Abstract
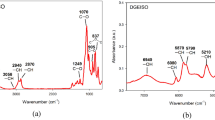
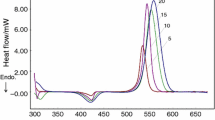
A multifunctional amine, 1,4-bis(2,4-diaminophenoxy)benzene (14BDAPOB), was prepared and used as a novel hardener for novolac epoxy resin (ER). The structure of 14BDAPOB was characterized with Fourier transform infrared (FT-IR) spectroscopy and differential scanning calorimetry (DSC). The curing kinetics of the novolac epoxy resin/1,4-bis(2,4-diaminophenoxy)benzene (ER/14BDAPOB) system was studied by means of non-isothermal DSC experiments at five heating rates and determined by the Kissinger, Ozawa and Crane methods. The results showed that the activation energy Ea of the ER/14BDAPOB (74.56 kJ/mol) system was higher than that of the epoxy resin/LCA-30 (ER/LCA-30, 68.85 kJ/mol), where LCA-30 is a commercial modified diamine. The reaction order, frequency factor and the reaction rate constant at peak temperature of the two systems were calculated. The initial decomposition temperatures (Tonset) were 398.8 °C (ER/14BDAPOB) and 334.3 °C(ER/LCA-30). The tensile shear strengths were 21.63 MPa (ER/14BDAPOB) and 21.28 MPa (ER/LCA-30). The results showed that the two cured systems exhibited good thermal and mechanical properties.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
May CA. Epoxy Resins Chemistry and Technology[M]. second ed. Yew York: Marcel Dekker, Inc, 1988
Musto P, Abbate M, Ragosta G, et al. A Study by Raman, Near-infrared and Dynamic-mechanical Spectroscopies on the Curing Behaviour, Molecular Structure and Viscoelastic Properties of Epoxy/Anhydride Networks[J]. Polymer, 2007, 48: 3703–3716
Wan J, Bu ZY, Xu CJ, et al. Model-fitting and Model-Free Nonisothermal Curing Kinetics of Epoxy Resin with a Low-volatile Five-armed Starlike Aliphatic Polyamine[J]. Thermochim. Acta, 2011, 525: 31–39
Wan J, Li BG, Fan H, et al. Nonisothermal Reaction Kinetics of DGEBA with Four-armed Atarlike Polyamine with Benzene Core (MXBDP) as Novel Curing Agent[J]. Thermochim. Acta, 2010, 510: 46–52
Wan J T, Li C, Bu ZY, et al. A Comparative Study of Epoxy Resin Cured with a Linear Diamine and a Branched Polyamine[J]. Chem. Eng. J., 2012, 188: 160–172
Montserrat S, Martin JG. Non-isothermal Curing of Diepoxide-Cycloaliphalic Diamine System by Temperature Modulated Differential Scanning Calorimetry[J]. Thermochim. Acta, 2002, 388: 343–354
Cai HY, Li P, Sui G, et al. Curing Kinetics Study of Epoxy Resin/Flexible Amine Toughness Systems by Dynamic and Isothermal DSC[J]. Thermochim. Acta, 2008, 473: 101–105
Wang HM, Zhang YC, Zhu LR, et al. Curing Behaviors and Kinetics of Epoxy Resins with a Series of Biphenyl Curing Agents Having Different Methylene Units[J]. Thermochim. Acta, 2011, 521: 18–25
Wan JT, Bu ZY, Xu CJ, et al. Preparation, Curing Kinetics and Properties of a Novel Low-volatile Starlike Aliphatic-Polyamine Curing Agent for Epoxy Resins[J]. Chem. Eng. J., 2011, 171: 357–367
Li C, Fan H, Hu JJ, et al. Novel Silicone Aliphatic Amine Curing Agent for Epoxy Resin: 1,3-Bis(2-aminoethylaminomethyl) tetramethyldisiloxane. 2. Isothermal Cure, and Dynamic Mechanical Property [J]. Thermochim. Acta, 2012, 549: 132–139
Maity T, Samanta B, Banthia A, et al. Synthesis, Characterisation, Curing Reaction of Aniline Acetaldehyde Condensate with Epoxy Resin[J]. Resin Technol., 2011, 40: 285–291
Ghaemy M, Barghamadi M, Behmadi H. Nonisothermal Cure Kinetics of DGEBA with Novel Aromatic Diamine[J]. J. Appl. Polym. Sci., 2007, 103: 3076–3083
