Summary
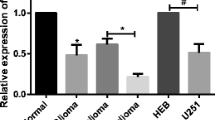
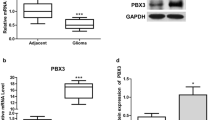
The effects of microRNA-34a (miR-34a)-regulated Notch1 gene on the proliferation and apoptosis of the human glioma cell line U87 were investigated in this study. The U87 cells were divided into miR-34a mimics, negative control, mock transfection and blank control groups in terms of different treatments. In miR-34a mimics group, human U87 glioma cells were transfected with miR-34a mimics by using lipofectamine 2000. The cells transfected with nonsense microRNA were set up as negative control group. Those treated with lipofectamine 2000 only were designated to the mock tranfection group. In the blank control group, the cells were cultured routinely and no treatment was given. The expression of miR-34a and Notch1 was detected by using real-time RT-PCR. Western blotting was employed to monitor the change in Notch1 protein. Cell proliferation and apoptosis were measured by CCK-8 and flow cytometry. The results showed that the proliferative ability of U87 cells was significantly reduced and the apoptotic cells increased in miR-34a mimics group relative to control groups. The expression of miR-34a was significantly up-regulated in mimics group as compared with control groups (P<0.05). Furthermore, Notch1 protein levels were significantly decreased in miR-34a mimics group when compared with control groups (P<0.05), but the mRNA expression of Notch1 showed no significant difference among these groups. It was concluded that miR-34a may suppress the proliferation and induce apoptosis of U87 cells by decreasing the expression of target gene Notch1, suggesting that miR-34a may become a promising gene therapeutic target for brain glioma.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Allen JC. Childhood brain tumors: current status of clinical trials in newly diagnosed and recurrent disease. Pediatr Clin North Am, 1985,32(3):633–651
Van Meir EG, Hadjipanayis CG, Norden AD, et al. Exciting new advances in neuro-oncology: the avenue to a cure for malignant glioma. CA Cancer J Clin, 2010,60(3):166–193
Yu J, Poulton J, Huang YC, et al. The hippo pathway promotes Notch signaling in regulation of cell differentiation, proliferation, and oocyte polarity. PLoS One, 2008,3(3):e1761
Jiang L, Wu J, Chen Q, et al. Notch1 expression is upregulated in glioma and is associated with tumor progression. J Clin Neurosci, 2011,18(3):387–390
Wang Z, Ahmad A, Li Y, et al. Targeting Notch to Eradicate Pancreatic Cancer Stem Cells for Cancer Therapy. Anticancer Res, 2011,31(4):1105–1113
Miele L. Notch signaling. Clin Cancer Res, 2006,12(4): 1074–1079
Inui M, Martello G, Piccolo S. MicroRNA control of signal transduction. Nat Rev Mol Cell Biol, 2010,11(4): 252–263
Chen CZ, Li L, Lodish HF, et al. MicroRNAs modulate hematopoietic lineage differentiation. Science, 2004,303(5654):83–86
Welch C, Chen Y, Stallings RL. MicroRNA-34a functions as a potential tumor suppressor by inducing apoptosis in neuroblastoma cells. Oncogene, 2007,26(34):5017–5022
Dutta KK, Zhong Y, Liu YT, et al. Association of microRNA-34a overexpression with proliferation is cell type-dependent. Cancer Sci, 2007,98(12):1845–1852
Li WB, Ma MW, Dong LJ, et al. MicroRNA-34a targets notch1 and inhibits cell proliferation in glioblastoma multiforme. Cancer Biol Ther, 2011,12(6):477–483
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods, 2001,25(4):402–408
Chiocca EA, Lawler SE. The many functions of microRNAs in glioblastoma. World Neurosurg, 2010,73(6): 598–601
