Abstract
To promote the rehabilitation of motor function in children with cerebral palsy (CP), we developed motor imagery (MI) based training system to assist their motor rehabilitation. Eighteen CP children, ten in short- and eight in long-term rehabilitation, participated in our study. In short-term rehabilitation, every 2 days, the MI datasets were collected; whereas the duration of two adjacency MI experiments was ten days in the long-term protocol. Meanwhile, within two adjacency experiments, CP children were requested to daily rehabilitate the motor function based on our system for 30 min. In both strategies, the promoted motor information processing was observed. In terms of the relative signal power spectra, a main effect of time was revealed, as the promoted power spectra were found for the last time of MI recording, compared to that of the first one, which first validated the effectiveness of our intervention. Moreover, as for network efficiency related to the motor information processing, compared to the first MI, the increased network properties were found for the last MI, especially in long-term rehabilitation in which CP children experienced a more obvious efficiency promotion. These findings did validate that our MI-based rehabilitation system has the potential for CP children to assist their motor rehabilitation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Cerebral palsy (CP) induces motor dysfunction, behavioral abnormality, and intellectual delay in patients (Aisen et al. 2011). As reported, CP is the most common cause of childhood-onset and lifelong physical disability and affects about 1 in 500 neonates (Graham et al. 2016). Currently, most of the used therapies depend on the physical execution of designed motor movements. Specifically, similar to motor execution (ME) (Chong and Stinear 2017), motor imagery (MI) relies on the mental simulation of actions and involves the motor representations in the brain in a similar way (i.e., neural structures and processes) with the actual physical movements (Hétu et al. 2013; Munzert et al. 2009). MI helps to extend the ability of normal people (Li et al. 2010), to provide compensative assistance to the disabled (Li et al. 2016), and to benefit the rehabilitation of motor function (Sharma et al. 2006).
The MI-based brain-computer interface (MI-BCI) has been considered as a promising way that benefits motor rehabilitation (Daly and Wolpaw 2008), and the MI-based therapy helps to promote the capacity of motor planning (Craje et al. 2010; Steenbergen et al. 2009) which is also found to avail the motor rehabilitation. Compared to control subjects, 6-week training with actual practice (i.e., ME) and mental imagery (i.e., MI) of trampolining improved the skill of actual movements of the training subjects, as assessed by five nationally qualified judges (Isaac 1992). Moreover, previous MI-based studies have reported corresponding changes in both central and peripheral systems. In this regard, a previous study manifested that MI training improved motor performance, which was attributed to the existence of a top-down mechanism based on the activation of a central representation of the movements (Papadelis et al. 2007). Meanwhile, individuals with lower visual MI abilities involved cognitive effort with greater cerebral activation, and those with poor kinaesthetic MI ability needed a continuous update of information to support MI-related executive functions (Menicucci et al. 2020).
The child patients have a higher possibility of brain neuroplasticity, when compared to the adults, the effective therapy may, therefore, achieve a satisfying rehabilitation quality in child patients (Page et al. 2001; Yan et al. 2013). During physical rehabilitation, patients perform motor training with assistance from clinicians, which cannot guarantee them to participate in this progress voluntarily, especially CP children. By contrast, MI emphasizes the active and voluntary participation of patients themselves. A vivid presentation of instruction (also feedback) has the potential to attract their attention, which then encourages them to voluntarily participate in motor rehabilitation. Herein, in this study, we developed an MI-based rehabilitation system to assist CP children with their motor rehabilitation.
Recently, remarkable evidence underlying the mechanism of CP has been uncovered (Bakhtiari et al. 2017; Papadelis et al. 2014). The disturbance of motor function occurs in distributed networks, such as the sensorimotor network (Bakhtiari et al. 2017). By using the network biomarkers, the individual motor capacity can be quantitatively evaluated (Hanakawa et al. 2003; Zhang et al. 2015), as well as the rehabilitation efficacy after motor rehabilitation. Actually, previous studies have manifested the involvement of somatosensory and motor cortex in the functional deficits observed in CP children (Papadelis et al. 2019; Wittenberg 2009). For example, abnormal cortical somatosensory evoked potentials were detected in CP children, which indicated that the disturbance of the central perception related to the sensory stimulus was due to the damage in the sensory cortex or subcortical areas of the central nervous system (Teflioudi et al. 2011). Additionally, the bilateral maladaptive functional changes in the ipsilesional primary somatosensory cortex of CP children, which were expressed as suppressed gamma responses and alterations in the cortical body representation, were also found to be correlated with the severity of axonal damage in the ascending sensory tracts projecting to the more affected hemisphere (Papadelis et al. 2018).
