Abstract
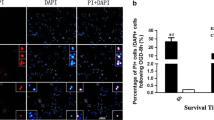
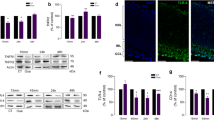
The purpose of this study was to investigate the effect of inhibition of calpain on retinal ganglion cell-5 (RGC-5) necroptosis following oxygen glucose deprivation (OGD). RGC-5 cells were cultured in Dulbecco’s-modified essential medium and necroptosis was induced by 8-h OGD. PI staining and flow cytometry were performed to detect RGC-5 necrosis. The calpain expression was detected by Western blotting and immunofluorescence staining. The calpain activity was tested by activity detection kit. Flow cytometry was used to detect the effect of calpain on RGC-5 necroptosis following OGD with or without N-acetyl-leucyl-leucyl-norleucinal (ALLN) pre-treatment. Western blot was used to detect the protein level of truncated apoptosis inducing factor (tAIF) in RGC-5 cells following OGD. The results showed that there was an up-regulation of the calpain expression and activity following OGD. Upon adding ALLN, the calpain activity was inhibited and tAIF was reduced following OGD along with the decreased number of RGC-5 necroptosis. In conclusion, calpain was involved in OGD-induced RGC-5 necroptosis with the increased expression of its downstream molecule tAIF.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Yuan L, Wang Z, Liu L, et al. Inhibiting histone deacetylase 6 partly protects cultured rat cortical neurons from oxygen-glucose deprivation-induced necroptosis. Mol Med Rep, 2015,12(2):2661–2667
Koo MJ, Rooney KT, Choi ME, et al. Impaired oxidative phosphorylation regulates necroptosis in human lung epithelial cells. Biochem Biophys Res Commun, 2015,464(3):875–880
Liu T, Zhao DX, Cui H, et al. Therapeutic hypothermia attenuates tissue damage and cytokine expression after traumatic brain injury by inhibiting necroptosis in the rat. Sci Rep, 2016,6:24547
Liu S, Wang X, Li Y, et al. Necroptosis mediates TNF-induced toxicity of hippocampal neurons. Biomed Res Int, 2014,2014:290182
Re DB, Le Verche V, Yu C, et al. Necroptosis drives motor neuron death in models of both sporadic and familial ALS. Neuron, 2014,81(5):1001–1008
Dvoriantchikova G, Degterev A, Ivanov D. Retinal ganglion cell (RGC) programmed necrosis contributes to ischemia-reperfusion-induced retinal damage. Exp Eye Res, 2014,123:1–7
Chen WW, Yu H, Fan HB, et al. RIP1 mediates the protection of geldanamycin on neuronal injury induced by oxygen-glucose deprivation combined with zVAD in primary cortical neurons. J Neurochem, 2012,120(1): c70–77
Wu HY, Tomizawa K, Matsui H. Calpain-calcineurin signaling in the pathogenesis of calcium-dependent disorder. Acta Med Okayama, 2007,61(3):123–137
Wang C, Shi D, Song X, et al. Calpain inhibitor attenuates ER stress-induced apoptosis in injured spinal cord after bone mesenchymal stem cells transplantation. Neurochem Int, 2016,97:15–25
Zhang X, Hu Y. Inhibitory effects of grape seed proanthocyanidin extract on selenite-induced cataract formation and possible mechanism. J Huazhong Univ Sci Technolog Med Sci, 2012,32(4):613–619
Autheman D, Wyder M, Popoff M, et al. Clostridium perfringens beta-toxin induces necrostatin-inhibitable, calpain-dependent necrosis in primary porcine endothelial cells. PLoS One, 2013,8(5):e64 644
Cabon L, Galan-Malo P, Bouharrour A, et al. BID regulates AIF-mediated caspase independent necroptosis by promoting BAX activation. Cell Death Differ, 2012,19(2):245–256
Samanta K, Kar P, Chakraborti T, et al. An overview of endoplasmic reticulum calpain system. Springer New York, 2013,7:3–19
Del Olmo-Aguado S, Núñez-Álvarez C, Osborne NN. Blue light action on mitochondria leads to cell death by necroptosis. Neurochem Res, 2016,41(9):2324–2335
