Abstract
Purpose
The aim of this study was to retrospectively assess the relationship between radiological and hemodynamic parameters in patients with chronic thromboembolic pulmonary hypertension (CTEPH). We introduced a new CT-score to evaluate hemodynamic changes, only employing CT-pulmonary angiography (CTPA).
Materials and methods
145 patients affected by CTEPH underwent hemodynamic and CTPA evaluation. Among these 145 patients, 69 underwent pulmonary endarterectomy (PEA) and performed a CTPA evaluation even after surgery. Hemodynamic assessment considered the values of mean pulmonary artery pressure (mPAP) and pulmonary vascular resistance (PVR), obtained through right heart catheterization (RHC). Radiological evaluation included CTPA signs of pulmonary hypertension.
Results
A highly significant statistical correlation was observed between the new CT-score and both mPAP and PVR (p < 0.000) in the whole sample and also in the subgroup who underwent PEA. In addition, mPAP and PVR showed an important association with the severity of mosaic perfusion (p < 0.000). mPAP also correlated with main pulmonary artery diameter (p < 0.01); a significant association was found in both between PVR and tricuspid regurgitation(p < 0.000) and with PVR and presence of unilateral or bilateral pulmonary thromboembolic occlusion (p < 0.05).
Conclusion
Our results confirm the diagnostic role of CTPA in evaluating patients with CTEPH and in addition open a new horizon in assessing hemodynamic changes in patients with CTEPH, only employing a CTPA, especially when RHC is contraindicated or not possible.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Chronic thromboembolic pulmonary hypertension (CTEPH) results from incomplete resolution of the vascular obstruction caused by pulmonary thromboemboli. If left untreated, over time, a gradual hemodynamic and symptomatic decline can be observed in these patients. As a consequence, prognosis in CTEPH is poor and proportional to the degree of pulmonary hypertension. Advanced CTEPH leads to cardiac remodeling, involving right ventricular (RV) dilatation and hypertrophy, tricuspid regurgitation (TR), and interventricular septal bowing (ISB), with consequent impact on cardiac function [1].
Pulmonary endarterectomy (PEA) represents the therapy of choice for patients with surgically accessible disease. PEA may be performed with a low mortality risk and results in clinical improvement and improved prognosis [2–4].
Radiologic assessment, most commonly with computed tomography (CT), is central in the evaluation of pulmonary hypertension (PH) and can assist in elucidating the underlying cause [5–9].
Our purpose was to retrospectively evaluate the relationship among radiological, clinical and hemodynamic parameters in patients with CTEPH and in a subgroup of them who underwent PEA. In the literature, different scores were introduced to quantify arterial obstruction in acute pulmonary embolism (modified Miller and Walsh, Qanadli and Mastora) [10–14], whereas a validated score for chronic pulmonary embolism does not exist yet.
Thus, we introduced a new CT-score to evaluate the severity of chronic pulmonary embolism; finally, we analyzed the relationships of our new CT-score with hemodynamic and clinical parameters.
Materials and methods
Study population
Between February 2006 and March 2015, 145 patients (M:F = 64:81, mean age 60) affected by CTEPH underwent hemodynamic and radiological evaluation at the Department of Cardio-Thoracic Surgery of our University Hospital.
All patients had undergone a cardiovascular examination with clinical, functional and hemodynamic assessment (New York Heart Association class determination, echocardiography, Right Heart Catheterization) and a radiological evaluation through CTPA.
CTEPH diagnosis was based on values of mean pulmonary artery pressure (mPAP) ≥25 mmHg measured through right heart catheterization (RHC) and on the presence of specific vascular and parenchymal signs on CTPA examination (in particular mPA diameter ≥2.9 cm and the presence of pulmonary artery occlusion).
Finally, cardio-surgical examination had been performed to verify eligibility criteria for PEA.
In addition, we analyzed a subgroup of 69 patients (M:F = 24:45, mean age 57) among 145 patients who underwent PEA and performed a cardiovascular and a radiological evaluation also after surgery; in particular New York Heart Association (NYHA) class determination, right heart catheterization (RHC) and CTPA were repeated postoperatively in a period of time ranging between 1 month and 6 years later.
