Abstract
Introduction
Organ complications like biliary or duodenal stenosis as well as intractable pain are current indications for surgery in patients with chronic pancreatitis (CP). We present here our experience with pancreatic resection for CP and focus on the long-term outcome after surgery regarding pain, exocrine/endocrine pancreatic function, and the control of organ complications in 224 patients with a median postoperative follow-up period of 56 months.
Methods
During 11 years 272 pancreatic resections were performed in our institution for CP. Perioperative mortality was 1%. Follow-up data using at least standardized questionnaires were available in 224 patients. The types of resection in these 224 patients were Whipple (9%), pylorus-preserving pancreato-duodenectomy (PD) (PPPD; 40%), duodenum-preserving pancreatic head resection (DPPHR; 41%, 50 Frey, 42 Beger), distal (9%) and two central pancreatic resections. Eighty-six of the patients were part of a randomized study comparing PPPD and DPPHR. The perioperative and follow-up (f/up) data were prospectively documented. Exocrine insufficiency was regarded as the presence of steatorrhea and/or the need for oral enzyme supplementation. Multivariate analysis was performed using binary logistic regression.
Results
Perioperative surgical morbidity was 28% and did not differ between the types of resection. At last f/up 87% of the patients were pain-free (60%) or had pain less frequently than once per week (27%). Thirteen percent had frequent pain, at least once per week (no difference between the operative procedures). A concomitant exocrine insufficiency and former postoperative surgical complications were the strongest independent risk factors for pain and frequent pain at follow-up. At the last f/up 65% had exocrine insufficiency, half of them developed it during the postoperative course. The presence of regional or generalized portal hypertension, a low preoperative body mass index, and a longer preoperative duration of CP were independent risk factors for exocrine insufficiency. Thirty-seven percent of the patients without preoperative diabetes developed de novo diabetes during f/up (no risk factor identified). Both, exocrine and endocrine insufficiencies were independent of the type of surgery. Median weight gain was 2 kg and higher in patients with preoperative malnutrition and in patients without abdominal pain. After PPPD, 8% of the patients had peptic jejunal ulcers, whereas 4% presented with biliary complications after DPPHR. Late mortality was analyzed in 233 patients. Survival rates after pancreatic resection for CP were 86% after 5 years and 65% after 10 years.
Conclusions
Pancreatic resection leads to adequate pain control in the majority of patients with CP. Long-term outcome does not depend on the type of surgical procedure but is in part influenced by severe preoperative CP and by postoperative surgical complications (regarding pain). A few patients develop procedure-related late complications. Late mortality is high, probably because of the high comorbidity (alcohol, smoking) in many of these patients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The knowledge about the pathophysiology and origin of inflammation and pain in chronic pancreatitis (CP) has increased during the past decades. Progress in interventional procedures, such as endoscopic retrograde cholangiopancreatography, and improved cross-sectional imaging (computerized tomography [CT] and magnetic resonance imaging, [MRI]) has helped to delineate the inflammatory processes better. The origin of pain was initially explained by pancreatic duct obstruction. Decompression was thought to control pain effectively. In a large analysis of more than 1,000 patients endoscopic drainage was successful in up to two-thirds of the patients.1 A short preinterventional duration of CP was associated with a larger success rate of endoscopic drainage.2 Nevertheless, a relevant number of interventionally treated patients had to undergo surgery because of failed symptom relief. Moreover, up to 50% of patients with chronic pancreatitis require surgical therapy during the long-term course of their disease.3
The hypothesis of pain origin by mechanisms other than increased intraductal pressure (e.g., fibrosis, alteration of pancreatic nerves, inflammatory enlargement) was strengthened during the last decade.4 Detection and description of an inflammatory mass predominantly in the pancreatic head was a further step in the understanding of CP.5,6 In an important follow-up analysis of patients with “failure of symptomatic relief after pancreatojejunal decompression”, the cause for recurrent pancreatitis was localized in the pancreatic head, which was consecutively declared as a pacemaker of the disease.6 With the publication of further disappointing results after surgical drainage, resectional procedures gained more importance in the treatment of CP, especially in patients with inflammatory enlarged areas of the pancreas. Duodenum preserving pancreatic head resections (DPPHR) as described by Beger7 and Frey8 and the pylorus preserving pancreatoduodenectomy (PPPD) as re-introduced by Traverso and Longmire9 are current resectional procedures in chronic pancreatitis predominantly of the pancreatic head. Removal of the inflammatory pancreatic head mass resulted in substantial pain relief and control of other organ complications in up to 90% of patients.10–14
Pancreatic resection is now a procedure with acceptable morbidity and low mortality. In many centers, a perioperative mortality rate of clearly less than 5% has been reported. As a result of these advances in the perioperative course after pancreatic surgery, the debate on the indications and results of resectional surgery for CP now focuses on the long-term outcome (quality of life, pain control, endocrine and exocrine function, control of organ complications).
The aim of our study was to evaluate the long-term course after resection for CP in more than 200 patients with a median follow-up of almost 5 years. Risk-factor analyses were performed to search for potential parameters influencing the long-term outcome.
