Abstract
Purpose
To evaluate the usefulness of the deep learning image reconstruction (DLIR) to enhance the image quality of abdominal CT, compared to iterative reconstruction technique.
Method
Pre and post-contrast abdominal CT images in 50 patients were reconstructed with 2 different algorithms: hybrid iterative reconstruction (hybrid IR: ASiR-V 50%) and DLIR (TrueFidelity). Standard deviation of attenuation in normal liver parenchyma was measured as the image noise on pre and post-contrast CT. The contrast-to-noise ratio (CNR) for the aorta, and the signal-to-noise ratio (SNR) of the liver were calculated on post-contrast CT. The overall image quality was graded on a 5-point scale ranging from 1 (poor) to 5 (excellent).
Results
The image noise was significantly decreased by DLIR compared to hybrid-IR [hybrid IR, median 8.3 Hounsfield unit (HU) (interquartile range (IQR) 7.6–9.2 HU); DLIR, median 5.2 HU (IQR 4.6–5.8), P < 0.0001 for post-contrast CT]. The CNR and SNR were significantly improved by DLIR [CNR, median 4.5 (IQR 3.8–5.6) vs 7.3 (IQR 6.2–8.8), P < 0.0001; SNR, median 9.4 (IQR 8.3–10.1) vs 15.0 (IQR 13.2–16.4), P < 0.0001]. The overall image quality score was also higher for DLIR compared to hybrid-IR (hybrid IR 3.1 ± 0.6 vs DLIR 4.6 ± 0.5, P < 0.0001 for post-contrast CT).
Conclusions
Image noise, overall image quality, CNR and SNR for abdominal CT images are improved with DLIR compared to hybrid IR.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Multidetector computed tomography (CT) plays a pivotal role in the diagnosis of abdominal diseases with continuous technological progressions. The recent technical developments include the introduction of image reconstruction algorithms that improve and optimize data processing to allow for radiation dose reduction while maintaining diagnostic image quality [1,2,3,4]. Since 2009, manufacturers have adapted several types of iterative reconstruction (IR) algorithms for use in CT [5,6,7,8]. IR techniques are classified as hybrid- and model-based [9]. In hybrid IR, the projection data are iteratively filtered to reduce artifacts, and after the backward projection step, the image data are iteratively filtered to reduce image noise. Hybrid IR technology has the potential to improve the image quality in the abdomen and facilitate a reduction in radiation dose compared to previous techniques [10]. Hybrid IR also has the advantage of allowing much faster image reconstruction than model-based IR (MBIR), since it is a less complex model with less emphasis on system optics than MBIR. One important drawback of hybrid IR is that high levels of IR results in over-smoothing or plastic image features. This is one reason that the maximal level of IR technique is typically not used in a clinical setting. MBIR uses a probabilistic method, deriving a statistical cost function by incorporating X-ray physics and CT optics modeling to reduce noise and artifacts. MBIR can provide dramatic noise reduction but requires significantly longer reconstruction time, and there are concerns regarding its associated altered image texture [3, 11,12,13].
Deep learning image reconstruction (DLIR) is a recently developed approach for CT reconstruction, which incorporates deep convolutional neural networks (DCNN) into the image reconstruction algorithm. The DLIR algorithms developed by GE Healthcare and Canon Medical system feature a DCNN, which is trained respectively with high-quality filtered back projection (FBP TrueFidelity™, GE Healthcare) and MBIR [Advanced intelligent Clear-IQ Engine (AiCE), Canon Medical System] datasets to learn how to differentiate noise from signals [14, 15]. During the training, the DCNN analyzes the data and synthesizes a reconstruction function (the inference engine), which is optimized through the learning process [16]. The inference engine has been tested and developed using a large test dataset for validation. The deep learning technology has recently demonstrated great potential, to reconstruct CT images while suppressing noise without changing noise texture or affecting anatomical and pathological structures [14, 16, 17].