CTPA acquisition and interpretation
Radiological evaluation has been performed with a CTPA exam, a first-line diagnostic technique with a high sensibility and specificity in the detection of imaging features of CTEPH, in the differentiation of this disease from other causes of PH and in the assessment of the technical feasibility of surgery.
After having acquired an informed consent, a first non-contrast scan was performed with a 16-detector Siemens Sensation Cardiac CT scanner; craniocaudal spiral scans have been acquired, with the patient in the supine position, during an inspiratory breath-hold without contrast enhancement. Scanning parameters were as follows: 120 kV, 210 mAs, slice thickness 0.75 mm with 0.5 mm increments, acquisition time 11 s; the images were reconstructed using high spatial frequency (bone) algorithm and visualized with window level of −500/−700 HU and window width of 1500/1800 HU.
CTPA was obtained after intravenous injection (brachial antecubital vein) of 100 mL of nonionic water-soluble contrast agent (Iomeron 400, Bracco, Milan, Italy), at a rate of 4.0 ml, followed by 40 ml of saline bolus chaser, using a double-syringe power injector (Stellant, MedRad, Pittsburg, PA, USA).
Imaging was automatically started 5 s after detection of contrast enhancement in the main pulmonary artery (mPA) (threshold, 80 HU) with bolus tracking software (CARE Bolus, Siemens Medical solutions, Forchheim, Germany).
Radiological parameters evaluated in our sample are listed in Table 1.
The main pulmonary artery (mPA) diameter was measured proximal to its bifurcation, perpendicular to the vessel wall. Right and left ventricular short axes, measured on a reformatted four-chamber view, were defined as the largest distance between the interventricular septum and the free wall of ventricle. Diameter of superior vena cava (SVC) was measured at the level of arch of azygos vein. Tricuspid regurgitation was split into four grades of gravity according to the degree of contrast medium reflux into the inferior vena cava and hepatic veins [15].
Among all parameters, special attention has been focused on pulmonary vascular signs, in particular on the evaluation of presence, location and degree of obstruction due to organized thromboemboli.
Since there does not exist a CT-scoring system to quantify the severity of CTEPH, we tried to create a new radiological CT-score that could reflect hemodynamic changes in patients with chronic pulmonary embolism, considering only radiological signs; in particular we considered: unilateral or bilateral disease (U/B), main pulmonary artery diameter (Ø mPA), mosaic perfusion pattern (MP) and tricuspid regurgitation (TR).
The new score was calculated as follows:
Scores assigned to each item are listed in Table 2.
MP was considered as mild if it was <10%, moderate if <20 and >30% and severe if >30%, using a visual score.
The total score can reach a maximum value of 15.
Radiological evaluation was made independently and with unblinded clinical information by two Radiologists of our Department, respectively, with 40 and 5 years of radiological experience. Discrepancies were resolved by consensus.
Hemodynamic evaluation
Hemodynamic study was performed with RHC, the reference technique for the assessment of PH severity. mPAP and Pulmonary Vascular Resistance (PVR) were measured through this invasive technique.
Indications and surgical technique
The selection criteria for patients undergoing PEA and operative technique have been thoroughly described by Jamieson and Kapelanski [16, 17]. In particular, surgery is considered technically feasible if at imaging the organized thrombi are not located distal to the lobar arteries or to the origin of the segmental vessels. Surgical resectability is further influenced by the severity of hemodynamic impairment, the presence of concomitant diseases, in particular underlying lung disease, and the patient’s age [18].