Overall, CP has raised extensive concern for its high pathogenic rate, whereas effective therapy improving the motor neuroplasticity of CP children is still lacking. Contrary to traditional physical rehabilitation, MI-BCI emphasizes patients’ voluntary participation in the rehabilitation training, which might be more helpful to promote motor function. To validate the effectiveness of our MI-based rehabilitation system, hence, in the current study, we firstly designed two rehabilitation strategies (i.e., short- and long-term rehabilitation). Particularly, every two days in short- and ten days in long-term progress, we collected related MI datasets; between two adjacency MI recording, CP children were requested to perform motor training based on our system. Then, we further combined signal power spectra, brain network, and graph theory analyses to investigate the capacity of our system that benefits the motor rehabilitation in CP children, and to which extend, their motor function could be promoted.
Materials and methods
Participants
The study was approved by the Ethics Committee of Sichuan Rehabilitation Hospital. A total of 10 CP children (5 girls, age range of 8–12 years) were recruited to take part in short-term rehabilitation, and 8 CP children (1 girl, age range of 8–12 years) participated in the long-term strategy. All of these children were diagnosed with a well-defined type of CP as determined by a clinical examination by an experienced child neurologist at the Sichuan Rehabilitation Hospital. Patients suffered from acquired lower limb deformity, but can stand and sit independently, which enabled them to perform MI tasks feasibly. No severe behavioral disorder, neurodegenerative disease, and other concomitant neurologic disorders could interfere with the rehabilitation tasks. Before the experiment, the therapists, CP children, and their guardians were told about the research purpose, potential benefits, and rehabilitation procedures. And thus, they could understand the purpose and process of our experiments, as well as cooperating with the therapists, to accomplish designed rehabilitation protocols. Moreover, the written informed consent was also given to the CP children and their guardians, they then signed their names on it.
Rehabilitation system
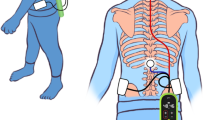
As described in the framework (Fig. 1a), CP children were instructed to perform the motor rehabilitation based on our MI system; meanwhile, based on the real-time feedback, they would dynamically adjust their MI strategies to achieve a better BCI control. In this study, to encourage them to voluntarily participate in the rehabilitation, we thereby developed different feedback modules that could provide a vivid scenario of MI task (Fig. 1b), which was further depicted as follows.
Rehabilitation protocol
Given the validation of the MI-based rehabilitation system needed a relatively long duration, following previous studies (Chowdhury et al. 2018; Foong et al. 2019) which adopted pre-post single-group designs to evaluate the effects of BCI therapy on the improvement of motor function, in this study, a strategy of self-control was applied by investigating any potential difference between their first and last time of MI recordings to measure the effect of our MI training. Moreover, to further testify the capacity of the MI-based motor rehabilitation for CP children, two different strategies were also designed, i.e., the short- and long-term rehabilitation protocols as displayed in Fig. 2, and the differences between both strategies were the duration of two adjacency electroencephalogram (EEG) MI recording sessions and the total rehabilitation period, as well.
In detail, for the short-term strategy (Exp. 1), the duration of two adjacency sessions was set as 2 days and the total period was 12 days; while for the long-term strategy (Exp. 2), both parameters were set as 10 and 60 days, respectively. Moreover, between two adjacency recording sessions, CP children were requested to perform the MI-based motor rehabilitation each day and to encourage them to voluntarily participate in the rehabilitation progress, the vivid feedback modules were developed, as displayed in Fig. 1b. Taking the floor blossom as an example, during their daily MI-based motor training, CP children would try to control the floor blossom. As predefined, the blossom was the effective indicator of successful MI-based training. When the floor bloomed, CP children would continue their imaged motor behaviors, if not they would be asked to dynamically adjust their MI strategies to achieve better control of the floor blossom. Then, on the last day of each rehabilitation session, the corresponding EEG datasets of MI tasks were collected and analyzed to validate the capacity of the daily MI-based motor training during this session.