Patients and Methods
Patients and Indications for Surgery
From July 1994 to December 2005, 272 patients underwent pancreatic resection for CP. Postoperative histological examination confirmed CP in all cases. Of these 272 patients, prospective postoperative follow-up data using standardized questionnaires could be gained in 224 (82%). Three patients (1%) died of postoperative complications. Of the remaining 45 patients, nine (3%) died without postoperative follow-up, eight (3%) were not contacted because of a postoperative observation period of less than 6 months, and 28 (10%) were lost to follow-up.
The 224 patients (80% male) included in this study had a median age of 44 years (range 27–79 years) at the time of surgery. Median preoperative duration of CP was 36 (1–444) months. Further preoperative characteristics and (co-) indications for surgery are listed in Table 1.
The leading indications for surgery in the 224 patients were pain (chronic or recurrent; n = 147, 66%), jaundice (n = 36; 16%), duodenal obstruction (n = 12; 5%), or one of various others (n = 29; 13%). It is of note, however, that many patients had more than one indication for pancreatic resection. Two hundred eight (93%) patients had pain (chronic or during recurrent episodes of pancreatitis) as indication or coindication for surgery (Table 1).
During the evaluation of the preoperative status the intake of pain medication was documented (as yes or no). In contrast to the postoperative follow-up data, we have no further details on preoperative pain medication like frequency of intake or type of analgesic taken. Preoperatively, 87 patients (39%) of the entire study group (and 42% of the 208 patients with pain and/or recurrent attacks of pancreatitis) had documented pain medication.
Preoperative Assessment
All patients had at least one cross-sectional imaging modality before surgery (CT in 94%, MRI in 34%). During the last years of the study period MRI included MRCP and MR-angiography in the majority of patients. Until 2001, most patients preoperatively underwent conventional angiography because of the high percentage of vascular involvement in our patients. Since 2001, with better vessel imaging by multislice CT and/or MR-angiography conventional angiography is restricted to selected patients only (79% had angiography). One hundred fifty-seven patients (70%) had an ERCP preoperatively, and 61 (27%) had a preoperative biliary drainage. Of our patients only a few preoperatively underwent endoscopic stenting of the pancreatic duct. Endoscopic ultrasound was performed in 55% of our study group.
Anatomical Description of Chronic Pancreatitis
In the entire patient group, 74% had documented pancreatic duct stenosis, and 77% had pancreatic duct dilatation (large duct disease). Pancreatic duct stones were present in 44% of the patients. Sixty percent of the patients had pseudocysts (Table 1), reflecting the high percentage of patients with large duct disease in our study group. Sixty-eight percent had calcifications. Calcifications were more frequent in alcoholic CP (74%) than in nonalcoholic CP (53%; p < 0.01). Seven percent of the patients had a pancreatic divisum (as potential etiology or coetiology of CP).
In the 201 patients undergoing pancreatic head resection (PD) or DPPHR, distal dilatation of the pancreatic duct was present in 78%. Sixty percent of those 201 patients had an inflammatory enlargement of the pancreatic head. Only 4% of the patients were documented as having neither an inflammatory mass of the pancreatic head nor a pancreatic duct dilatation nor a radiological stenosis of the common bile duct.
Surgery and Perioperative Management
The following types of pancreatic resection were performed in the 224 patients: PPPD (n = 89; 40%), DPPHR (n = 92; 41%; Beger 42 and Frey 50), classic Whipple operation (n = 20; 9%), distal pancreatectomy (n = 21; 9%), and two central pancreatic resections (1%).
The perioperative management of our patients has recently been described in detail.15 After the resectional part, the pancreatic duct was always cannulated to exclude remaining pancreatic duct stones or stenosis. After PD pancreatic anastomosis was performed as a single-layer end-to-side pancreatojejunostomy (91%), a duct-to-mucosa-technique with a pancreatic duct catheter (6%) or as pancreatogastrostomy (3%). After DPPHR according to Beger, the pancreatic anastomosis was also performed in an end-to-side technique using interrupted full-thickness polydioxanone sutures. During the Beger operation, a bilioenteric anastomosis to the posterior wall of the jejunal loop was included in 24 (57%) of the 42 patients. After the Frey resection, reconstruction consisted in a side-to-side pancreatojejunostomy using running polydioxanone sutures.
The bilioenteric anastomosis after PD was performed in an interrupted technique with polydioxanone sutures in almost all patients. A few patients with large common hepatic duct caused by extensive cholestasis underwent end-to-side hepaticojejunostomy using running sutures.
After distal pancreatic resection, 16 of 21 (76%) patients had a single-layer pancreatojejunostomy and 5 (24%) had a suture closure of the pancreatic stump. Stapler closure of the pancreatic stump was not used in our study patients.
Perioperative octreotide was almost always applied for 5 to 7 days in the first years of this study period. Its routine use was abandoned in 2003. Before abdominal closure, flat silicon drains were placed at the pancreatic anastomosis (and at the bilioenteric anastomosis, when performed) and taken out through the abdominal wall. These drains were left in place for at least 3 postoperative days.
Definitions
Our standardized definition of pancreatic leakage was reported in detail before15 and consisted in increased amylase in the drain output beyond the sixth postoperative day, the need of interventional drainage of abdominal fluid collections with a high amylase concentration or visible anastomotic insufficiency found during relaparotomy. All intraabdominal complications including gastrointestinal bleeding and wound infections were summarized as surgical complications.