The purpose of this study was to evaluate our initial experience with the DLIR algorithm TrueFidelity in abdominal CT using qualitative reader evaluations and quantitative assessments in comparison with our standard hybrid IR method.
Materials and methods
Study population
Fifty consecutive patients (23 men, 27 women, age range 21–83 years; mean age, 60 ± 16 years) who had pre-contrast CT and post-contrast CT acquired at 2 min after contrast injection covering the upper abdomen at our institution between August and September 2019 were retrospectively evaluated in this study. Written informed consent for undergoing contrast enhanced CT were obtained in all patients. The body mass index of the subjects ranged from 15.9 to 33.0 (mean, 23.0 ± 4.9). The clinical indications for the abdominal CT were as follows: screening for liver tumor (n = 33), follow-up after surgery for gastric tumor (n = 7), renal tumor (n = 5) and duodenal tumor (n = 1), evaluation of adrenal grand (n = 2), and follow-up for liver metastases (n = 2). This retrospective study was approved by our institutional review board and written informed consent was waived since this study used existing clinical CT image data. The opportunity to opt-out of the inclusion to this study was given through a notice in the hospital website. No patient showed intention for an exclusion from this study.
CT image acquisition and reconstruction
All acquisitions were performed with a multidetector CT scanner (Revolution CT, GE Healthcare, Milwaukee, WI, USA). The scan protocols for abdominal CT were identical to the setting used clinically at our institution and were as follows: matrix 512 × 512, noise index 20, tube voltage 120 kV, tube current 100–740 mA with automatic exposure control, rotation time 0.6 s, pitch factor 0.992, z-axis coverage 80 mm. Nonionic contrast material (approximately 520 mgI/kg body weight) was administered intravenously for 80 s by means of a power injector. The scanning delays of the post-contrast CT were approximately 120 s after the start of the contrast material injection. All images were reconstructed with a slice thickness of 1.25 mm combined with two different algorithms: (a) hybrid IR (ASiR-V, GE Healthcare, Milwaukee, WI, USA) at 50% blending factor with a standard kernel, and (b) DLIR (TrueFidelity, GE Healthcare, Milwaukee, WI, USA). For the setting of TrueFidelity, there are three selectable reconstruction strength levels (Low, Medium, High) to control the amount of noise reduction. In this study, the highest strength level was used for the DLIR. The calculation time of ASiR-V and DLIR in the abdominal CT of this study was approximately 16 s and 50 s, respectively.
Quantitative analysis of CT images
The pre and post-contrast CT images with a slice thickness of 1.25 mm was objectively analyzed by one board-certified radiologist (Y.K. with 6 years of radiology experience). Liver attenuation was measured as the mean CT value of four regions of interest (ROIs) with approximately 150 mm2 in the right anterior, right posterior, left medial, and left lateral segment of the liver on pre and post-contrast CT images. Care was taken to avoid superimposition of the ROIs on the large vessels in the liver. In addition, ROIs were placed within the aorta and the portal vein for the attenuation measurements on post-contrast CT. The aortic attenuation was recorded as the mean measurement value of two ROIs at the celiac artery level and superior mesenteric artery level. The portal vein attenuation was recorded as the mean measurement value of two ROIs in the right and left portal vein. The ROIs in the aorta and portal veins were manually drawn as large as the vessel lumen.
On post-contrast CT, aortic and portal vein contrast-to-noise ratio (CNR) was calculated as follows:
where ROIvessel is the mean attenuation of the vessel of interest, ROIliver the mean attenuation of the liver parenchyma, and N is the noise determined as SD in the liver parenchyma.