After median sternotomy and full heparinization, cardio-pulmonary bypass (CPB) is instituted by cannulation of the ascending aorta and both inferior and superior venae cavae. A passive drain is placed in the main pulmonary trunk. The left ventricle is drained through the right upper pulmonary vein with negative pressure. While cooling, before 18 °C nasopharyngeal temperature is reached, the aorta is clamped and cold crystalloid cardioplegia (Custodiol—Dr Koehler GMBH Chemie-Alsbach-Haehlein) is administered. Right pulmonary artery is exposed and opened. When the cleavage plane is identified proximally, the systemic circulatory arrest is achieved and endarterectomy performed distally towards the lobar, segmental and sub-segmental arteries. Every 15–20 min of arrest is followed by 15 min of systemic reperfusion. After right pulmonary artery is reconstructed, cardioplegia is reinfused. As previously described, the endarterectomy in the left side is completed. CPB is suspended when normothermia is reached.
Statistical analysis
Pearson correlation was used to analyze linear associations between continuous radiological parameters and hemodynamic data.
Student’s T test was used to assess differences between pre- and post-surgical values of continuous radiological and hemodynamic parameters.
Comparisons of categorical radiological parameters were performed by Chi-square, McNemar Test or Wilcoxon signed-rank test.
Values of p < 0.05 were considered statistically significant.
All analyses were performed with Stata/SE 12.1 Statistical Software.
Results
Patient characteristics
145 patients (M:F = 64:81, mean age 60) were selected for the study and their radiological and hemodynamic parameters were evaluated at the baseline before PEA.
In addition, was identified a subgroup of 69/145 (M:F = 24:45, mean age 57) that underwent PEA and had a radiological and hemodynamic assessment before and after PEA.
Cardiovascular risk factors and thrombophilic disorders assessed are displayed in Table 3.
In physical examination before surgery, only 2/145 patients were asymptomatic, while all others had a variety of symptoms reported in Table 3.
All 145 patients had hemodynamic evidence of PH (mPAP values >25 mmHg), which was also confirmed by radiological evaluation through the presence of typical CT signs (in particular mPA diameter ≥2.9 cm and the presence of pulmonary artery occlusion).
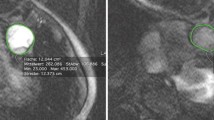
The distribution of pulmonary vascular occlusion (Figs. 1, 2) assessed on CTPA is displayed in Table 4.
Results in the whole sample
In all 145 patients, highly significant statistical correlations were observed between the new CT-score and hemodynamic parameters (mPAP, PVR) with a p < 0.000 (Table 5).
mPAP was also correlated with the main pulmonary artery diameter (p < 0.01); a significant association was found between PVR both with tricuspid regurgitation (p < 0.000) and presence of unilateral or bilateral pulmonary thromboembolic occlusion (p = 0.05); in addition, mPAP and PVR showed an important relationship with the severity of mosaic perfusion (p < 0.000) in all 145 patients.
Finally, we observed that bronchial artery hypertrophy did not show a significant correlation either with mPAP or PVR.
Radiological, hemodynamic and clinical variations
The relationship between variation of radiological, hemodynamic and clinical parameters evaluated at baseline and after surgery in the subgroup of 69 patients is displayed in Table 6.
All radiological and hemodynamic parameters evaluated showed an improvement except for collateral circles (CC), interventricular septum bowing (ISB) and BAH (Fig. 3) which were unchanged, respectively, in 92, 81 and 79% (Table 6).
In particular, MP evaluated before and after PEA showed a normalization in the 40% and a reduction of the group with severe grade of MP from 30 to 4% (Fig. 4); mPA diameter presented an improvement in the 80% (Fig. 5); TR grades were grouped as 1 as mild, 2 as moderate, 3 and 4 as severe and the severe grade declined from 30 to 4%.
As regards NYHA class variations the group that included NYHA III and IV passed from 92% of pre-surgery to 34% in post-PEA. Moreover, NYHA I and II passed from 8 to 66%.
A significant statistical difference was observed for mPAP and PVR (p < 0.01) and for radiological parameters evaluated before and after PEA (Table 6).
Therefore, statistical correlations between radiological and hemodynamic parameters (mPAP and PVR) before and after PEA are displayed in Table 7. In particular, mPAP presented a significant correlation with the MP and mPA diameter both before and after PEA; PVR correlated with MP before and after PEA, while presented a significant statistical correlation with mPA diameter only after PEA.