Hu JH, Shan J Y, Zhao J, et al. Water Resistance and Curing Kinetics of Epoxy Resins with a Novel Curing Agent of Biphenyl-containing Amine Synthesized by One-pot Method[J]. Thermochim. Acta, 2015, 606: 58–65
Tang XP, Huang CH. Curing Kinetics of Epoxy Resin by Amine-containing Agents[J]. Guangzhou Huagong, 2013, 41(8): 86–87, 204
Liu XD, Cheng J, Lin X, et al. Curing Kinetics of Epoxy Resins/Amine System and Epoxy/Episulfide Resin/Amine System[J]. Huagong Xuebao (Chinese Edition), 2013, 64(11): 4046–4053
Mustafa MF, Cook WD, Schiller TL, et al. Curing Behavior Andthermal Properties of TGDDM Copolymerized with a New Pyridine-containing Diamine and with DDM or DDS [J]. Thermochim. Acta, 2014, 575: 21–28
Bhuvana S, Sarojadevi MJ. Synthesis and Characterization of Epoxy/Amine Terminated Amide-Imide-Imide Blends[J]. J. Appl. Polym. Sci., 2008, 108: 2001–2009
Supriya N, Catherine KB. DSC-TG Studies on Kinetics of Curing and Thermal Decomposition of Epoxy-Ether Amine Systems[J]. J. Therm. Anal. Calorim., 2013, 112: 201–208
Wan JT, Li BG, Fan H, et al. Nonisothermal Reaction, Thermal Stability and Dynamic Mechanical Properties of Epoxy System with Novel Nonlinear Multifunctional Polyamine Hardener[J]. Thermochim. Acta, 2010, 511: 51–58
Luda MP, Balabanovich AI, Zanetti M, et al. Thermal Decomposition of Fire Retardant Brominated Epoxy Resins Cured with Different Nitrogen Containing Hardeners[J]. Polym. Degrad. Stab., 2007, 92: 1088–1100
Woo EM, Seferis JC, Schaffnit RS. Viscoelastic Characterization of High Performance Epoxy Matrix Composites[J]. Polym. Compos., 1991, 12: 273–280
Tripathi G, Srivastava D. Cure Kinetics of Ternary Blends of Epoxy Resins Studiedby Nonisothermal DSC Data[J]. J. Appl. Polym. Sci., 2009, 112: 3119–3126
Jagadeesh KS, Gururaja RJ. Effect of Type of Substitution in 4,4’-Bis-(diaminodiphenyl) Methane Hardener on Cure Kinetics, Mechanical, and Flame Retardant Properties of Tetrafunctional Epoxy Resins[J]. J. Appl. Polym. Sci., 2006, 101: 480–491
Wan JT, Bu ZY, Xu CJ, et al. Model-fitting and Model-free Nonisothermal Curing Kinetics of Epoxy Resin with a Low-volatile Fivearmed Starlike Aliphatic Polyamine[J]. Thermochim. Acta, 2011, 525: 31–39
Van AG, Van HA, Rahier H, et al. Modulated Differential Scanning Calorimetry: Isothermal Cure and Vitrification of Thermosetting Systems[J]. Thermochim. Acta, 1996, 286: 209–224
Girard-Reydet E, Riccardi C, Sautereau CH, et al. Epoxy-Aromatic Diamine Kinetics. 1. Modeling and Influenceof the Diamine Structure[J]. Macromolecules, 1995, 28: 7599–7607
Ghaffaria M, Ehsania M, Khonakdara HA, et al. Evaluation of Curing Kinetic Parameters of an Epoxy/Polyaminoamide/Nano-Glassflake System by Non-isothermal Differential Scanning Calorimetry[J]. Thermochim. Acta, 2012, 533: 10–15
Zvetkov VL. Comparative DSC Kinetics of the Reaction of DGEBA with Aromatic Diamines. I. Non-isothermal Kinetic Study of the Reaction of DGEBA with m-Phenylene Diamine[J]. Polymer, 2001, 42: 6687–6697
Kissinger HE. Reaction Kinetics in Differential Thermal Analysis[J]. Anal. Chem., 1957, 29: 1702–1706
Ozawa T. Kinetic Analysis of Derivative Curves in Thermal Analysis[J]. Bull. Chem. Soc. Jpn., 1965, 38: 1881–1886
Crane LW, Dynes PJ, Kaelble DH. Analysis of Curing Kinetics in Polymer Composites[J]. J. Polym. Sci. Polym. Lett. Ed., 1973, 11: 533–540
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yu, X., Chen, S. & Xu, Y. Curing Kinetics and Properties of Epoxy Resin with 1, 4-bis (2, 4-diaminophenoxy)benzene. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 33, 1256–1262 (2018). https://doi.org/10.1007/s11595-018-1960-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-018-1960-3