On pre and post-contrast CT images, liver signal-to-noise ratios (SNR) was calculated as follows:
Qualitative analysis of CT images
Two experienced radiologists independently performed quantitative analysis of the pre and post-contrast CT images. For the qualitative image analysis, there were two steps—(1) training session and (2) assessment session. In the training session, the two readers overviewed the image noise and image quality on pre and post-contrast abdominal CT images reconstructed with hybrid-IR in 20 patients (11 men, 9 women; age range 43–80 years; mean age, 67 ± 11 years; mean body mass index, 22.0 ± 4.2) not included in this study subjects. The images were presented on a preset soft tissue window; the window width and level were 240 and 40 Hounsfield unit (HU), respectively. The averaged image noise and quality observed in the training session was regarded as reference standard for the subsequent visual analysis. In the assessment session, the readers qualitatively evaluated image noise and overall image quality on the pre and post-contrast CT reconstructed with hybrid-IR and DLIR in 50 subjects. The images were presented in random order on a preset soft tissue window; the window width and level were 200 HU and 20 HU on pre-contrast CT, and 240 HU and 40 HU on post-contrast CT, respectively. The observers were allowed to scroll and adjust window level and width while reviewing the cases. The readers were blinded to all patient demographics and CT parameters. Image noise was qualitatively assessed and was graded on a 5-point scale: score 1 = substantially increased in image noise, score 2 = mildly increased in image noise, score 3 = comparable image noise to CT observed in the training session, score 4 = mildly decreased in image noise, and score 5 = substantially decreased in image noise. Vessel conspicuity (visibility of small structures, especially the detection of the segmental branch level of the portal vein) on post-contrast CT images was graded on a 5-point scale: score 1 = very poor, score 2 = suboptimal, score 3 = comparable vessel conspicuity to CT observed in the training session, score 4 = above average, and score 5 = excellent. The overall image quality was also scored on a 5-point scale: score 1 = poor image quality, score 2 = slightly degraded image quality, score 3 = comparable image quality observed in the training session, score 4 = slightly improved image quality, and score 5 = substantially improved image quality.
Radiation dose
To assess radiation exposure, the CT dose index (CTDIvol), the dose-length product (DLP), the effective radiation, and the size-specific dose estimate (SSDE) [18, 19] was recorded for each patient. The effective radiation dose was calculated as the product of the DLP multiplied by a conversion coefficient for the abdomen (k = 0.015) [20].
Statistical analysis
All continual valuables are expressed as median and interquartile range (IQR). The scores for image noise and overall image quality are expressed as mean ± standard deviation. Wilcoxon matched-pairs signed-rank test was used to analyze differences in the image noise, CNR, SNR, and the scores of image noise, vessel conspicuity, and overall image quality between hybrid-IR and DLIR CT images. Differences were considered to be statistically significant at P < 0.05. Statistical analysis was performed with IBM SPSS Statistics version 25 (IBM Corporation, New York, USA).
Results
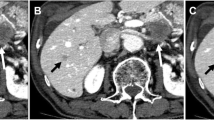
Representative cases are shown in Figs. 1 and 2.
Radiation exposure
The median CTDIvol, DLP, effective dose, and SSDE values for abdominal scans were 15.1 mGy (IQR 11.9–20.9 mGy), 964.7 mGy · cm (IQR 692.4–1389.2 (mGy · cm)), 14.5 mSv (IQR 10.4–20.8 (mSv)), and 22.1 mGy (IQR 18.2–28.1 mGy), respectively. They were slightly lower compared to conventional abdominal CT reported as the Japanese diagnostic reference levels [21].