Meanwhile, during daily training, four MI runs were carried out, and between two adjacency runs, a 10-min break was set. In each run, CP patients were requested to mentally imagine the left- or right-limb movement according to the instruction presented on the computer screen, and 40 trials, i.e., 20 trials of left-limb MI (lMIs) and 20 trials of right-limb MI (rMIs) were included. In each trial, a 3-s preparation was first set; thereafter, CP children were given 6 s to perform the required MI task (e.g., raising the left or right limb), and during the 6-s-length MI, the appearance of instruction on the computer screen implied the onset of the lMIs or rMIs. Finally, a 3-s rest was given to children after the MI task. As illustrated in Fig. 2c, during each time of MI-based motor rehabilitation, the first two runs were taken as offline training sets, and the remaining runs were used as online testing sets. Based on the training datasets, the corresponding classifier was accordingly trained, which would be further applied in two testing sets to achieve MI-BCI control.
EEG data recording
Sparse electrodes were selected around the primary sensorimotor cortex, premotor and supplementary motor cortices, as well as the parietal cortex, which were reported to have neuroplastic potential and were extensively activated in CP children during MI tasks (Ehrsson et al. 2003; Neuper et al. 2005; Shin et al. 2012; Stippich et al. 2002). Additionally, the visual cortex area in the occipital lobe that is responsible for visual-motor control during MI was also selected (Kerr et al. 2011; Solodkin et al. 2004). Therefore, sparse 16 Ag/AgCl electrodes (BrainMaster, Inc., Shenzhen, China) locating at the posterior area (i.e., Cp1, Cp2, C3, C4, Po3, Po4, P3, P4, O1, O2, Cp3, Cp4, T3, T4, T5, and T6) were used to record related MI EEG data by the Symtop amplifier (Symtop Instrument, Beijing, China), and the scalp distribution of these electrodes was displayed in Fig. 3. The default parameters of the amplifier are that the online band-pass filtering is set within a narrow band of 0.5–45 Hz, the online sampling rate is set as 1000 Hz, and the electrode AFz is set as the reference. Meanwhile, during the whole experiment, the impedance for each electrode was kept below 5 kΩ.
EEG data analysis
To acquire artifact-free trials, task MI-EEG was first preprocessed with procedures including the averaging referencing, 0.5–30 Hz offline bandpass filtering, 6-s data segmentation, and artifact trial removal. As depicted, the first two MI runs were taken as offline training sets, and the remaining runs were used as online testing sets. And when recording related MI EEG datasets, only the first two runs were collected, including 40 lMIs and 40 rMIs. Therefore, in this study, relying on the onsets of lMIs or rMIs, we extracted a 6-s length of MI segments, resulting in 40 trials for lMIs or rMIs. Unfortunately, few trials were left for the last time of the MI experiment in short-term rehabilitation, in this study, we thereby analyzed the first five times of MI tasks.
In the current work, we mainly focused on the brain’s sensorimotor activity since MI corresponded to the situation when a person imaging movement. Hence, brain activities of 8–15 Hz were considered in the following analysis since numerous studies have implied a functional relation between cerebral processes such as movement or primary sensory processing and 8–15 Hz oscillations (Başar-Eroğlu et al. 2013; Ishii et al. 2009; Olson et al. 2006). In particular, EEG recordings over the sensorimotor cortex show a distinctive oscillatory pattern (Cheyne et al. 2003; Gunji et al. 2007; Howe and Sterman 1972) and is associated with inhibition of motor activity (Sterman 1977) in a frequency range of 8–15 Hz, which plays an essential role for biofeedback therapy (Egner and Gruzelier 2001) and appears to be dominant during imagination of body movements (Neuper et al. 2006; Pfurtscheller et al. 2006).
Cortical oscillations have been validated to play an important role in a wide range of neural activities. In particular, task-related changes in spectral power within and between neuronal assemblies in specific frequency bands have been reported (Niemarkt et al. 2011). As the relative power spectral density (rPSD) emphasizes relative power magnitude alterations over a range of frequencies (Cohen 2014), the rPSD within 8–15 Hz was evaluated for each measurement of the short- and long-term rehabilitation, respectively. Specifically, using Welch's method, the absolute power spectral density (aPSD) of each frequency bin f was first computed, the rPSD was then defined and calculated as the ratio of the aPSD in 8–15 Hz to that of the entire band. Finally, the whole brain rPSD was formulated as,
where ξ denotes the types of short- or long-term rehabilitation.