The presence of diabetes was defined by the criteria of the WHO classification. Many patients underwent oral glucose tolerance tests or 24-h glucose profile determination.
Exocrine insufficiency was defined as the presence of steatorrhea and/or the need for oral pancreatic enzyme supplementation. In our complete study group, we did not routinely measure other parameters for exocrine function (e.g., stool elastase).
Follow-up
Postoperative follow-up examinations were performed in several chronological steps since 1996 in the form of mailed questionnaires (with or without additional telephone contact to the patient or home physician) or outpatient visits. They always included standardized questionnaires asking (among others) the presence of pain, pain intensity (including visual analog scales), pain frequency (none/daily/weekly/monthly/yearly), the presence of diabetes or steatorrhea, and the current specific medication (pancreatic enzymes, analgesics). In all follow-up questionnaires, the type of pain medications and the frequencies of their intake were evaluated. Furthermore, the need of reoperation was investigated.
In November and December 2005, mailed questionnaires were (again) sent to all eligible patients, and 130 surviving patients answered with completed questionnaires until end of January 2006 (among those 21 patients without prior follow-up data).
Eighty-six patients included in this series were part of a randomized study comparing PPPD and DPPHR. These patients all had regular outpatient follow-up including the questionnaires mentioned above plus quality-of-life measurements and oral glucose tolerance tests. The specific results of this randomized study (especially regarding quality of life) are not included in this manuscript and will be reported separately.
For this study, the results of the last follow-up evaluation per patient were considered. Median postoperative follow-up in all 224 patients was 56.3 (4–141) months. Median follow-up was about 1 year longer after PD (57 months) than after DPPHR (44 months). This may reflect the fact that DPPHR was performed with increasing frequency during the later study period.
Statistics
All perioperative and outcome data were entered into a computerized database (SPSS 13.0, SPSS Inc., Illinois, USA). Data acquisition after pancreatic resection is performed prospectively in our department.15 During subgroup analysis, comparisons were made by the chi-square and Mann–Whitney tests where appropriate. Potential risk factors for the long-term outcome parameters pain, endocrine and exocrine insufficiency were multivariately analyzed by a binary logistic regression model with a forward selection strategy using likelihood ratio statistics (inclusion and exclusion probability, p = 0.2). For the subgroup analyses of the influence of the different types of surgery on the outcome (n = 222 with PD, DPPHR, or distal resection), the two patients with segmental resection were excluded.
Results
Surgery and Perioperative Course
Median duration of surgery in all 224 patients was 400 min (range 160–870 min). Duration of surgery was 442.5 min (285–870) for PD, 377.5 min (195–740) for DPPHR, and 242.5 min (160–405) for distal resections.
The median number of intraoperatively transfused units of blood was 2 (range 0–36). The median of transfused units was 4 during PD and 2 during DPPHR and distal resection.
The total postoperative complication rate was 38%, with 28% of the patients having surgical complications (Table 2). The frequency of surgical complications did not differ significantly after PD, DPPHR, or distal resection. A reoperation for complications was necessary in 16 patients (7%). Median postoperative length of stay was 14 days (7–120).
Pain Assessment
At the last follow-up evaluation 134 (60%) patients reported no abdominal pain at all and 90 (40%) of the patients had abdominal pain. Of the 90 patients with abdominal pain, 12 (13%) had pain every day, 19 (21%) had pain at least once per week, 29 (32%) at least once per month, and 30 (33%) at least once per year. Subgroup analysis could not demonstrate a correlation between pain presence and type of surgery performed (Table 3). In addition, preoperative pain medication was not associated with pain or frequent pain during follow-up (Table 4). Univariate analysis of other potential risk factors for pain at the last follow-up evaluation showed that the absence of diabetes, a concomitant exocrine insufficiency, postoperative surgical complications, a shorter postoperative follow-up period (less than 5 years), and a shorter total duration of CP (less than 8 years) were significantly associated with a higher rate of abdominal pain (Table 4). In multivariate analysis, the concomitant absence of diabetes, a concomitant exocrine insufficiency, postoperative surgical complications, and a short postoperative follow-up period were independent risk factors for abdominal pain at the last follow-up evaluation (Table 5).
To further assess pain after resection for CP, the subgroup of patients with frequent abdominal pain (pain daily or at least once per week; n = 31) underwent again uni- and multivariate evaluations. Here, uni- and multivariate analysis revealed that the presence of exocrine insufficiency and former postoperative surgical complications were independent risk factors for the occurrence of frequent pain (at least once per week; Tables 4 and 5).
Pain Medication at Follow-up
At the last follow-up evaluation, 65 of the 90 patients (72%) complaining of abdominal pain took pain medication (10 had analgesics every day, 17 at least once per week, 20 at least once per month, and 18 at least once per year). Of these 65 patients 38 (58%) had opioids and 27 (42%) had peripheral analgesics. Univariate analysis could not identify risk factors for the use of pain medication at follow-up (in those patients with pain).
Endocrine Function
Sixty-eight patients (30%) had documented endocrine insufficiency preoperatively. Of those 68 patients with preoperative diabetes, five (7%) had no evidence of diabetes at the last follow-up (two after PD, two after DPPHR, and one after distal resection).