Quantitative analysis of image noise, CNR, and SNR
The image noise was significantly decreased by DLIR compared to hybrid IR on pre-contrast CT (hybrid IR: median, 8.3 Hounsfield unit (HU) (IQR, 7.6–9.2 HU); DLIR: median, 5.2 HU (IQR 4.6–5.8 HU), P < 0.0001) as well as post-contrast CT (hybrid IR 8.9 HU (IQR 8.2–9.8 HU); DLIR 5.5 HU (IQR 5.1–6.2 HU), P < 0.0001) (Fig. 3). The mean reduction rate of the image noise by DLIR on pre and post-contrast CT between 35.5 ± 15.8% and 38.0 ± 2.5%, respectively, compared to hybrid-IR method. There was significant improvement of the CNR of aorta and portal vein by DLIR method on post-contrast CT [aorta: median, 4.5 (IQR 3.8–5.6) vs 7.3 (IQR 6.2–8.8), P < 0.0001; portal vein: 5.3 (IQR 4.5–6.3) vs 8.5 (IQR 6.8–10.3), P < 0.0001]. In addition, SNRs on pre and post-contrast CT were significantly improved by DLIR compared to hybrid-IR [pre-contrast CT: 6.1 (IQR 5.4–6.7) vs 9.6 (IQR, 8.5–10.9), P < 0.0001; post-contrast CT: 9.4 (IQR 8.3–10.1) vs 15.0 (IQR 13.2–16.4), P < 0.0001].
Qualitative image analysis
The results of image quality scores are shown in Table 1. There was significant improvement in image noise score by DLIR method compared to hybrid IR [observer 1: hybrid-IR 3.0 ± 0.7 vs DLIR 4.4 ± 0.6, P < 0.0001 (pre-contrast CT); observer 2: hybrid IR 3.1 ± 0.6 vs DLIR 4.0 ± 0.5, P < 0.0001 (pre-contrast CT)]. There was significant improvement in vessel conspicuity by DLIR method compared to hybrid IR (observer 1: hybrid IR 3.1 ± 0.8 vs DLIR 3.7 ± 1.0, P < 0.0001; observer 2: hybrid IR 3.2 ± 0.9 vs DLIR 4.2 ± 0.4, P < 0.0001). Overall image quality score on DLIR images was significantly higher than that on hybrid IR images [observer 1: hybrid-IR 3.1 ± 0.7 vs DLIR 4.3 ± 0.6, P < 0.0001 (pre-contrast CT); observer 2: hybrid IR 3.2 ± 0.5 vs DLIR 3.9 ± 0.6, P < 0.0001 (pre-contrast CT)]. All DLIR images received a score of 3 or higher for both image noise score and overall image quality score. Pre-contrast DLIR images received a rating of score 4 or 5 for image noise in 94% (47/50) and 84% (42/50) of subjects from observer 1 and 2, respectively. Post-contrast DLIR images received a rating of score 4 or 5 for image noise in 94% (47/50) and 96% (48/50) of subjects from observer 1 and 2, respectively.
Discussion
The present study demonstrated that the recently developed DLIR substantially reduced image noise and improved image quality on pre- and post-contrast abdominal CT, compared to hybrid-IR technique with a blending factor of 50%.
CT manufacturers have recently developed algorithms that use deep learning convolutional neural networks to perform image reconstruction. There are currently two commercialized CT reconstruction tools using DLIR—Advanced intelligent Clear IQ Engine (AiCE; Canon Medical Systems) and TrueFidelity (GE Healthcare). AiCE is the first commercialized deep learning reconstruction tool. For the deep learning approach, training pairs of hybrid-IR images and high-dose MBIR images are given [14]. Tatsugami et al. [17] recently conducted a retrospective study that investigated the value of the DLIR method for improving image quality of coronary CT angiography in 30 patients. They found that the mean image noise, represented by SD of attenuation in the paraspinal muscle, was significantly improved by the DLIR method compared to hybrid IR (18.5 ± 2.8 HU vs. 23.0 ± 4.6 HU, P < 0.01). The study also demonstrated that the CNR in the proximal left coronary arteries was significantly higher than that on hybrid IR (22.8 ± 5.0 HU vs. 26.3 ± 5.0 HU, P < 0.01). Akagi et al. [14] demonstrated that image noise, overall image quality, and CNR on hepatic ultra-high-resolution dynamic CT were significantly improved with the DLIR compared to hybrid-IR and MBIR. As the deep learning convolutional neural network is trained with MBIR images, the DLIR approach using AiCE has potential to generate comparable image quality to the MBIR images [14].