The coherence is then used to measure the couplings between each pair of electrodes. Considering two time series, x(t) and y(t), in each MI trial, a high coherence value denotes a great synchronization of neuronal oscillations between two electrodes, while a low coherence implies functional segregation. The coherence is formulated as,
where Pxy(f) is the cross-spectral density between x(t) and y(t) at frequency f, Pxx(f) and Pyy(f) are the auto-spectral densities of x(t) and y(t) at frequency f, respectively, and Cxy(f) is the frequency-dependent coherence.
In this study, the sixteen electrodes were set as network nodes, and the network edge was estimated by averaging the Cxy within the concerned frequency range, which resulted in a 16 × 16 weighted adjacency matrix for each MI trial. For each CP child, the final EEG network was obtained by averaging the adjacency matrices across all artifact-free trials.
Brain connectivity toolbox (BCT, http://www.nitrc.org/projects/bct/) (Rubinov and Sporns 2010) was finally used to calculate the weighted network properties, such as the clustering coefficient (CC) and characteristic path length (CPL). Thereafter, we analyzed the fluctuations of the network properties for both rehabilitation strategies.
Results
Figure 4 displays the varied rPSD in short- and long-term rehabilitation protocols, and an obvious increased rPSD from first to last lMI/rMI could be found. Meanwhile, one-way repeated measures analysis of variance (ANOVA) identified that, CP children experienced the promoted rPSD during both rehabilitation protocols, as reveled by a main effect of timing. In particular, in short-term rehabilitation, the significantly or marginally enhanced rPSD of the last rMIs (F(4, 36) = 7.04, p = 0.00) and lMIs (F(4, 36) = 2.44, p = 0.06) were found, when compared to that of the first MI; in the meantime, for long-term strategy, the rPSD of both lMIs (F(5, 35) = 31.15, p = 0.00) and rMIs (F(5, 35) = 24.02, p = 0.00) corresponding to the last MI recording were also found to be significantly promoted. Besides, post hoc tests further uncovered that the last MI recording (lMIs: 0.83 ± 0.03, rMIs: 0.84 ± 0.02) presented a significant lager rPSD compared to that of the first one (lMIs: 0.80 ± 0.01, rMIs: 0.80 ± 0.03) for short-term rehabilitation (lMIs: p = 0.01, rMIs: p = 0.00). As for the long-term rehabilitation, compared to the first MI (lMIs: 0.66 ± 0.04, rMIs: 0.70 ± 0.03), the last MI (lMIs: 0.84 ± 0.04, rMIs: 0.83 ± 0.06) also exhibited the significant higher rPSD (lMIs: p = 0.00, rMIs: p = 0.00).
Figure 5 displays the varied weighted network properties of the short- and long-term rehabilitation strategies, in which the increased CC and decreased CPL from first to last MI are illustrated for both strategies. Besides, to investigate the effect of training duration on rehabilitation efficacy in CP children, we then analyzed the dynamic fluctuations of related brain networks. However, in terms of the ANOVA results, we found no significant main effect of time in network properties for both rehabilitation processes due to the relatively small sample size. The Wilcoxon signed-rank test (McCrum-Gardner 2008; Rosner et al. 2006) was then utilized to statistically quantify the potential differences in related network properties between the first and last MI-based rehabilitation session.
Particularly, in short-term rehabilitation (Fig. 6a), the CC of lMIs (p = 0.03) and rMIs (p = 0.02) were identified to be significantly larger for the last MI task, while the CPL of lMIs (p = 0.04) and rMIs (p = 0.06) of the last MI were found to be significantly and marginally significantly shorter than that of the first MI. Meanwhile, similar to short-term rehabilitation, Fig. 6b further demonstrates that the CC (lMIs: p = 0.03; rMIs: p = 0.01) of the last MI is significantly larger than that of the first MI, whereas the CPL (lMIs: p = 0.03; rMIs: p = 0.01) of last MI is significantly shorter than that of first MI.
