, number of vibrations per group, and interval between groups were also calculated.
Results
The xiphoid sound frequency was increased after NF, and the esophageal transit time and xiphoid sound duration were significantly decreased. A significant correlation was found between xiphoid sound duration and LES–diaphragm displacement. The number of vibration groups and interval between groups were reduced after NF.
Conclusion
The acoustic technique for swallowing revealed the effects of NF upon the dynamic profile of the EGJ. The organization of vibration groups at the EGJ suggested that the passage of the bolus was modified by hiatus hernia, i.e., dissociation between the LES and the diaphragm and regularized by NF. Concomitant acoustic and radiologic study should contribute to better understanding of sound related to EGJ structure and boli.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cervical auscultation has been applied to the study of pharyngeal swallowing.1–4 Hamlet et al.5 demonstrated that the sounds are generated by the flow across the cricopharyngeus, and Cichero et al.6 suggested an analogy between heart sounds and swallowing sounds because of the valve and pump system in the pharynx as in the heart.
With our digital acoustic recording technique for swallowing, we have performed combined cricoid (cervical) and xiphoid (thoracic) auscultation to study bolus displacement through the esophagus and the lower esophageal sphincter (LES) during swallowing.7 Using concurrent perfusion manometry and acoustic technique, we demonstrated that xiphoid sound occurs in the second half of the LES relaxation.7 Swallowing sounds recorded by noninvasive acoustic techniques are thus produced in the upper esophagus and also in the lower esophagus during the relaxation of these zones of constriction and during the passage of the bolus.
Incompetence of LES mechanisms leads to gastroesophageal reflux disease (GERD), i.e., when the LES has a low resting pressure8,9 or when transient lower esophageal sphincter relaxation is too frequent and too long.10,11 The presence of a hiatus hernia enhances reflux by inducing dissociation between intrinsic LES and extrinsic crural diaphragmatic high pressure zones and reducing esophagogastric junction pressure.12,13 Fundoplication is currently used mainly to treat GERD patients and is performed under laparoscopic conditions.14,15 The gastric wrap restores the high pressure zone of the distal esophagus and reduces the triggering of transient LES relaxation.16,17 Fundoplication achieves closure of the esophageal hiatus and reduction of the hiatus hernia to create a new anti-reflux barrier.
The xiphoid sounds are thus produced in the esophagogastric junction (EGJ) where the hiatus hernia induces dissociation between the LES and the crural diaphragm, both EGJ components. We therefore hypothesize that the xiphoid sound analysis could provide information concerning the dynamic function and integrity of the EGJ to assess clinical diagnosis of GERD and results of Nissen fundoplication (NF). The aim of this study was to describe and evaluate the changes in swallowing sound parameters at the EGJ after its surgical reorganization. Patients were examined using acoustic technique before and after NF.
Material and Methods
Patients
Twenty-one patients with GERD (11 men, ten women, mean age 43.2 ± 10.3 years) were enrolled. All patients were hospitalized in the Department of Visceral Surgery at the University Hospital of Tours. The study was approved by the Ethics Committee of the Hospital, and informed written consent was obtained in advance from each patient. None of the patients had a history of upper swallowing disorder. All patients had a hiatus hernia. The length of the intrathoracic displacement was 3.2 ± 0.7 cm, calculated during endoscopic examination before surgery. Esophageal manometry was performed with the slow pull-through technique to evaluate proximal (55.7 ± 16.9 mmHg) and distal (87.5 ± 35.5 mmHg) esophageal peristaltic pressure and length (2.9 ± 0.7 cm) and resting pressure (8.8 ± 4.2 mmHg) of LES before surgery. Gastroesophageal reflux was treated by 360° laparoscopic NF by the same surgeon (NH) to reproduce the surgical technique precisely. After closure of the crura and reduction of hiatus hernia, the wrap (approximately 3 cm in length) was realized. Patients were excluded if they had had esophageal or gastric surgery before NF and if the laparoscopic intervention was switched to a laparotomy approach during surgery.