Following AiCE, TrueFidelity has recently become available for clinical use, and its usefulness in improving CT image quality has been verified by phantom experiments [22,23,24]. Several studies on the clinical application of TrueFidelity have also been reported [16, 25,26,27], and it is expected to be used in daily clinical practice. Benz et al. retrospectively evaluated the value of the TrueFidelity for improving image quality of coronary CT angiography in 43 patients, compared to ASiR-V 70% [25]. They demonstrated that TrueFidelity yielded a substantial reduction in image noise of up to 43% on coronary CT angiography, compared to ASiR-V 70%, while diagnostic accuracy of CCTA reconstruction by TrueFidelity remained equal to ASiR-V 70% using invasive coronary angiography as the standard of reference. The usefulness of TrueFidelity in the abdominal region has been reported by Jensen et al. [16]. They retrospectively investigated the value of TrueFidelity for improving image quality of portal venous abdominal CT, compared to ASiR-V 30%, and found that TrueFidelity was scored significantly better for overall image quality, lesion diagnostic confidence, artifacts, and image noise and texture. TrueFidelity, like AiCE, is a promising technology for improving CT image quality, and further accumulation of clinical evidence will be necessary for its effective use in the future.
IR algorithms have limitations on significantly reducing the radiation dose on abdominal CT while preserving diagnostic performance for low-contrast detection tasks (e.g., detection of liver metastases on abdominal CT images) [11, 13, 28, 29]. Jensen et al. [13] recently performed a prospective study that included 52 adults with 233 colorectal liver metastases who underwent routine-dose (mean CTDIvol, 25.8 mGy) and reduced dose (mean dose reduction, 54%) scans within the same breath hold, and compared observer performance between routine-dose FBP and reduced-dose IR (ASiR-V 60%). Lower-dose CT examinations (mean CTDIvol, 11.8 mGy) using IR demonstrated decreased observer performance, primarily owing to suboptimal detection of subcentimeter lesions. Whether the new DLIR technology will enable further radiation dose reduction while preserving diagnostic performance for low-contrast detection tasks compared to current IR algorithms are still unknown. This issue needs to be explored in further studies on the diagnostic performance of DLIR images for the detection of low-contrast lesions.
Our study has some limitations. The study population was relatively small, and the research was retrospectively conducted at a single institution. Second, the current study focused on subjective and objective assessments of image quality. We did not assess the impact of DLIR on the diagnostic performance. Third, while there are three selectable reconstruction strength levels (Low, Medium, High) to control the amount of noise reduction for TrueFidelity, this study evaluated only one strength level parameter (High) for the DLIR algorithm. Prior to the clinical implementation of this new technology, further rigorous evaluation may be necessary to determine the optimal intensity level for the DLIR algorithm. Furthermore, only the blending factor of 50% was used for ASiR-V in this study. Changing the blending factor for ASiR-V may have a significant impact on the study results. In this study, the potential ability of the recently-developed DLIR to reduce image noise was examined in comparison to ASiR-V with an intermediate blend factor setting, which is commonly used in abdominal CT. Forth, analysis of the noise texture on the CT images was not performed in this study.
Conclusion
Image noise, overall image quality, CNR and SNR on pre- and post-contrast abdominal CT images are significantly improved with the DLIR method compared to hybrid-IR technique.
References
Shuman WP, Chan KT, Busey JM, Mitsumori LM, Choi E, Koprowicz KM, Kanal KM. Standard and reduced radiation dose liver CT images: adaptive statistical iterative reconstruction versus model-based iterative reconstruction-comparison of findings and image quality. Radiology. 2014;273(3):793–800.
Kataria B, Althen JN, Smedby O, Persson A, Sokjer H, Sandborg M. Assessment of image quality in abdominal CT: potential dose reduction with model-based iterative reconstruction. Eur Radiol. 2018;28(6):2464–73.