At the last follow-up evaluation, a total of 120 patients (54%) had diabetes. Fifty-seven of the 156 patients (37%) without preoperative diabetes became diabetic (de novo diabetes). Of those 156 patients, seven (4%) developed diabetes directly after surgery (three after PD, three after DPPHR, and one after distal resection), whereas 50 (32%) became diabetic during the postoperative follow-up period.
Univariate (Tables 6 and 7) and multivariate analyses revealed no statistically significant risk factors for the presence of diabetes or the development of postoperative de novo diabetes, although patients with regional/generalized portal hypertension, after distal pancreatic resection or with concomitant exocrine insufficiency, had slightly higher risks to be diabetic at the end of follow-up.
Exocrine Function
Preoperatively, 71 patients (32%) were assessed to have exocrine pancreatic insufficiency. During the postoperative follow-up period, 75 other patients (33.5%) developed exocrine insufficiency leading to a total number of 146 patients (65.2%) with exocrine pancreatic insufficiency at the last follow-up assessment (Table 8).
Univariate analysis revealed that the presence of portalvenous hypertension at the time of surgery, a longer preoperative duration of CP, and malnutrition (preoperative body mass index <20) were univariately associated with a higher probability of exocrine insufficiency at the last follow-up (Table 9). These three parameters were also independent risk factors for exocrine insufficiency after multivariate analysis (Table 5).
Malnutrition (low BMI) was the only risk factor for postoperative de novo exocrine insufficiency (Table 8). A multivariate analysis was, therefore, not performed for this entity.
Postoperative Weight Change
In all 224 patients, the median weight gain from the time of surgery to the last follow-up evaluation was 2 (range −31 to +37) kg. In subgroup analysis, patients without frequent pain (median weight difference +2 kg vs. −2 kg in patients with daily or weekly pain; p < 0.001) and patients with a preoperative BMI below 20 (median weight gain +4 kg vs. +1 kg in patients with a BMI > 20) had significantly higher weight gain. In other subgroup analyses (endocrine and exocrine function, type of surgery, surgical complication), no statistical differences regarding the postoperative body weight changes were found between the groups.
Anatomical Classification
The preoperative anatomical classification of CP (pancreatic duct dilatation, presence of an inflammatory mass of the pancreatic head, calcifications) did not show any influence on the main outcome parameters. The presence or absence of these mentioned characteristics had no effect on long-term pain, pain classification, exocrine or endocrine function (data not shown).
Late Morbidity/Organ Complications
Eleven patients underwent surgery for incisional hernia and two for small bowel obstruction caused by postoperative adhesions. The reasons for surgery for specific complications after pancreatic resection are listed in Table 10.
Symptomatic peptic ulcer disease was documented in nine patients (4%) during follow-up. After PPPD, seven (8%) of 109 patients developed peptic anastomotic or jejunal ulceration leading to reoperation in one of those. After classical Whipple procedure (n = 20), one patient had a peptic ulcer requiring surgery and one patient required completion pancreatectomy for recurrent CP.
Relevant biliary complications occurred in seven (3%) of the 224 patients (two stenoses [one after Beger procedure, one after Frey procedure]) and two impacted common bile duct stones (Frey) after DPPHR treated by surgery or ERCP; two stenoses of the bilioenteric anastomosis after PPPD; one new stenosis of the distal bile duct after former distal pancreatic resection as a result of recurrent CP in the pancreatic head.
Late Mortality
Long-term survival could be evaluated in the 224 patients of this study and nine more patients who died during the postoperative period without undergoing clinical follow-up examination. Thirty of those 233 patients died, a median of 3.2 years after pancreatic resection. Reasons for death were cardiac (n = 6), pancreatic cancer (n = 3), liver cirrhosis (n = 3), suicide (n = 3), ENT cancer (n = 2), one lung cancer, one bile duct cancer, and complications of diabetes in another patient. Reason for death is unknown in 10 of 30 patients.
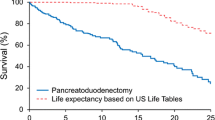
Five- and 10-year survival rates were 86 and 65%, respectively, in these 233 patients with a median age of 44 years at the time of pancreatic resection.
Discussion
In this study, we report the long-term outcome after pancreatic resection for CP in 224 patients with a postoperative follow-up period of up to 11.7 years. Prospective documentation of many perioperative parameters and standardized follow-up questionnaires enabled us to perform risk-factor analyses for the late postoperative outcome.
Most patients were referred by gastroenterologists, frequently after failed nonsurgical therapy of pain or complications of adjacent organs. The high frequencies of large duct disease, inflammatory mass, pseudocysts, and bile duct stenoses reflect the fact that many patients had a rather severe and/or advanced form of CP. Patients with small duct disease without complications other than pain were exceptional in our study group.