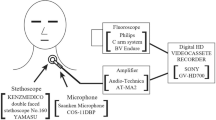
Acoustic Acquisition and Parameters
Acoustic data were obtained in an identical environment for each subject. Two omnidirectional microphones (Electret tie clip Sony® microphone, frequency range 50–18,000 Hz) were used. The cricoid microphone was placed in direct contact with the skin, on the anterolateral surface of the neck pressing just below the cricoid cartilage. The other microphone was inserted in a standard stethoscope and the flat diaphragm was placed on the xiphoid cartilage to obtain xiphoid sound. The microphones were kept in place by a fabric collar or belt. The microphones were connected to an amplifier linked to a computer audio acquisition card to obtain stereo signals under “.wave” form. The patients were instructed to fast for at least 6 h before the sound recordings. Each patient remained standing upright and was asked to perform a six-swallow sequence with 10 mL of 50% barium sulfate suspension (Micropaque® Laboratoire Guerlet, France) 1 day before and 2 days after surgery. The bolus volume was measured by syringe, placed in the mouth, and then swallowed. Each swallow was separated by 30 s.
All the recordings were analyzed using the Cool Edit Pro software program (Syntrillium Software Corporation, Phoenix, USA). Each recording was filtered with a band-pass filter (500–1,200 Hz) to extract xiphoid sounds. The stereo signal (cricoid and xiphoid sounds) obtained after filtering and zoom is illustrated in Fig. 1.
Stereo swallowing sounds recorded at the cricoid (above) and at the xiphoid (below) positions. Initial stereo swallowing sounds (upper). Stereo swallowing sounds after band-pass filtering, the xiphoid sound appears more clearly (middle). The zoom allows precise measurement of the xiphoid sound parameters (lower). The xiphoid sound (total duration, 2,060 s) was composed of three vibration groups. First group was composed of 13 sound vibrations (587 ms), second group was composed of five sound vibrations (122 ms), and third group was composed of five sound vibrations (229 ms). Intervals between bursts were 386 and 229 ms.
After all the acoustic signals had been heard, the frequencies of cricoid and xiphoid sounds were calculated before and after surgery in percent (%). For each sound recording, the esophageal transit time (the time between the start of the cricoid swallowing sound and the start of the xiphoid swallowing sound) and the duration of the xiphoid sound (the time elapsed between the start and the end of each sound) were measured in milliseconds (ms). We defined the xiphoid sound as composed of vibration groups separated by periods >100 ms. The zoom allows precise measurement of the sound components (Fig. 1). The number of vibration groups, number of vibrations per group, duration of each group (the time elapsed between the start and the end of each group), and intervals between groups (ms) were calculated for each xiphoid sound before and after surgery. The mean umbers and mean durations were calculated.
Statistic Analysis
Data were expressed as mean with their standard deviation (SD). A paired Student’s t test was used to compare the sound parameters before and after NF. Pearson correlation test was applied to establish correlations between the sound parameters and the EGJ displacement, or the LES pressure, or the distal esophageal pressure before NF. A p value <0.05 was considered significant.
Results
We obtained a total of 252 recordings. Two hundred twenty-six of them (90%) were good enough to permit analysis.
The cricoid sound was always heard before and after NF (100%).
Frequency of xiphoid sound was significantly enhanced after NF: 54% vs 84.8% (p < 0.05). For one man, no xiphoid sound was recorded before or after NF, and for one woman, no xiphoid sound was heard before NF. These two patients were excluded from the analysis because comparison between before and after NF was impossible (16 recordings).
Esophageal transit time was significantly higher before NF than after (6,865 ± 809 vs 5,194 ± 726 ms, p < 0.05). Individual values are shown before and after NF in Fig. 2. A significant positive correlation between esophageal transit time and EGJ displacement was present (r = 0.78, p < 0.001) before NF (Fig. 3). No significant correlation was found between esophageal transit time and LES pressure or distal pressure of esophagus.
Duration of xiphoid sound was significantly reduced after NF (1,671 ± 580 vs 758 ± 261 ms, p < 0.05). Individual values of xiphoid sound duration are shown before and after NF in Fig. 2. A significant positive correlation between xiphoid sound duration and EGJ displacement was present (r = 0.76, p < 0.001) before NF (Fig. 3). No significant correlation was found between this parameter and LES pressure or distal pressure of esophagus.