Ichikawa Y, Kitagawa K, Nagasawa N, Murashima S, Sakuma H. CT of the chest with model-based, fully iterative reconstruction: comparison with adaptive statistical iterative reconstruction. BMC Med Imaging. 2013;13:27.
Deak Z, Grimm JM, Treitl M, Geyer LL, Linsenmaier U, Korner M, Reiser MF, Wirth S. Filtered back projection, adaptive statistical iterative reconstruction, and a model-based iterative reconstruction in abdominal CT: an experimental clinical study. Radiology. 2013;266(1):197–206.
Mitsumori LM, Shuman WP, Busey JM, Kolokythas O, Koprowicz KM. Adaptive statistical iterative reconstruction versus filtered back projection in the same patient: 64 channel liver CT image quality and patient radiation dose. Eur Radiol. 2012;22(1):138–43.
Singh S, Kalra MK, Hsieh J, Licato PE, Do S, Pien HH, Blake MA. Abdominal CT: comparison of adaptive statistical iterative and filtered back projection reconstruction techniques. Radiology. 2010;257(2):373–83.
Silva AC, Lawder HJ, Hara A, Kujak J, Pavlicek W. Innovations in CT dose reduction strategy: application of the adaptive statistical iterative reconstruction algorithm. AJR Am J Roentgenol. 2010;194(1):191–9.
Khawaja RD, Singh S, Otrakji A, Padole A, Lim R, Nimkin K, Westra S, Kalra MK, Gee MS. Dose reduction in pediatric abdominal CT: use of iterative reconstruction techniques across different CT platforms. Pediatr Radiol. 2015;45(7):1046–55.
Willemink MJ, Noël PB. The evolution of image reconstruction for CT-from filtered back projection to artificial intelligence. Eur Radiol. 2019;29(5):2185–95.
Kwon H, Cho J, Oh J, Kim D, Cho J, Kim S, Lee S, Lee J. The adaptive statistical iterative reconstruction-V technique for radiation dose reduction in abdominal CT: comparison with the adaptive statistical iterative reconstruction technique. Br J Radiol. 2015;88(1054):20150463.
Pickhardt PJ, Lubner MG, Kim DH, Tang J, Ruma JA, del Rio AM, Chen GH. Abdominal CT with model-based iterative reconstruction (MBIR): initial results of a prospective trial comparing ultralow-dose with standard-dose imaging. AJR Am J Roentgenol. 2012;199(6):1266–74.
Ehman EC, Yu L, Manduca A, Hara AK, Shiung MM, Jondal D, Lake DS, Paden RG, Blezek DJ, Bruesewitz MR, et al. Methods for clinical evaluation of noise reduction techniques in abdominopelvic CT. Radiographics. 2014;34(4):849–62.
Jensen CT, Wagner-Bartak NA, Vu LN, Liu X, Raval B, Martinez D, Wei W, Cheng Y, Samei E, Gupta S. Detection of colorectal hepatic metastases is superior at standard radiation dose CT versus reduced dose CT. Radiology. 2019;290(2):400–9.
Akagi M, Nakamura Y, Higaki T, Narita K, Honda Y, Zhou J, Yu Z, Akino N, Awai K (2019) Deep learning reconstruction improves image quality of abdominal ultra-high-resolution CT. Eur Radiol.
Jiang Hsieh EL, Brian Nett, Jie Tang, Jean-Baptiste Thibault, Sonia Sahney (2019) A new era of image reconstruction: TrueFidelity™-Technical white paper on deep learning image reconstruction. https://wwwgehealthcare.com/-/jssmedia/040dd213fa89463287155151fdb01922pdf.