Finally mPAP percent difference correlated significantly with NYHA clinical classification (p = 0.002, each one evaluated in terms of improvement, stability or worsening).
The new CT-score
The statistical analysis of comparison between radiological and hemodynamic parameters is shown in Table 5. In particular, the CT-score correlated both with mPAP and PVR in the whole sample (145 patients).
We calculated a cutoff of the CT-score = 9 as the median value of all the scores considering the CT-scores and the hemodynamic parameters at baseline (145 patients); so we divided patients into two groups as follows:
-
patients with a baseline CT-score ≤9;
-
patients with a baseline CT-score >9.
We found that a baseline CT-score ≤9 corresponded to a mean value of mPAP of 46 mmHg ± 11 of standard deviation (SD) and a mean value of PVR of 7.9 mmHg ± 3.6 SD; whereas for a baseline CT-score >9 we obtained a mean value of mPAP of 50.7 mmHg ± 9.3 SD and a mean value of PVR of 10.3 mmHg ± 4 SD.
Comparison of the mean values of mPAP in the group with a baseline CT-score ≤9 and the mean values of mPAP in the group with a baseline CT-score >9 was significant (p = 0.006) and the same was for the mean values of PVR (p = 0.000) in both groups.
Another step of our study consisted in evaluating the subgroup of patients who underwent surgery (69 patients); we compared the new CT-score with mPAP and PVR before and after surgical intervention and observed highly significant correlation (p ≤ 0.005) as displayed in Table 8.
In addition, as before in all 145 patients, considering the CT-score and the hemodynamic parameters after PEA in the subgroup of 69 patients, we recognized a CT-score = 6 as the median of all the scores and divided the patients into two groups as follows:
-
patients with a CT-score after PEA ≤6;
-
patients with a CT-score after PEA >6.
We observed that a CT-score after PEA ≤ 6 corresponded to a mean value of mPAP of 24.3 mmHg ± 8.5 SD and a mean value of PVR of 2.9 mmHg ± 1.5 SD; whereas for a CT-score after PEA >6 we found out a mean value of mPAP of 31.2 mmHg ± 7.7 SD and a mean value of PVR of 4.3 mmHg ± 1.5 SD. Comparison of the mean values of mPAP in the group with a CT-score after PEA ≤6 and the mean values of mPAP in the group with a CT-score after PEA >6 was significant (p = 0.001) and the same was for the mean values of PVR (p = 0.000) in both groups.
Discussion
Pulmonary endarterectomy offers curative treatment for CTPEH patients with profound improvements in hemodynamics, symptoms, and survival [19–21].
The results of the present study confirm the importance of CTPA in the evaluation of patients with CTEPH undergoing PEA. Initially, CT scanning was employed mostly to identify occlusion in the central pulmonary arteries, which has been considered as a criterion for the operability of CTEPH. Nowadays, it has been recognized the role of CT scanning in providing more information on different components of the disease, predicting surgical success in the individual patient [22–24].
In particular, the highly significant correlation emerged for each radiological parameter, evaluated before and after PEA (p < 0.01), allows us to reconfirm the fundamental role of CTPA in assessing the outcome of PEA [3, 25].
As shown in Table 6, some radiological parameters did not show substantial modifications on CT executed after surgery; in particular, it was observed that collateral circles, BAH and leftward interventricular septum bowing did not change much after PEA, which could be linked to a minor reversibility of these parameters after surgical intervention. The study by Herre J. Reesink [1] analyzed the effect of pulmonary endarterectomy on the restoration of right ventricular remodeling in patients with chronic thromboembolic pulmonary hypertension by magnetic resonance imaging and observed that after pulmonary endarterectomy, pulmonary hemodynamic improved, as we demonstrated as well. They also assessed the normalization of leftward ventricular septal bowing; this last result is different from ours, probably due to the absence of the ECG gating in our study and/or a better evaluation of it through MRI [1].