The xiphoid sound is composed of vibration groups separated by periods >100 ms (Fig. 1). Number of vibration groups and interval between groups were significantly reduced after NF (3.8 ± 1.7 vs 2.05 ± 0.8, p < 0.05 and 228 ± 98 vs 128 ± 51 ms, p < 0.05, respectively). In contrast, number of vibrations per group and duration of groups significantly increased after NF (3.7 ± 2.0 vs 6.3 ± 4.2, p < 0.05 and 283 ± 137 vs 360 ± 97 ms, p < 0.05). Significant positive correlations between number of groups or interval between groups and EGJ displacement were present (r = 0.68, p < 0.001 and r = 0.59, p < 0.05, respectively) before NF. Significant negative correlation between duration of groups and EGJ displacement was present (r = −0.59, p < 0.001) before NF. Typical example of xiphoid sounds before and after NF for the same patient is shown in Fig. 4.
Discussion
By using the digital acoustic recording technique for swallowing, we demonstrated that the frequency of xiphoid sound enhanced after NF. Fundoplication significantly decreased the durations of esophageal transit time and xiphoid sound. We also described the xiphoid sound, which is composed of vibration groups, and showed that the number of groups were significantly reduced after NF, separated by shorter intervals between groups. The number of vibrations per group and the durations of groups were significantly increased after NF.
In our study, the patients were placed in an upright position as in scintigraphy,18 impedancemetry,19 or high-resolution manometry.20
In the present study, the beginning of the cricoid sound was used to determine initiation of the swallowing reflex, as in the manometry technique where a displacement receptor was positioned on the neck near the cricoid cartilage.21,22 Cricoid sounds were always heard because patients with dysfunction of the upper esophagus were excluded from this protocol. The frequency of the xiphoid sound was low before NF. Bolus displacement was perhaps affected because the hiatus hernia created esophageal shortening,23 influenced the primary peristalsis by attenuation of shortening,24 and decreased the distal amplitude of the esophagus.22 These disorders of peristalsis might affect the speed of displacement of the bolus, which would then pass without making a sound. This is also the case when the LES pressure is low, the bolus met little resistance, and passed over the LES without sound. The frequency of xiphoid sound increased after NF, although without reaching normal values (i.e., 95%).7 It was demonstrated that NF reestablished the anti-reflux function by creating pressure on the distal esophagus by means of the gastric wrap, suppressing the hiatus hernia and reducing the diameter of the diaphragmatic hiatus.17,25,26 However, residual pressure was maintained with this restructuring during swallowing.27
We found that esophageal transit time was significantly longer before NF. With hiatus hernia, the LES and hiatus canal were dislocated and bolus transit was slowed down by the first passage across the LES and the second passage over the diaphragmatic hiatus.28,29 Fifteen patients had impaired peristalsis in the distal esophagus, which helped to decrease the rate of bolus displacement.30,31 Esophageal transit time decreased after NF because a single zone of high pressure was reconstructed at the EGJ. A significant positive correlation between esophageal transit time and EGJ displacement was found which would explain the effect of the hiatus hernia on the lengthening of bolus transit. Esophageal transit time was about 6–7 s after NF, the time required to pass through the entire esophagus, and was comparable to values reported by other authors32 (i.e., 7.2 s for water).
On the window of the Cool Edit Pro software, the xiphoid sound is simultaneously heard and visualized to facilitate the sound analysis. Computer programs able to process the xiphoid signal are being developed by our team to provide more detailed analysis of such signals.