The network properties for the first and last time of MI in short- (a) and long-term rehabilitation (b). The blue and red filled bars denote the network properties corresponding to the first and last time of the MI recording, respectively. The black solid lines and asterisks denote the statistical difference of network property (p < 0.05) between the two conditions. Values are the means and standard deviations (Mean ± STD) of network properties
Eventually, the promoted relative change ratio (RCR) from the first to the last MI recording was calculated for both strategies, which was defined as a ratio of the rPSD or property difference between last and first MI versus that of the first MI. Table 1 then illustrates a similar tendency between both strategies. Specifically, a larger RCR was found for long-term rehabilitation, when compared to the short-term training.
Discussion
The MI is regarded as a potential strategy during motor rehabilitation (Stevens and Stoykov 2003) and has the potential to evaluate the rehabilitation efficacy in patients (Zimmermann-Schlatter et al. 2008). In the current study, we developed an MI-based rehabilitation system to "tell" CP children how to voluntarily participate in motor rehabilitation. Thereafter, two strategies, i.e., the short- and long-term rehabilitation, were probed to validate the potential application of our system.
MI requires participants to voluntarily mentally imagine motor manners. Compared to traditional MI that used words or arrows as instructions, a vivid presentation seems to be more attractive in CP children. In our present study, we thereby presented the instruction (also feedback) in different ways that were illustrated in Fig. 1 when developing this system. Concretely, by presenting MI instructions in such a way, we intended to make it easier for the CP children, which then tutored them to implement the same motion according to the instruction; and presenting the feedback in such different ways aimed to remind CP children to dynamically adjust their imagery after they noticed the feedback of their last imagery.
The rPSD which represents the relative power distribution of EEG series in the frequency domain (Wang et al. 2015) was used to evaluate the dynamic fluctuations of the brain activity related to MI. As for both lMIs and rMIs after the short- and long-term MI rehabilitation, a significantly promoted brain power related to the motor information processing in CP children was verified in Fig. 4, in which a main effect of time was found to further support the effectiveness of the intervention. As reported previously, the power spectrum changes of CP children during power mobility training found the alpha power increased during intervention phases, suggesting that the mobility training could induce enhancement of cognitive processing (Kenyon et al. 2018). Moreover, a distinctive oscillatory pattern over the sensorimotor cortex within 8–15 Hz (Cheyne et al. 2003; Gunji et al. 2007; Howe and Sterman 1972) plays an essential role for biofeedback therapy (Egner and Gruzelier 2001) and appears to be dominant during imagination of body movements (Neuper et al. 2006; Pfurtscheller et al. 2006). And the rPSD of 8–15 Hz has been regarded as a better EEG biomarker (Elgendi et al. 2011) in classification performance than the traditional frequency bands. This is consistent with our findings as the rPSD across 8–15 Hz did stably capture the promoted power in CP children, which verified the enhanced cognitive functions during the rehabilitation training and further manifested the potential role of the MI system for CP children’s motor rehabilitation.
Considering the topological features shared by structural and functional networks in the brain, the human brain is organized in a way with a highly efficient small-world manner (Bullmore and Sporns 2009; Iturria-Medina et al. 2008; Stam and Van Straaten 2012). A large CC ensures the information to be processed by the brain efficiently, a short CPL supports a high level of global communication efficiency across distributed areas (Li et al. 2015; Rubinov and Sporns 2010). Our present study demonstrated a promoted brain efficiency related to the motor information processing in CP children (Fig. 5), after the short- and long-term MI rehabilitation. For both lMIs and rMIs, after MI training, a significantly larger CC of MI network was found in Exp. 1, along with a shorter CPL, compared to that before MI training (Fig. 6a). Besides, the long-term rehabilitation aimed to investigate the effect of MI training duration on rehabilitation efficacy in another CP group. As displayed in Fig. 5, the promoted network efficiency related to motor information processing was also found in long-term rehabilitation. Particularly, the obvious difference between the two strategies was that from Table 1, a larger RCR of efficiency (i.e., larger rates of increased CC and shorter CPL) could be found for the long-term condition, and the significant promotion (p < 0.05) of last MI that deviated from first MI were further demonstrated in Fig. 6b.
One possible limitation was that although a pre-post single-group design was used to validate the capacity of our MI-based rehabilitation system, a matched control group is still necessary. For example, a controlled-group design regarding the use of traditional physical rehabilitation in combination with the application of our MI-BCI should be considered to manifest that MI training is more effective than using other devices. Hence, in the future, a control group will be recruited and investigated. Another would be the relatively small patient size that reduced the statistical power, this was apparent when quantitatively investigating the promotion of motor networks for both strategies, therefore, more patients will be recruited in our future study to further validate these findings.