Delavallee L, Cabon L, Galan-Malo P, et al. AIF-mediated caspase-independent necroptosis: a new chance for targeted therapeutics. IUBMB Life, 2011,63(4):221–232
Cao G, Xing J, Xiao X, et al. Critical role of calpain I in mitochondrial release of apoptosis-inducing factor in ischemic neuronal injury. J Neurosci, 2007,27(35):9278–9293
Shang L, Huang J F, Ding W, et al. Calpain: a molecule to induce AIF-mediated necroptosis in RGC-5 following elevated hydrostatic pressure. BMC Neurosci, 2014,15:63
Li Y, Chen Y, Sun M, et al. Inhibition on apoptosis induced by elevated hydrostatic pressure in retinal ganglion cell-5 via laminin upregulating ß1-integrin/focal adhesion kinase/protein kinase B signaling pathway. Chin Med J, 2016,129(8):976–983
Qing GP, Zhang S, Wang B, et al. Functional MRI signal changes in primary visual cortex corresponding to the central normal visual field of patients with primary open-angle glaucoma. Invest Ophthalmol Vis Sci, 2010,51(9):4627–4634
Huang JF, Yu HM, Shang L, et al. The correlation between rat retinal nerve fiber layer thickness around optic disc by using optical coherence tomography and histological measurements. Int J Ophthalmol, 2013,6(4):415–421
Cui YH, Huang JF, Cheng S, et al. Study on establishment and mechanics application of finite element model of bovine eye. BMC Ophthalmol, 2015,15(1): 101
Sappington RM, Chan M, Calkins DJ. Interleukin-6 protects retinal ganglion cells from pressure-induced death. Invest Ophthalmol Vis Sci, 2006,47(7):2932–2942
Li GY, Li T, Fan B, et al. The D1 dopamine receptor agonist, SKF83959, attenuates hydrogen peroxide-induced injury in RGC-5 cells involving the extracellular signal-regulated kinase/p38 pathways. Mol Vision, 2012,18:2882–2895
Chen RF, Zhang T, Sun YY, et al. Oxygen-glucose deprivation regulates BACE1 expression through induction of autophagy in Neuro-2a/APP695 cells. Neural Regen Res, 2015,10(9):1433–1440
Du S, Mao G, Zhu T, et al. TIMP1 in conditioned media of human adipose stromal cells protects neurons against oxygenglucose deprivation injury. Neurosci Lett, 2015,584:56–59
Li N, Shang L, Wang S, et al. The toxic effect of ALLN on primary rat retinal neurons. Neurotox Res, 2016, [Epub ahead of print]
Debiasi R L, Squier M K, Pike B, et al. Reovirus-induced apoptosis is preceded by increased cellular calpain activity and is blocked by calpain inhibitors. J Virology, 1999,73(1):695–701
Jiang SH, Shang L, Xue LX, et al. The effect and underlying mechanism of Timosaponin B-II on RGC-5 necroptosis induced by hydrogen peroxide. BMC Complement Altern Med, 2014,14:459
Kaji T, Boland B, et al. Calpain mediates calcium-induced activation of the erk1, 2 MAPK pathway and cytoskeletal phosphorylation in neurons: relevance to Alzheimer’s disease. Am J Pathol, 2004,165(3):795–805
Yamashima T. Reconsider Alzheimer’s disease by the ‘calpain-cathepsin hypothesis’—A perspective review. Prog Neurobiol, 2013,105:1–23
Fifre A, Sponne I, Koziel V, et al. Microtubule-associated protein MAP1A, MAP1B, and MAP2 proteolysis during soluble amyloid beta-peptide-induced neuronal apoptosis. J Biol Chem, 2006,281(1):229–240
Marcilhac A, Raynaud F, Clerc I, et al. Detection and localization of calpain 3-like protease in a neuronal cell line: possible regulation of apoptotic cell death through degradation of nuclear Ikappa Balpha. Int J Biochem Cell Biol, 2006,38(12):2128–2140
Zhang DW, Shao J, Lin J, et al. RIP3, an energy metabolism regulator that switches TNF-induced cell death from apoptosis to necrosis. Science, 2009,325(5938):332–336
Dong Y, Bao C, Yu J, et al. Receptor-interacting protein kinase 3-mediated programmed cell necrosis in rats subjected to focal cerebral ischemia-reperfusion injury. Mol Med Rep, 2016,14(1):728–736