Xiphoid sound duration and the organization of the vibration groups seem to be linked. We showed that the xiphoid sound was composed of vibration groups and that these groups were more numerous and more spaced out before NF. In consequence, xiphoid sound was longer. The sounds heard before NF seemed to reflect intermittent passage. These “hiccups” may be due to dysfunction in the distal esophagus in which turbulence occurs and to variations in speed of peristalsis. Three-dimensional imaging of the LES has shown that the LES can be defective over the entire length of the sphincter, or only partially in the intra-abdominal portion.9
With a hiatus hernia, a bolus successively transverses two zones of differing resistance, as well visualized by the pressure topography12 and pressure profile22 of the EGJ. The duration of the xiphoid sound was correlated with displacement of the EGJ; in other words, the greater the LES–diaphragmatic displacement, the longer the duration. This would appear to explain the difficulties in the passage of a bolus to the EGJ. Optimum elasticity of the EGJ is achieved by superposition of the smooth muscle of the LES and the striated muscle of the diaphragm, a reduced zone which is distended by passage of the bolus.33 This double musculature has an active part in the anti-reflux function.34
Xiphoid sound duration was shorter after NF (1,671 vs 758 ms) and there were fewer groups of vibrations. Kahrilas et al.25 and Scheffer et al.20 have shown that opening duration at the EGJ assessed with 10 ml barium suspension was unaffected by fundoplication, i.e., 13.4 vs 12.5 (supine) and 5.1 vs 5.0 s (upright), respectively. However, data on EGJ transit time before and after fundoplication are limited. A significant correlation between the increased EGJ transit time and the dysphagia score was reported.35,20 EGJ transit time was 2.8 s before NF and 5.8 s after NF with dysphagia.20 Our values were assessed before NF with hiatus hernia and after NF without dysphagia. Moreover, in all these techniques, the volume and consistency of the bolus were different. Our new acoustic technique using bolus of barium suspension and its results can be compared to other EGJ investigations to test differences, but more physiological boluses (water, semi-liquid, solid) might be used to standardize this noninvasive acoustic EGJ exploration. It can be hypothesized that the bolus traversed the EGJ under the effect of more regular pressure and that its passage was more rapid with a more compact bolus. Gastric wrap surrounds the esophagus over 3 cm and maintains high pressure throughout its length,36 but the distensibility of this new muscle arrangement decreases.37 Stretching of the body of the esophagus to make this wrap may contribute to the development of more effective peristalsis,38 but only in certain patients.39
Conclusion
The digital acoustic recording technique for swallowing is noninvasive and permits evaluation of the passage of a swallowed bolus through the esophagogastric junction. We demonstrated for the first time that the xiphoid sound analysis can reveal the dissociation between the LES and the crural diaphragm in patients with GERD and the xiphoid sound modifications in the same patients after NF. Since it provides additional information, particularly concerning the displacement of a bolus, displacement through the EGJ appears to be regularized by the fundoplication. Studies combining acoustic and radiological techniques are in progress to visualize the bolus and the sound at the same time and to provide greater understanding of the origins of the acoustic changes.
References
Zenner PM, Losinski DS, Mills RH. Using cervical auscultation in the clinical dysphagia examination in long-term care. Dysphagia 1995;10:27–31. doi:10.1007/BF00261276.
Boiron M, Rouleau P, Metman EH. Exploration of pharyngeal swallowing by audiosignal recording. Dysphagia 1997;12:86–92. doi:10.1007/PL00009524.
Perlman AL, Ettema SL, Barkmeier J. Respiratory and acoustic signal associated with bolus passage during swallowing. Dysphagia 2000;15:89–94.
Moriniere S, Boiron M, Beutter P. Sound components duration of healthy human pharyngo-esophageal swallowing. Dysphagia 2008;23:267–73. doi:10.1007/s00455-007-9134-z.
Hamlet JA, Nelson RJ, Patterson RL. Interpreting the sound of swallowing: fluid flow through the cricopharygeus. Ann Otol Rhinol Laryngol 1990;99:749–752.
Cichero JA, Murdoch BE. The physiologic cause of swallowing sounds: answers from heart sounds and vocal tract acoustics. Dysphagia 1998;13:39–52. doi:10.1007/PL00009548.
Boiron M, Rouleau P, Atipo B, Picon L, Metman EH. Esophageal swallowing phase assessed by audio recording. Relationship with manometry in gastroesophageal reflux disease patients. Dig Dis Sci 1999;44:529–535. doi:10.1023/A:1026649205827.