Conclusion
This study demonstrated the potential application of our MI-based system in the motor rehabilitation for CP children, by investigating the EEG power and brain networks related to MI recording sessions. As demonstrated, motor rehabilitation based on our system significantly promoted brain efficiency when processing motor-related information, which was accomplished by promoting the efficient reallocation of brain resources, as well as enhancing the inter-regional activity, during motor imagery.
References
Aisen ML, Kerkovich D, Mast J, Mulroy S, Wren TAL, Kay RM, Rethlefsen SA (2011) Cerebral palsy: clinical care and neurological rehabilitation. Lancet Neurol 10:844–852. https://doi.org/10.1016/s1474-4422(11)70176-4
Bakhtiari R, Cummine J, Reed A, Fox CM, Chouinard B, Cribben I, Boliek CA (2017) Changes in brain activity following intensive voice treatment in children with cerebral palsy. Hum Brain Mapp. https://doi.org/10.1002/hbm.23669
Başar-Eroğlu C, Schmiedt-Fehr C, Mathes B (2013) Auditory-evoked alpha oscillations imply reduced anterior and increased posterior amplitudes in schizophrenia. Supplements to Clinical neurophysiology, vol 62. Elsevier, Amsterdam, pp 121–129
Bullmore E, Sporns O (2009) Complex brain networks: graph theoretical analysis of structural and functional systems. Nat Rev Neurosci 10:186–198. https://doi.org/10.1038/nrn2575
Cheyne D, Gaetz W, Garnero L, Lachaux J-P, Ducorps A, Schwartz D, Varela FJ (2003) Neuromagnetic imaging of cortical oscillations accompanying tactile stimulation. Cogn Brain Res 17:599–611
Chong BW, Stinear CM (2017) Modulation of motor cortex inhibition during motor imagery. J Neurophysiol 117:1776–1784. https://doi.org/10.1152/jn.00549.2016
Chowdhury A et al (2018) Active physical practice followed by mental practice using BCI-driven hand exoskeleton: a pilot trial for clinical effectiveness and usability. IEEE J Biomed Health Informatics 22:1786–1795
Cohen MX (2014) Analyzing neural time series data: theory and practice. MIT Press, Cambridge
Craje C, van Elk M, Beeren M, van Schie HT, Bekkering H, Steenbergen B (2010) Compromised motor planning and motor Imagery in right. Hemiparetic Cerebral Palsy Res Dev Disabilities 31:1313–1322. https://doi.org/10.1016/j.ridd.2010.07.010
Daly JJ, Wolpaw JR (2008) Brain-computer interfaces in neurological rehabilitation. Lancet Neurol 7:1032–1043
Egner T, Gruzelier JH (2001) Learned self-regulation of EEG frequency components affects attention and event-related brain potentials in humans. NeuroReport 12:4155–4159
Ehrsson HH, Geyer S, Naito E (2003) Imagery of voluntary movement of fingers, toes, and tongue activates corresponding body-part-specific motor representations. J Neurophysiol
Elgendi M, Vialatte F, Cichocki A, Latchoumane C, Jeong J, Dauwels J (2011) Optimization of EEG frequency bands for improved diagnosis of Alzheimer disease. 2011 Annual International Conference of the IEEE Engineering in Medicine and Biology Society. IEEE, New York, pp 6087–6091
Foong R et al (2019) Assessment of the efficacy of EEG-based MI-BCI with visual feedback and EEG correlates of mental fatigue for upper-limb stroke rehabilitation. IEEE Trans Biomed Eng 67:786–795
Graham H et al. (2016) Cerebral palsy. Nat Rev Disease Primers 2
Gunji A, Ishii R, Chau W, Kakigi R, Pantev C (2007) Rhythmic brain activities related to singing in humans. Neuroimage 34:426–434
Hanakawa T, Immisch I, Toma K, Dimyan MA, Van Gelderen P, Hallett M (2003) Functional properties of brain areas associated with motor execution and imagery. J Neurophysiol 89:989–1002
Hétu S, Grégoire M, Saimpont A, Coll M-P, Eugène F, Michon P-E, Jackson PL (2013) The neural network of motor imagery: an ALE meta-analysis. Neurosci Biobehav Rev 37:930–949