Although there is an ongoing debate on the type of surgery in subgroups of patients with large duct disease (resection versus drainage procedures alone), we believe that the majority of our patients had a clear indication for resection as defined by the aforementioned criteria (together with suspicion of malignancy in some of them). Our results, therefore, cannot be compared with studies also reporting good outcomes after surgical drainage alone.16,17
Follow-up evaluation was performed in several steps since 1996. By analysis of the questionnaires and/or documentation of postoperative outpatient visits, we judge the data regarding the presence of pain and diabetes as very reliable. About half of the patients pre- and postoperatively had oral glucose tolerance tests further determining endocrine function. Regarding pain, however, one cannot exclude that postoperative pain has other reasons than recurrent or ongoing pancreatitis (like, e.g., postoperative bowel adhesions) as indicated by the fact that postoperative surgical complications were a risk factor for abdominal pain. By analyzing the questionnaires or by telephone contact with the home physicians, however, we had the strong impression that many severe pain attacks in the postoperative period were caused by episodes of recurrent CP and often related to alcohol abuse. The evaluation of postoperative alcohol intake by patient interview or questionnaire has certainly a strong bias and was not performed in our study.
Because some patients were treated for postoperative (non-surgical) problems by their home-gastroenterologists or in the initially referring hospital, we have certainly not recorded all recurrent attacks of pancreatitis. We, therefore, did not include a detailed analysis of recurrent CP episodes in this paper. Regarding necessary reoperations, however, we probably have sufficient data of all patients.
Our results confirm many other reports that pancreatic resections can be performed with a very low mortality (1% in our overall patient group) and an acceptable morbidity in patients with chronic pancreatitis.10,13,18–20 Therefore, the long-term control of pain is the most important outcome parameter after surgical treatment of CP. In our patients, we could achieve a complete or satisfactory control of pain in more than 80%. Pain origin in patients with CP is not fully understood. Beyond increased intraductal pressure other factors like nerve alterations, fibrosis, or an inflammatory mass per se (“pacemaker”) may be responsible4,6. A concomitant exocrine insufficiency was one of two strong risk factors for abdominal pain in our study. Possible explanations here are advanced CP leading to exocrine insufficiency by parenchyma destruction and to recurrent pain by one of the aforementioned mechanisms. As already mentioned, former postoperative surgical complications were also an independent risk factor for late pain. The explanation for this phenomenon is difficult, and, we cannot exclude that intraabdominal adhesions may be (in part) responsible for pain in these patients. By uni- and/or multivariate analysis, a longer duration (total and preoperative, respectively) of CP was associated with fewer pain at last follow-up compared to patients with a shorter CP history. It is possible that in these patients with longstanding disease a “burn-out” of CP may be responsible as suggested by Ammann et al.21 or Layer et al.22
At the end of the follow-up period, 54% of the patients were diabetic. About half of those developed diabetes during the postoperative period (de novo diabetes). Only a small minority of de novo diabetes occurred directly as a consequence of surgery, but most de novo endocrine insufficiency developed later, probably as a consequence of further ongoing parenchyma destruction by CP.12,13,23 Surprisingly, we could not identify any risk factor for diabetes or de novo diabetes in our patients. It has been well described that distal resection bears a higher risk of postoperative diabetes,23 but we could at best see a tendency for a higher rate of de novo diabetes after distal resection. The presence of calcifications as risk factor for diabetes as described by Malka et al.23 was not confirmed in our patients.
As already reported by Beger et al.,20 we also had five patients with improvement of endocrine function after surgery, four after head resection, and even one after distal resection. This may be because of removal of an inflammatory process and/or pancreatic duct stenosis with subsequent improvement of function in the remaining pancreas.
Similar to endocrine insufficiency, about one-third of the patients presented with exocrine insufficiency before surgery and half of the remaining patients developed exocrine insufficiency during the postoperative follow-up period of almost 5 years. In contrast to diabetes, we could identify risk factors for late exocrine insufficiency. Patients with preoperative malnutrition (as defined by a BMI <20) developed postoperative de novo exocrine insufficiency significantly more frequently than patients with a BMI above 20 (although the rate of exocrine insufficiency was comparable before surgery). Summarized with further risk factors like a longer preoperative duration of CP or the presence of portalvenous hypertension, we can suggest that more severe or advanced forms of CP bear a higher risk to develop exocrine insufficiency.
It is somewhat surprising that the anatomical classification of CP (presence or absence of large duct disease, inflammatory mass, or calcifications) and the different types of surgery did not influence the main outcome parameters. Since 1996, the type of surgery was always adapted to the underlying pathological condition (also in the 86 patients randomized for PPPD or DPPHR) with the intention to leave as much pancreatic tissue as possible after removal of the part potentially responsible for the complication leading to surgery. It is, therefore, possible that this adapted surgical strategy may be responsible for those comparable long-term results.
Reoperation or reintervention for pain or recurrence of organ complications was rarely necessary in our patients. As published before,24 peptic ulcer disease, often in the form of jejunal ulcers, is a specific complication after PD (especially after PPPD) and required reoperation in two cases. Biliary complications occurred in a few patients because of stenoses of a bilioenteric anastomosis, recurrent CP, or insufficient biliary decompression during primary Frey operation.
