Abstract
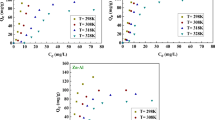
Adsorption of 2, 4, 6-trichlorophenol (TCP) onto the calcined Mg/Al-CO3 layered double hydroxide (CLDH) was investigated. The prepared Mg/Al-CO3 layered double hydroxide (LDH) and CLDH were characterized by powder X-ray diffraction (XRD) and thermo gravimetric analyzer-differential scanning calorimeters (TG-DSC). Moreover, 2,4,6-trichlorophenol (TCP) was removed effectively (94.7% of removal percentage in 9 h) under the optimized experimental conditions. The adsorption kinetics data fitted the pseudo-second-order model well. The Freundlich, Langmuir, and Tempkin adsorption models were applied to the experimental equilibrium adsorption data at different temperatures of solution. The adsorption data fitted the Freundlieh adsorption isotherm with good values of the correlation coefficient. A mechanism of the adsorption process is proposed according to the intraparticle diffusion model, which indicates that the overall rate of adsorption can be described as three steps.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Jiang M, Niu SQ, Zhan HY, et al. Research Advances in Biodegradation of Chlorophenols in Environment[J]. Chinese Journal of Applied Ecology, 2003, 14(6): 1003–1006
Nikolaou AD, Kostopoulou MN, Lekkas TD. Organic By-roducts of Drinking Water Chlorination[J]. Global NEST: The International Journal, 1999, 1(3): 143–156
Fingler S, Dravenkar V. Chlorophenols in the Sava River before, in and after the Zagreb City Area. Impact on the Purity of the City Ground and Drinking Waters[J]. Toxicol. Environ. Chem., 1988, 17: 319–328
Chen GC, Shan XQ, Wang YS, et al. Adsorption of 2,4,6-Trichlorophenol by Multi-walled Carbon Nanotubes as Affected by Cu(II) [J]. Water Res., 2009, 43: 2409–2418
Tan IA, Ahmad AL, Hameed BH. Adsorption Isotherms, Kinetics, Thermodynamics and Desorption Studies of 2,4,6-Trichlorophenol on Oil Palm Empty Fruit Bunch-based Activated Carbon[J]. J. Hazard. Mater., 2009, 164: 473–482
Eker S, Kargi F. Biological Treatment of 2,4,6-Trichlorophenol (TCP) Containing Wastewater in a Hybrid Bioreactor System with Effluent Recycle[J]. J. Environ. Manage., 2009, 90 (2), 692–698
Konsyantinos CC, Eleni S, Maria L, et al. Mechanism of Catalytic Degradation of 2,4,6-Trichlorophenol by a Fe-porphyrin Catalyst[J]. Appl. Catal. B-Environ., 2011, 101: 417–424
Gaya UI, Abdullah AH, Hussein M Z, et al. Photocatalytic Removal of 2,4,6-Trichlorophenol from Water Exploiting Commercial ZnO Powder[J]. Desalination, 2010, 263: 176–182
Bulut Y, Gozubenli N, Aydin H. Equilibrium and Kinetics Studies for Adsorption of Direct Blue 71 from Aqueous Solution by Wheat Shells[J]. J. Hazard. Mater., 2007, 144: 300–306
Wang XP, Shan XQ, Luo L, et al. Sorption of 2,4,6-Trichlorophenol in Model Humic Acid-clay Systems[J]. J. Agric. Food. Chem., 2005, 53: 3548–3555
Goh KH, Lim TT, Dong ZL. Application of Layered Double Hydroxides for Removal of Oxyanions: A Review[J]. Water Res., 2008, 42: 1343–1368
Turk T, Aip I, De veci H. Adsorption of As(V) from Water Using Mg-Fe-based Hydrotalcite (FeHT)[J]. J. Hazard. Mater., 2009, 171: 665–670
Angeles M, Gabriela JA, Getsemani MM, et al. Photoassisted Degradation of 4-Chlorophenol and p-Cresol Using MgAl Hydrotalcites[J]. Ind. Eng. Chem.Res., 2011, 50: 2762–2767
Iyi N, Sasaki T. Deintercalation of Carbonate Ions and Anion Exchange of an Al-rich MgAl-LDH (Layered Double Hydroxide)[J]. Appl.Clay. Sci., 2008, 42: 246–251
Kavitha D, Namasivayam C. Experimental and Kinetic Studies on Methylene Blue Adsorption by Coir Pith Carbon[J]. Bioresour. Technol., 2007, 98: 14–21
Tseng RL, Wu FC. Inferring the Favorable Adsorption Level and the Concurrent Multi-stage Process with the Freundlich Constant[J]. J. Hazard. Mater., 2008, 155(1-2): 277–287
Karam, Yuzer H, Sabah E, et al. Adsorption of Cobalt from Aqueous Solutions onto Sepiolite[J]. Water Res., 2003, 37 (1): 224–232
Li KQ, Zheng Z, Huang XF, et al. Equilibrium, Kinetic and Thermodynamic Studies on the Adsorption of 2-Nitroaniline onto Activated Carbon Prepared from Cotton Stalk Fibre[J]. J. Hazard. Mater., 2009, 166: 213–220
Zaghouane-Boudiafa H, Boutahala M, Tiar C, et al. Treatment of 2,4,5-Trichlorophenol by MgAl-SDBS organo-layered Double Hydroxides: Kinetic and Equilibrium Studies[J]. Chem. Eng. J., 2011, 173(1): 36–414
Ho YS, Mckay GA. Comparison of Chemisorption Kinetic Models Applied to Pollutant Removal on Various Sorbents[J]. Process Saf. Environ. Prot., 1998, 76: 332–340
Ozcan A, Oncu EM, Ozcan AS. Kinetics, Isotherm and Thermodynamic of Adsorptiom of Acid Blue 193 from Aqueous Solutions onto Natural Sepiolite[J]. Colloids Surf. A, 2006, 277: 90–97
Ho YS, Ng JCY, Mckay G. Kinetics of Polutant Sorption by Biosorbents: Review[J]. Sep. Purif. Methods, 2000, 29: 189–232
Guibal E, Mccarrick P, Tobin JM. Comparison of the Adsorption of Anionic Dyes on Activated Carbon and Chitosan Derivatives from Dilute Solutions[J]. Sep. Sci. Technol., 2003, 38: 3049–3073
Mckay G, Otterburn MS, Sweeney AG. The Removal of Colour from Effluent Using Various Adsorbents-III Silica: Rate Processes[J]. Sweeney. Water Res., 1980, 14 (152): 11–20
Liu W, Li LQ, Yao XL, et al. Role of Pore Structure of Activated Carbon in Adsorption for Acetone[J]. Journal of Central South University: Science and Technology, 2012, 43 (4): 1574–1583 (in Chinese)
Lazaridis NK, Asouhidou DD. Kinetics of Sorptive Removal of Chromium(VI) from Aqueous Solutions by Calcined Mg-Al-CO3 Hydrotalcite[J]. Water Res., 2003, 37: 2875–2882
Author information
Authors and Affiliations
Corresponding author
Additional information
Funded by the National Natural Science Foundation of China (No.21476269)
Rights and permissions
About this article
Cite this article
Zhang, D., Zhao, G., Yu, J. et al. Thermodynamic and kinetic studies of effective adsorption of 2,4,6-trichlorophenol onto calcine Mg/Al-CO3 layered double hydroxide. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 31, 1211–1218 (2016). https://doi.org/10.1007/s11595-016-1514-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-016-1514-5