Howe RC, Sterman M (1972) Cortical-subcortical EEG correlates of suppressed motor behavior during sleep and waking in the cat. Electroencephalogr Clin Neurophysiol 32:681–695
Isaac AR (1992) Mental practice-does it work in the field? Sport Psychol 6:192–198
Ishii R et al (2009) Cortical oscillatory power changes during auditory oddball task revealed by spatially filtered magnetoencephalography. Clin Neurophysiol 120:497–504
Iturria-Medina Y, Sotero RC, Canales-Rodríguez EJ, Alemán-Gómez Y, Melie-García L (2008) Studying the human brain anatomical network via diffusion-weighted. MRI Graph Theory NeuroImage 40:1064–1076. https://doi.org/10.1016/j.neuroimage.2007.10.060
Kenyon LK, Farris JP, Aldrich NJ, Rhodes S (2018) Does power mobility training impact a child’s mastery motivation and spectrum of EEG activity? An exploratory project. Disabil Rehabil Assist Technol 13:665–673
Kerr AL, Cheng S-Y, Jones TA (2011) Experience-dependent neural plasticity in the adult damaged brain. J Commun Disorders 44:538–548
Li F et al (2015) Relationships between the resting-state network and the P3: evidence from a scalp EEG study Sci Rep 5:15129. https://doi.org/10.1038/Srep15129
Li Y, Long J, Yu T, Yu Z, Wang C, Zhang H, Guan C (2010) An EEG-based BCI system for 2-D cursor control by combining Mu/Beta rhythm and P300 potential. IEEE Trans Bio-med Eng 57:2495–2505. https://doi.org/10.1109/Tbme.2010.2055564
Li Y, Pan J, Long J, Yu T, Wang F, Yu Z, Wu W (2016) Multimodal BCIs: target detection, multidimensional control, and awareness evaluation in patients with disorder of consciousness. P IEEE 104:332–352. https://doi.org/10.1109/Jproc.2015.2469106
McCrum-Gardner E (2008) Which is the correct statistical test to use? Br J Oral Maxillofacial Surg 46:38–41
Menicucci D, Di Gruttola F, Cesari V, Gemignani A, Manzoni D, Sebastiani L (2020) Task-independent electrophysiological correlates of motor imagery ability from kinaesthetic and visual perspectives. Neuroscience 443:176–187
Munzert J, Lorey B, Zentgraf K (2009) Cognitive motor processes: the role of motor imagery in the study of motor representations. Brain Res Rev 60:306–326. https://doi.org/10.1016/j.brainresrev.2008.12.024
Neuper C, Scherer R, Reiner M, Pfurtscheller G (2005) Imagery of motor actions: differential effects of kinesthetic and visual–motor mode of imagery in single-trial. EEG Cogn Brain Res 25:668–677
Neuper C, Wörtz M, Pfurtscheller G (2006) ERD/ERS patterns reflecting sensorimotor activation and deactivation. Progress Brain Res 159:211–222
Niemarkt HJ et al (2011) Maturational changes in automated EEG spectral power analysis in preterm infants. Pediatric Res 70:529–534
Olson AK, Eadie BD, Ernst C, Christie BR (2006) Environmental enrichment and voluntary exercise massively increase neurogenesis in the adult hippocampus via dissociable pathways. Hippocampus 16:250–260
Page SJ, Levine P, Sisto S, Johnston MV (2001) A randomized efficacy and feasibility study of imagery in acute stroke. Clin Rehabil 15:233–240
Papadelis C, Kourtidou-Papadeli C, Bamidis P, Albani M (2007) Effects of imagery training on cognitive performance and use of physiological measures as an assessment tool of mental effort. Brain Cogn 64:74–85
Papadelis C, Ahtam B, Nazarova M, Nimec D, Snyder B, Grant PE, Okada Y (2014) Cortical somatosensory reorganization in children with spastic cerebral palsy: a multimodal neuroimaging study. Front Hum Neurosci 8:725. https://doi.org/10.3389/fnhum.2014.00725
Papadelis C et al (2018) Reorganization of the somatosensory cortex in hemiplegic cerebral palsy associated with impaired sensory tracts. NeuroImage Clin 17:198–212