Zaninotto G, De Meester TR, Swizer W. The lower esophageal sphincter in health and disease. Am J Surg 1999;155:104–111. doi:10.1016/S0002-9610(88)80266-6.
Wetscher GJ, Hinder RA, Perdikis G, Wieschemeier T, Stalzer R. Three-dimensional imaging of the lower esophageal sphincter in healthy subjects and gastroesophageal reflux. Dig Dis Sci 1996;41:2377–2382. doi:10.1007/BF02100131.
Mittal RK, Holloway RH, Penagini R, Blackshaw LA, Dent J. Transient lower esophageal sphincter relaxation. Gastroenterology 1995;109:601–610. doi:10.1016/0016-5085(95)90351-8.
Holloway RH, Penagini R, Ireland AC. Criteria for objective definition of transient lower esophageal sphincter relaxation. Am J Physiol 1995;268:G128–G133.
Karhilas PJ, Lin S, Chen J, Manka M. The effect of hiatus hernia on gastro-oesophageal junction pressure. Gut 1999;44:476–482.
Mattioli S, D’Ovidio F, Pilotti V, Di Simone MP, Lugaresi ML, Bassi F, Brusori S. Hiatus hernia and intrathoracic migration of esophagogastric junction in gastroesophageal reflux disease. Dig Dis Sci 2003;48:1823–1831. doi:10.1023/A:1025471801571.
Spivak H, Lelcuk S, Hunter JG. Laparoscopic surgery of the gastroesophageal junction. World J Surg 1999;23:356–367. doi:10.1007/PL00012310.
Mattioli S, Lugaresi ML, Di Simone MP, D’Ovidio F, Pilotti V, Bassi F, Brusori S, Gavelli G. The surgical treatment of the intrathoracic migration of the gastro-oesophageal junction and of short oesophagus in gastro-oesophageal reflux disease. Eur J Cardiothorac Surg 2004;25:1079–1088. doi:10.1016/j.ejcts.2004.02.009.
Ireland AC, Holloway RH, Tooli J, Dent J. Mechanisms underlying the antireflux action of fundoplication. Gut 1993;34:303–308. doi:10.1136/gut.34.3.303.
Rydberg L, Ruth M, Lundell L. Mechanism of action of antireflux procedures. Br J Surg 1999;86:405–410. doi:10.1046/j.1365-2168.1999.01025.x.
Stier AW, Stein HJ, Siewert JR, Schwaiger M. Image processing in esophageal scintigraphy: topography of transit times. Dis Esophagus 2000;13:152–160. doi:10.1046/j.1442-2050.2000.00105.x.
Bredenoord AJ, Weusten BLA, Timmer R, Akkermans LMA, Smout AJP. Relationships between air swallowing, intragastric air, belching and gastro-oesophageal reflux. Neurogastroenterol Motil 2005;17:341–347. doi:10.1111/j.1365-2982.2004.00626.x.
Scheffer RCH, Samsom M, Haverkamp A, Oors GS, Hebbard GS, Gooszen HG. Impaired bolus transit across the esophagogastric junction in postfundoplication dysphagia. Am J Gastroenterol 2005;100:1684–1677. doi:10.1111/j.1572-0241.2005.42009.x.
Roling GT, Farrell RL, Castell DO. Cholinergic response of the lower esophageal sphincter. Am J Physiol 1972;222:967–972.
Cuomo R, Sarnelli G, Grasso R, Alfieri M, Bottiglieri ME, Paternuosto M, Budillon G. Manometric study of hiatal hernia and its correlation with esophageal peristalsis. Dig Dis Sci 1999;44:1747–1753. doi:10.1023/A:1018861715957.
Mittal RK, Balaban DH. The esophagogastric junction. N Engl J Med 1997;27:924–32. doi:10.1056/NEJM199703273361306.
Karhilas PJ, Scott WU, Lin S, Pouderoux P. Attenuation of esophageal shortening during peristalsis with hiatus hernia. Gastroenterology 1995;109:1818–1825. doi:10.1016/0016-5085(95)90748-3.