Jensen CT, Liu X, Tamm EP, Chandler AG, Sun J, Morani AC, Javadi S, Wagner-Bartak NA. Image quality assessment of abdominal CT by use of new deep learning image reconstruction: initial experience. AJR American J Roentgenol. 2020;21:1–8.
Tatsugami F, Higaki T, Nakamura Y, Yu Z, Zhou J, Lu Y, Fujioka C, Kitagawa T, Kihara Y, Iida M, et al. Deep learning-based image restoration algorithm for coronary CT angiography. Eur Radiol. 2019;29(10):5322–9.
Christner JA, Braun NN, Jacobsen MC, Carter RE, Kofler JM, McCollough CH. Size-specific dose estimates for adult patients at CT of the torso. Radiology. 2012;265(3):841–7.
Brady SL, Kaufman RA. Investigation of American Association of Physicists in Medicine Report 204 size-specific dose estimates for pediatric CT implementation. Radiology. 2012;265(3):832–40.
The measurement, reporting, and management of radiation dose in CT: report of AAPM Task Group 23 of the Diagnostic Imaging Council CT Committee. AAPM report no. 96. College Park (MD): American Association of Physicists in Medicine.
J-RIME JNfRaIoME: National diagnostic reference levels in Japan (2020)—Japan DRLs 2020. http://www.radherjp/J-RIME/report/DRL2020_Engverpdf 2020.
Racine D, Becce F, Viry A, Monnin P, Thomsen B, Verdun FR, Rotzinger DC. Task-based characterization of a deep learning image reconstruction and comparison with filtered back-projection and a partial model-based iterative reconstruction in abdominal CT: A phantom study. Phys Med. 2020;76:28–37.
Solomon J, Lyu P, Marin D, Samei E: Noise and spatial resolution properties of a commercially available deep learning-based CT reconstruction algorithm. Med Phys 2020.
Greffier J, Hamard A, Pereira F, Barrau C, Pasquier H, Beregi JP, Frandon J. Image quality and dose reduction opportunity of deep learning image reconstruction algorithm for CT: a phantom study. Eur Radiol. 2020;30(7):3951–9.
Benz DC, Benetos G, Rampidis G, von Felten E, Bakula A, Sustar A, Kudura K, Messerli M, Fuchs TA, Gebhard C et al. (2020) Validation of deep-learning image reconstruction for coronary computed tomography angiography: Impact on noise, image quality and diagnostic accuracy. J Cardiovasc Comput Tomogr.
Kim I, Kang H, Yoon HJ, Chung BM, Shin NY (2020) Deep learning-based image reconstruction for brain CT: improved image quality compared with adaptive statistical iterative reconstruction-Veo (ASIR-V). Neuroradiology.
Park C, Choo KS, Jung Y, Jeong HS, Hwang JY, Yun MS (2020) CT iterative vs deep learning reconstruction: comparison of noise and sharpness. Eur Radiol.
Pooler BD, Lubner MG, Kim DH, Chen OT, Li K, Chen GH, Pickhardt PJ. Prospective evaluation of reduced dose computed tomography for the detection of low-contrast liver lesions: direct comparison with concurrent standard dose imaging. Eur Radiol. 2017;27(5):2055–66.
Mileto A, Guimaraes LS, McCollough CH, Fletcher JG, Yu L. State of the art in abdominal CT: the limits of iterative reconstruction algorithms. Radiology. 2019;293(3):491–503.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was approved by the local institutional review board at Mie University Hospital (approval No. H2019-207).
Informed consent
Written informed consent was waived since this study used existing clinical CT image data. The opportunity to opt-out of the inclusion to this study was given through a notice in the hospital website. No patient showed intention for an exclusion from this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Ichikawa, Y., Kanii, Y., Yamazaki, A. et al. Deep learning image reconstruction for improvement of image quality of abdominal computed tomography: comparison with hybrid iterative reconstruction. Jpn J Radiol 39, 598–604 (2021). https://doi.org/10.1007/s11604-021-01089-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11604-021-01089-6