Wang L, Wang T, Li H, et al. Receptor interacting protein 3-mediated necroptosis promotes lipopolysaccharide-induced inflammation and acute respiratory distress syndrome in mice. PLoS One, 2016,11(5):e0155723
Huang JF, Shang L, Zhang MQ, et al. Differential neuronal expression of receptor interacting protein 3 in rat retina: involvement in ischemic stress response. BMC Neurosci, 2013,14:16
Xiong K, Liao HD, Long LL, et al. Necroptosis contributes to methamphetamine-induced cytotoxicity in rat cortical neurons. Toxicol In Vitro, 2016,35:163–168
Wu JR, Wang J, Zhou SK, et al. Necrostatin-1 protection of dopaminergic neurons. Neural Regen Res, 2015,10(7):1120–1124
Li J, McQuade T, Siemer AB, et al. The RIP1/RIP3 necrosome forms a functional amyloid signaling complex required for programmed necrosis. Cell, 2012,150(2):339–350
Ding W, Shang L, Huang JF, et al. Receptor interacting protein 3-induced RGC-5 cell necroptosis following oxygen glucose deprivation. BMC Neurosci, 2015,16:49
Sosna J, Voigt S, Mathieu S, et al. The proteases HtrA2/Omi and UCH-L1 regulate TNF-induced necroptosis. Cell Commun Signal, 2013,11:76
Zhao XM, Chen Z, Zhao JB, et al. Hsp90 modulates the stability of MLKL and is required for TNF-induced necroptosis. Cell Death Dis, 2016,7:e2089
Jacobsen AV, Lowes KN, Tanzer MC, et al. HSP90 activity is required for MLKL oligomerisation and membrane translocation and the induction of necroptotic cell death. Cell Death Dis, 2016,7:e2051
Linkermann A, De Zen F, Weinberg J, et al. Programmed necrosis in acute kidney injury. Nephrol Dial Transplant, 2012,27(9):3412–3419
Sosna J, Voigt S, Mathieu S, et al. TNF-induced necroptosis and PARP-1-mediated necrosis represent distinct routes to programmed necrotic cell death. Cell Mol Life Sci, 2013,71(2):331–348
Averna M, Stifanese R, De TR, et al. Regulation of calpain activity in rat brain with altered Ca2+ homeostasis. J Biol Chem, 2007,282(4):2656–2665
Shioda N, Moriguchi S, Shrasaki Y, et al. Generation of constitutively active calcineurin by Calpain contributes to delayed neuronal death following mouse brain ischemia. J Neurochem, 2006,98(1):310–320
Das A, Wallace GC, Holmes C, et al. Hippocampal tissue of patients with refractory temporal lobe epilepsy is associated with astrocyte activation, inflammation, and altered expression of channels and receptors. Neuroscience, 2012,220:237–246
Matsumoto M, Baba Y. Role of STIM-dependent Ca2+ influx in regulatory B cells. Yakugaku Zasshi, 2013,133 (4):419–425
McKernan DP, Guerin MB, O’ Brien CJ, et al. A key role for calpains in retinal ganglion cell death. Invest Ophthalmol Vis Sci, 2007,48(12):5420–5430
Bollino D, Balan I, Aurelian L. Valproic acid induces neuronal cell death through a novel calpain-dependent necroptosis pathway. J Neurochem, 2015,133(2):174–186
Author information
Authors and Affiliations
Corresponding author
Additional information
This project was supported by grants from National Natural Science Foundation of China (No. 81371011, and No. 81571939), Wu Jie-Ping Medical Foundation of the Minister of Health of China (No. 320.6750.14118) and Natural Science Foundation of Hunan Province (No. 2015JJ2187), Teacher Research Foundation of Central South University (No. 2014JSJJ026), Science Foundation for the Youth Scholars of Central South University (No. 120973) and Project of Innovation- driven Plan of Central South University (No. 2015CXS022).
Rights and permissions
About this article
Cite this article
Chen, S., Yan, J., Deng, Hx. et al. Inhibition of calpain on oxygen glucose deprivation-induced RGC-5 necroptosis. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 36, 639–645 (2016). https://doi.org/10.1007/s11596-016-1639-y
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-016-1639-y