Kahrilas PJ, Lin S, Spiess AE, Brasseur JG, Joehl RJ, Manka M. Impact of fundoplication on bolus transit across esophagogastric junction. Am J Physiol 1998;275:G1386–G1393.
Chrysos E, Athanasakis E, Pechlivanides G, Tzortzinis A, Mantides A, Xynos E. The effect of total and anterior partial fundoplication on antireflux mechanisms of the gastroesophageal junction. Am J Surg 2004;188:39–44. doi:10.1016/j.amjsurg.2003.10.023.
Crookes PF, Ritter MP, Johnson WE, Bremner CG, Peters JH, DeMeester TR. Static and dynamic function of the lower esophageal sphincter before and after laparoscopic Nissen fundoplication. J Gastrointest Surg 1997;1:499–504. doi:10.1016/S1091-255X(97)80064-9.
Mittal RK, Lange RC, Mc Callum RW. Identification and mechanism of delayed esophageal acid clearance in subjects with hiatal hernia. Gastroenterology 1987;92:130–135.
Sloan S, Kahrilas PJ. Impaired of esophageal emptying with hiatal hernia. Gastroenterology 1991;100:596–605.
Karhilas PJ, Dodds WJ, Hogan WJ. Effect of peristaltic dysfunction on esophageal volume clearance. Gastroenterology 1988;94:73–80.
Cunningham ET, Horowitz M, Riddell PS, Maddern GJ, Myers JC, Holloway RH, Wishart JM, Jamieson GG. Relations among autonomic nerve dysfunction, oesophageal motility and gastric emptying in gastrooesophageal reflux disease. Gut 1991;32:1436–1440. doi:10.1136/gut.32.12.1436.
Wise JL, Murray JA, Conklin JL. Regional differences in oesophageal motor function. Neurogastroenterol Motil 2004;16:31–37. doi:10.1046/j.1365-2982.2003.00455.x.
Shaker R, Bardan E, Gu C, Massey BT, Sanders T, Kern MK, Hoffmann RG, Hogan WJ. Effect of lower esophageal sphincter tone and crural diaphragm contraction on distensibility of the gastroesophageal junction in humans. Am J Physiol 2004;287:G815–G821.
Mittal RK. Current concepts of the antireflux barrier. Gastroenterol Clin North Am 1990;19:501–516.
Tatum RP, Shi G, Manka MA, Brasseur JG, Joehl RJ, Kahrilas PJ. Bolus transit assessed by an esophageal stress test in postfundoplication dysphagia. J Surg Res 2000;91:56–60. doi:10.1006/jsre.2000.5907.
Karhilas PJ, Lin S, Manka M, Shi G, Joehl RJ. Esophagogastric junction pressure topography after fundoplication. Surgery 2000;127:200–208. doi:10.1067/msy.2000.102045.
Blom D, Bajaj S, Liu J, Hofmann C, Rittmann T, Derksen T, Shaker R. Laparoscopic Nissen fundoplication decreases gastroesophageal junction distensibility in patients with gastroesophageal reflux disease. J Gastrointest Surg 2005;9:1318–1325. doi:10.1016/j.gassur.2005.08.032.
Gill RC, Bowes KL, Murphy PD, Kingma YJ. Esophageal motor abnormalities in gastroesophageal reflux and the effects of fundoplication. Gastroenterology 1986;91:364–369.
Fibbe C, Layer P, Keller J, Strate U, Emmermann A, Zoring C. Esophageal motility in reflux disease before and after fundoplication: a prospective, randomized, clinical and manometric study. Gastroenterology 2001;121:5–14. doi:10.1053/gast.2001.25486.
Acknowledgments
This study was supported by the French Ministry of Education, Research and Technology with the ACI program “Telemedicine and Technologies for Health” and by the University François-Rabelais of Tours.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Boiron, M., Benchellal, Z. & Huten, N. Study of Swallowing Sound at the Esophagogastric Junction Before and After Fundoplication. J Gastrointest Surg 13, 1570–1576 (2009). https://doi.org/10.1007/s11605-009-0937-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-009-0937-0