Late mortality in patients with CP is clearly higher than in the normal population.3,12,22,24 The 10-year survival in our patient group was even poorer than the data reported from the Mayo group,25 although the median age of our patients was almost 4 years lower than in their study. Strate et al.12 from Hamburg reported a mortality rate of 24% of the patients during a follow-up of almost 9 years. Although patients with alcoholic CP were found to have a higher mortality rate this was not the case in our patients. The reasons for death were heterogeneous, and we had only a few patients dying from pancreatic cancer in our series. The high late mortality rate is probably mainly caused by a high comorbidity from alcohol abuse and smoking.3,12,24
During the last decade, several reports of the long-term outcome after surgery for chronic pancreatitis with follow-up periods between 34 and 104 months and relevant number of patients (57–504 patients) have been published.12,13,16–18,20,24,26,27 One study randomized patients to undergo either the Frey or the Beger procedure; all other studies reported results of one or more types of surgery (including also drainage operations without resection;16,17) in a nonrandomized fashion.12 Complete or substantial pain relief was reported in between 65 and 91%. The frequency of exocrine and endocrine insufficiency (total or postoperative de novo) was not reported in all of those papers. However, when data were available, between 30%19 and almost 60%12 of the patients had diabetes at the end of the observation period, in part dependent on the length of follow-up. In our patients, one-third of preoperatively nondiabetic patients developed de novo diabetes (25% of all patients), which is in the range of other reports. In analysis of the literature, it seems evident that the majority of postoperative de novo diabetes is not related to surgery itself, but develops during the later disease course even in patients without pain. Long-term exocrine insufficiency is reported at even higher rates than diabetes with frequencies up to 83%12 in patients with long follow-up periods. As for diabetes, there seems to be no or less impact of surgery on the development of exocrine insufficiency, but rather an effect of longstanding disease as outlined by Ammann et al.21
There is still a controversial discussion about the appropriate type of surgery in CP, especially in those patients with large duct disease and absence of other organ complications, large inflammatory masses, or suspicion of malignancy. In North America, many centers prefer drainage procedures, whereas other centers in Europe promote resectional procedures. Arguments to favor resections may be a frequent failure of pain relief by drainage procedures reported in some studies.6,21,28
Because of rather comparable results especially regarding pain relief, the current literature, however, suggests that the choice of the type of surgery depending on the underlying complications other than pain (e.g., duct stenosis, portalvenous compression) and anatomy (size of the pancreatic duct, inflammatory masses) may be the most important measure to obtain these rather good outcome reported in most studies. In the only randomized study with a substantial follow-up time, the group from Hamburg found comparable long-term outcomes after the Frey or the Beger procedure after almost 9 years.12 In our study, outcomes were comparable between the different procedures. During the initial learning curve of our group, more PDs were performed for CP predominantly of the pancreatic head. Outside of our randomized study performed between 1997 and 2001, we since then have chosen the type of head resection dependent on the presence and type of local complications with less extended operations (Frey) preferred when possible.
Conclusion
Pancreatic resection leads to adequate pain relief and control of organ complications in the majority of patients with CP. Long-term outcome does not depend on the type of surgical procedure, but is in part influenced by severe and/or advanced preoperative CP and by postoperative surgical complications (regarding pain). As reported before, however, the majority of patients develop exocrine and endocrine insufficiency unrelated to surgery. A few patients develop procedure-related late complications requiring reintervention. Late mortality is high, probably because of the high comorbidity (alcohol, smoking) in many of these patients.
References
Rosch T, Daniel S, Scholz M, Huibregtse K, Smits M, Schneider T, Ell C, Haber G, Riemann JF, Jakobs R, Hintze R, Adler A, Neuhaus H, Zavoral M, Zavada F, Schusdziarra V, Soehendra N. Endoscopic treatment of chronic pancreatitis: a multicenter study of 1000 patients with long-term follow-up. Endoscopy 2002;34:765–771.
Delhaye M, Arvanitakis M, Verset G, Cremer M, Deviere J. Long-term clinical outcome after endoscopic pancreatic ductal drainage for patients with painful chronic pancreatitis. Clin Gastroenterol Hepatol 2004;2:1096–1106.
Steer ML, Waxman I, Freedman S. Chronic pancreatitis. N Engl J Med 1995;332:1482–1490.
Di Sebastiano P, di Mola FF, Bockman DE, Friess H, Buchler MW. Chronic pancreatitis: the perspective of pain generation by neuroimmune interaction. Gut 2003;52:907–911.
Beger HG, Buchler M. Duodenum-preserving resection of the head of the pancreas in chronic pancreatitis with inflammatory mass in the head. World J Surg 1990;14:83–87.
Markowitz JS, Rattner DW, Warshaw AL. Failure of symptomatic relief after pancreaticojejunal decompression for chronic pancreatitis. Strategies for salvage. Arch Surg 1994;129:374–379.
Beger HG, Krautzberger W, Bittner R, Buchler M, Limmer J. Duodenum-preserving resection of the head of the pancreas in patients with severe chronic pancreatitis. Surgery 1985;97:467–473.
Frey CF, Smith GJ. Description and rationale of a new operation for chronic pancreatitis. Pancreas 1987;2:701–707.
Traverso LW, Longmire WP, Jr. Preservation of the pylorus in pancreaticoduodenectomy. Surg Gynecol Obstet 1978;146:959–962.
Sakorafas GH, Farnell MB, Nagorney DM, Sarr MG, Rowland CM. Pancreatoduodenectomy for chronic pancreatitis: long-term results in 105 patients. Arch Surg 2000;135:517–523.