Wong JW. MicroRNA-induced silencing of glioma progression. J Neurosci, 2010,30(11):3868–3869
Hébert SS, De Strooper B. Alterations of the microRNA network cause neurodegenerative disease. Trends Neurosci, 2009,32(4):199–206
Denli AM, Tops BB, Plasterk RH, et al. Processing of primary microRNAs by the Microprocessor complex. Nature, 2004,432(7014):231–235
Sassen S, Miska EA, Caldas C. MicroRNA: implications for cancer. Virchows Arch, 2008,452(1):1–10
Zhang B, Pan X, Cobb GP, et al. microRNAs as oncogenes and tumor suppressors. Dev Biol, 2007,302(1):1–12
Pang RT, Leung CO, Ye TM, et al. MicroRNA-34a suppresses invasion through downregulation of Notch1 and Jagged1 in cervical carcinoma and choriocarcinoma cells. Carcinogenesis, 2010,31(6):1037–1044
Ji Q, Hao X, Meng Y, et al. Restoration of tumor suppressor miR-34 inhibits human p53-mutant gastric cancer tumorspheres. BMC Cancer, 2008,8:266
Carthew RW. Gene regulation by microRNAs. Curr Opin Genet Dev, 2006,16(2): 203–208
Berezikov E, Guryev V, van de Belt J, et al. Phylogenetic shadowing and computational identification of human microRNA genes. Cell, 2005,120(1):21–24
Betel D, Wilson M, Gabow A, et al. The microRNA.org resource: targets and expression. Nucleic Acids Res, 2008,36:D149–D153
Li Y, Guessous F, Zhang Y, et al. MicroRNA-34a inhibits glioblastoma growth by targeting multiple oncogenes. Cancer Res, 2009,69(19):7569–7576
Milner LA, Bigas A. Notch as a mediator of cell fate determination in hematopoiesis: evidence and speculation. Blood, 1999,93(8):2431–2448
Zhang XP, Zheng G, Zou L, et al. Notch activation promotes cell proliferation and the formation of neural stem cell-like colonies in human glioma cells. Mol Cell Biochem, 2008,307(1–2):101–108
Katoh M, Katoh M. Integrative genomic analyses on HES/HEY family: Notch-independent HES1, HES3 transcription in undifferentiated ES cells, and Notch-dependent HES1, HES5, HEY1, HEY2, HEYL transcription in fetal tissues, adult tissues, or cancer. Int J Oncol, 2007,31(2):461–466
Zweidler-McKay PA. Notch signaling in pediatric malignancies. Curr Oncol Rep, 2008,10(6):459–468
Wang Q, Li H, Liu N, et al. Correlative analyses of notch signaling with resveratrol-induced differentiation and apoptosis of human medulloblastoma cells. Neurosci Lett, 2008,438(2):168–173
Fan X, Mikolaenko I, Elhassan I, et al. Notch1 and notch2 have opposite effects on embryonal brain tumor growth. Cancer Res, 2004,64(21):7787–7793
Fèvre-Montange M, Champier J, Durand A, et al. Microarray gene expression profiling in meningiomas: differential expression according to grade or histopathological subtype. Int J Oncol, 2009,35(6):1395–1407
Monk CE, Hutvagner G, Arthur JS. Regulation of miRNA transcription in macrophages in response to Candida albicans. PLoS One, 2010,5(10):e13669
Ji Q, Hao X, Zhang M, et al. MicroRNA miR-34 inhibits human pancreatic cancer tumor-initiating cells. PLoS One, 2009,4(8):e6816
Author information
Authors and Affiliations
Corresponding author
Additional information
This project was supported by grants from the Key Clinic Programs of Ministry of Health (2010–2012) and the 11th Five-year Plan of National Science and Technology Supporting Project (No. 2006BAI05A07).
Rights and permissions
About this article
Cite this article
Yu, X., Zhang, W., Ning, Q. et al. MicroRNA-34a inhibits human brain glioma cell growth by down-regulation of notch1. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 32, 370–374 (2012). https://doi.org/10.1007/s11596-012-0064-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-012-0064-0