Papadelis C, Kaye H, Shore B, Snyder B, Grant PE, Rotenberg A (2019) Maturation of corticospinal tracts in children with hemiplegic cerebral palsy assessed by diffusion tensor imaging and transcranial magnetic stimulation. Front Human Neurosci 13:254
Pfurtscheller G, Brunner C, Schlögl A, Da Silva FL (2006) Mu rhythm (de) synchronization and EEG single-trial classification of different motor imagery tasks. Neuroimage 31:153–159
Rosner B, Glynn RJ, Lee MLT (2006) The Wilcoxon signed rank test for paired comparisons of clustered data. Biometrics 62:185–192
Rubinov M, Sporns O (2010) Complex network measures of brain connectivity: Uses and interpretations. Neuroimage 52:1059–1069. https://doi.org/10.1016/j.neuroimage.2009.10.003
Sharma N, Pomeroy VM, Baron J-C (2006) Motor imagery a backdoor to the motor system after stroke? Stroke 37:1941–1952. https://doi.org/10.1161/01.Str.0000226902.43357.Fc
Shin YK, Lee DR, Hwang HJ, You SJH, Im CH (2012) A novel EEG-based brain mapping to determine cortical activation patterns in normal children and children with cerebral palsy during motor imagery tasks. NeuroRehabilitation 31:349–355
Solodkin A, Hlustik P, Chen EE, Small SL (2004) Fine modulation in network activation during motor execution and motor imagery. Cereb Cortex 14:1246–1255
Stam Cv, Van Straaten E (2012) The organization of physiological brain networks clinical neurophysiology: official. J Int Federation Clin Neurophysiol 123:1067–1087. https://doi.org/10.1016/j.clinph.2012.01.011
Steenbergen B, Crajé C, Nilsen DM, Gordon AM (2009) Motor imagery training in hemiplegic cerebral palsy: a potentially useful therapeutic tool for rehabilitation. Dev Med Child Neurol 51:690–696
Sterman M (1977) Sensorimotor EEG operant conditioning: experimental and clinical effects. Pavlovian J Biol Sci Official J 12:63–92
Stevens JA, Stoykov MEP (2003) Using motor imagery in the rehabilitation of Hemiparesis. Arch Phys Med Rehab 84:1090–1092
Stippich C, Ochmann H, Sartor K (2002) Somatotopic mapping of the human primary sensorimotor cortex during motor imagery and motor execution by functional magnetic resonance imaging. Neurosci Lett 331:50–54
Teflioudi EP, Zafeiriou DI, Vargiami E, Kontopoulos E, Tsikoulas I (2011) Somatosensory evoked potentials in children with bilateral spastic cerebral palsy. Pediatric Neurol 44:177–182
Wang R, Wang J, Yu H, Wei X, Yang C, Deng B (2015) Power spectral density and coherence analysis of Alzheimer’s. EEG Cogn Neurodyn 9:291–304
Wittenberg GF (2009) Motor mapping in cerebral palsy. Dev Med Child Neurol 51(Suppl 4):134–139
Yan J, Sun J, Guo X, Jin Z, Li Y, Li Z, Tong S (2013) Motor imagery cognitive network after left ischemic stroke: study of the patients during mental rotation task. PLoS ONE 8:e77325. https://doi.org/10.1371/journal.pone.0077325
Zhang R et al (2015) Efficient resting-state EEG network facilitates motor imagery performance. J Neural Eng 12:066024. https://doi.org/10.1088/1741-2560/12/6/066024
Zimmermann-Schlatter A, Schuster C, Puhan MA, Siekierka E, Steurer J (2008) Efficacy of motor imagery in post-stroke rehabilitation: a systematic review. J Neuroeng Rehabil 5:8. https://doi.org/10.1186/1743-0003-5-8
Acknowledgements
This work was supported in part by the Key Research and Development Program of Guangdong Province, China (#2018B030339001), in part by the National Natural Science Foundation of China (#81330032, #81771925, #61961160705, #U19A2082, and #61901077), in part by the National Key Research and Development Plan of China (#2017YFB1002501).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xie, J., Jiang, L., Li, Y. et al. Rehabilitation of motor function in children with cerebral palsy based on motor imagery. Cogn Neurodyn 15, 939–948 (2021). https://doi.org/10.1007/s11571-021-09672-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11571-021-09672-3