Schafer M, Mullhaupt B, Clavien PA. Evidence-based pancreatic head resection for pancreatic cancer and chronic pancreatitis. Ann Surg 2002;236:137–148.
Strate T, Taherpour Z, Bloechle C, Mann O, Bruhn JP, Schneider C, Kuechler T, Yekebas E, Izbicki JR. Long-term follow-up of a randomized trial comparing the beger and frey procedures for patients suffering from chronic pancreatitis. Ann Surg 2005;241:591–598.
Traverso LW, Kozarek RA. Pancreatoduodenectomy for chronic pancreatitis: anatomic selection criteria and subsequent long-term outcome analysis. Ann Surg 1997;226:429–435.
Buchler MW, Friess H, Muller MW, Wheatley AM, Beger HG. Randomized trial of duodenum-preserving pancreatic head resection versus pylorus-preserving Whipple in chronic pancreatitis. Am J Surg 1995;169:65–69.
Adam U, Makowiec F, Riediger H, Schareck WD, Benz S, Hopt UT. Risk factors for complications after pancreatic head resection. Am J Surg 2004;187:201–208.
Nealon WH, Matin S. Analysis of surgical success in preventing recurrent acute exacerbations in chronic pancreatitis. Ann Surg 2001;233:793–800.
Lucas CE, McIntosh B, Paley D, Ledgerwood AM, Vlahos A. Surgical decompression of ductal obstruction in patients with chronic pancreatitis. Surgery 1999;126:790–795.
Sohn TA, Campbell KA, Pitt HA, Sauter PK, Coleman JA, Lillemo KD, Yeo CJ, Cameron JL. Quality of life and long-term survival after surgery for chronic pancreatitis. J Gastrointest Surg 2000;4:355–364.
Jimenez RE, Fernandez-Del Castillo C, Rattner DW, Chang Y, Warshaw AL. Outcome of pancreaticoduodenectomy with pylorus preservation or with antrectomy in the treatment of chronic pancreatitis. Ann Surg 2000;231:293–300.
Beger HG, Schlosser W, Friess HM, Buchler MW. Duodenum-preserving head resection in chronic pancreatitis changes the natural course of the disease: a single-center 26-year experience. Ann Surg 1999;230:512–519.
Ammann RW, Muellhaupt B. The natural history of pain in alcoholic chronic pancreatitis. Gastroenterology 1999;116:1132–1140.
Layer P, Yamamoto H, Kalthoff L, Clain JE, Bakken LJ, DiMagno EP. The different courses of early- and late-onset idiopathic and alcoholic chronic pancreatitis. Gastroenterology 1994;107:1481–1487.
Malka D, Hammel P, Sauvanet A, Rufat P, O’Toole D, Bardet P, Belghiti J, Bernades P, Ruszniewski P, Levy P. Risk factors for diabetes mellitus in chronic pancreatitis. Gastroenterology 2000;119:1324–1332.
Sakorafas GH, Farnell MB, Farley DR, Rowland CM, Sarr MG. Long-term results after surgery for chronic pancreatitis. Int J Pancreatol 2000;27:131–142.
Sakorafas GH, Sarr MG. Pancreatic cancer after surgery for chronic pancreatitis. Dig Liver Dis 2003;35:482–485.
Witzigmann H, Max D, Uhlmann D, Geissler F, Schwarz R, Ludwig S, Lohmann T, Caca K, Keim V, Tannapfel A, Hauss J. Outcome after duodenum-preserving pancreatic head resection is improved compared with classic Whipple procedure in the treatment of chronic pancreatitis. Surgery 2003;134:53–62.
Evans JD, Wilson PG, Carver C, Bramhall SR, Buckels JA, Mayer AD, McMaster P, Neoptolemos JP. Outcome of surgery for chronic pancreatitis. Br J Surg 1997;84:624–629.
Greenlee HB, Prinz RA, Aranha GV. Long-term results of side-to-side pancreaticojejunostomy. World J Surg 2006;14:70–76.
Author information
Authors and Affiliations
Corresponding author
Additional information
DISCUSSION
Dr. W. Nealon (Galveston, TX): Dr. Kaufman, Dr. Joseph, members and guests. The authors from Freiburg have contributed yet another thorough review of their outcomes in the management of chronic pancreatitis. They report today their experience with 272 resections, including pancreaticoduodenectomy, classic Whipple, and duodenum-preserving pancreatic head resection either by the Frey or Beger technique, and they provide detailed follow-up data, including measures of pain relief, functional derangements, and nutritional outcomes.
Their operative mortality of 1% is striking, particularly considering their inclusion of patients with portal vein or splenic vein thrombosis, which they have seen in 25% of their resected patients, and often this entity raises considerably the risk for hemorrhage in these resections. Complete abolition of pain was achieved in 60% and some reduction in pain was achieved in 86%. These are superb outcomes. Two-thirds of the patients had steatorrhea in follow-up, half acquired after surgery; 37% developed new-onset diabetes. Again, all these are acceptable rates. Notably, 86% of patients survived 5 years and 65% 10 years, reflecting the known chronic nature of this disease and the likely ongoing ravages of alcoholism. Nutritional improvements were noted in a high percentage of patients independent of their pancreatic function after operation, an observation we have made in the past, and a striking one, I believe.
I have three questions. Can you review with us your formula for choosing among the operations that are available for resection, particularly how you decide between a Frey’s procedure and a Begar procedure?
Number two, you don’t mention narcotics use, and I must say in my huge experience with these patients this overshadows every bit of patient management. I am wondering if you monitor this and whether you have any thoughts on the impact of narcotic dependence on the management of these patients after surgery?
And number three, regarding exocrine insufficiency, there are some mysteries you have exposed. It was shown patients with postop exocrine insufficiency correlated with persistent pain, and your patients with low pre-op BMI had a higher chance of developing exocrine insufficiency and a higher risk of persistent pain. Do you have any thoughts on the connection between this exocrine function and the pain?
I congratulate you and your colleagues on a thorough and superbly analyzed review of these very complex patients.
Dr. Makowiec: Thank you, Dr. Nealon. Your first question was about the choice of the type of operation. After all the experience we have with our patients and our studies, we try to perform the less invasive or the smallest operation possible. That means, in ascending order of complexity, the Frey operation, the Beger operation and PD depending on the anatomy of chronic pancreatitis and the organ complications. For example, patients with pancreatic duct dilatation and a small or medium sized pancreatic head mass will receive a Frey operation. If a patient has a larger mass, a lot of fibrosis in the pancreatic head with concomitant bile duct stenosis, he will undergo Beger’s procedure. In more than 50% of the Beger operations in our series a bilioenteric anastomosis was included, with good results in the vast majority regarding clearance of the bile ducts. If patients have a suspicion of malignancy or an enormous inflammatory mass of the pancreatic head with duodenal destruction, destruction of the antrum of the stomach, we perform a pylorus-preserving pancreaticoduodenectomy.
Regarding the second question, we have no reliable data on the use of narcotics before and after surgery. Preoperatively about one-fourth of the patients took opioids, and three-fourths of the patients had some form of peripheral analgesics. In our follow-up examinations we always asked for the use of narcotics and other analgetics, but I think that the results were not very reliable, again related to the use of alcohol. So we don’t think that the data given by the patients regarding pain medications are scientifically reliable.
Your third question was about the correlation between pain, exocrine insufficiency, and body mass index. We saw that patients with exocrine insufficiency had, more frequently, pain. I think that this is a sign of advanced disease. Another reason is probably, especially patients with severe pain and frequently recurrent pain, the continuous use of alcohol. As for informations about pain medication, this information is also not very reliable. However, during many phone contacts with home physicians, we heard that in most cases with severe or frequent pain these patients continue to drink.
Low body mass index is probably a sign of severe and advanced chronic pancreatitis. These patients are malnourished just because they can’t eat more due to abdominal pain.
Dr. L. Traverso (Seattle, WA): Frank, thank you for showing us all these details in just 10 minutes. I have three questions. When we reviewed our patients after a five-year follow-up following the Whipple operation for chronic pancreatitis those with diabetes preoperatively had better pain relief. Did you see that association?
Number two, we found it valuable to compare the patient’s pre-op pain to their post-op pain and whether they had received some benefit from the operation. With that method we observed every patient indicated they were improved and 76% had complete pain relief.
Third, we all have to be accountable when deciding to resect the head of the pancreas in these patients with documented chronic pancreatitis and chronic abdominal pain. The best way is to make sure they really have severe chronic pancreatitis before resection. The first slide you showed on preoperative ductal anatomy indicated that about 87% or so had large duct disease. From that slide, a little over 10% of your patients could have had a normal pancreatic duct. Was that the case? Did they all have abnormal pancreatic ducts? The method that we used was the Cambridge Classification of Image Severity described by Axon (Axon ATR, Classen M, Cotton PB, et al. Pancreatography in chronic pancreatitis: international definitions. Gut 1984; 25:1107-1112). To qualify for resection a patient had to have the worst case of image severity, i.e., stage IV. The minimal disease required to have that category was a major pancreatic duct stricture in the head, with or without stones, with or without duct dilatation. When pancreatic surgeons are accountable that way then almost every patient after head resection for true chronic pancreatitis will get pain relief, provided they don’t start drinking alcohol again.
Dr. Makowiec: Regarding our data, I can confirm some inverse correlation between the presence of diabetes and pain. We found that the absence of diabetes was a risk factor for pain. I have no explanation for this phenomenon.
Many of the patients who noted to have pain on their questionnaires also noted that they are satisfied with the operation because they had clearly less pain. Regarding pain assessment we had one problem: In 60% of our patients we had no complete preoperative pain documentation regarding frequency or a visual analog scale. So we can hardly compare the data.
About 20 % of our patients had no relevant dilatation of the pancreatic duct. However, most of those had inflammatory masses and/or bile duct stenosis. I, therefore, agree completely with you that pancreatic head resection may be appropriate in these cases, even without large duct disease.
Rights and permissions
About this article
Cite this article
Riediger, H., Adam, U., Fischer, E. et al. Long-term Outcome After Resection for Chronic Pancreatitis in 224 Patients. J Gastrointest Surg 11, 949–960 (2007). https://doi.org/10.1007/s11605-007-0155-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-007-0155-6