A further step of our study consisted in evaluating the relationship between variation of radiological and hemodynamic parameters. From this analysis, it resulted that the absolute values of mPAP and PVR were strongly correlated with the degree of MP evaluated before and after PEA (Table 7). Also Marc Heinrich et al. [22] correlated CT scan findings with hemodynamic measurements in sixty patients who had undergone PEA, analyzing preoperative and postoperative hemodynamic with preoperative CT imaging features. They found out the MP as the best parameter for correlation with preoperative hemodynamic measurements, considering MP as a perfusion score demonstrating a strong correlation with mPAP and PVR.
These outcomes strengthen ours in which patients with a major reduction of mPAP after PEA also showed a consistent improvement of mosaic perfusion pattern on CT. This result can be probably explained as surgical disobstruction of pulmonary vessels causes both a reduction in mPAP values and the reperfusion of lung areas which were poorly vascularised before PEA, with the consequent disappearance of hypo-perfused lung areas on chest CT after surgical intervention. So the MP is the most sensible radiological parameter of the surgical success.
In addition the significant relationship between mPAP and mPA diameter, evaluated both as absolute value and as percent difference, confirms the relationship between mPA size and PH severity. Over the last three decades, this relationship has been extensively investigated by several studies which demonstrated a moderate to strong correlation between mPA diameter and mPAP, to the extent that a cutoff value of PA size has been proposed to help in the diagnosis or exclusion of PH [26–32]. Some authors [22, 33] also investigated this relationship in patients with CTEPH who underwent PEA, coming to the same conclusions, in particular, among other parameters, mPA diameter measured on CT showed the strongest correlation with mPAP and the most significant reversibility after surgery [33].
Another aim of our study was evaluating the relationship between mPAP percent difference and variation in NYHA class after PEA; this variation was also expressed dividing our sample into improved, unchanged and worsened patients. We observed that mPAP percent difference correlated significantly with variation of NYHA class in our sample. These data confirm the good correspondence between clinical and hemodynamic improvement after PEA and, as many authors already demonstrated [34, 35], further confirm NYHA class as a useful parameter in the evaluation of clinical severity in patients with CTEPH.
As concerns the scoring system of CTEPH, in past years, different scores (modified Miller and Walsh scores and, more recently, Qanadli and Mastora scores) have been proposed for the quantification of clot burden with CTPA in acute pulmonary embolism. However, until now, there is not still a CT index designed to quantify the severity of chronic PE. As some authors already did [26, 36], we preliminarily applied Qanadli score to our patients with chronic PE and similar to other authors’ results, we established that Qanadli score could not accurately reflect the severity of pulmonary artery obstruction in CTEPH (statistical analysis with linear regression did not show a significant correlation between post-surgical change in Qanadli score and hemodynamic parameters with p > 0.05).
We introduced a new CT-score using those radiological parameters with the best statistical correlation with hemodynamic parameters (MP, mPA diameter, TR and the presence of unilateral or bilateral pulmonary artery occlusion).
In the course of our analysis, we demonstrated that the CT-score proposed could accurately reflect hemodynamic changes in patients with CTEPH. In fact, it was observed both in whole sample and in the subgroup of patients who underwent PEA that a greater reduction of CT-score corresponded to a consistent reduction of both hemodynamic parameters considered. We correlated each radiological parameter of the CT-score with mPAP and PVR and we also tried to use central or peripheral occlusion of pulmonary arteries; our results led to a stronger correlation of both hemodynamic parameters with the presence of unilateral or bilateral occlusion in pre- and post-surgical evaluations.
From these results, it follows that the CT-score is directly proportional to the hemodynamic severity of CTEPH; therefore, it could be used as an index of hemodynamic changes in all CTEPH patients for a quick assessment of hemodynamic impairment. The difference between the median value of the CT-score in the two groups of patients is probably due, in our opinion, to the amount and the different characteristics of the two samples. In fact, the median value of all CT-score in the group of 145 patients is evaluated only at the baseline CT, while the one of the other group (69 patients) is evaluated on the pre- and post-surgical CT. These characteristics determine a lower median value of all the CT-score in the subgroup of 69.
In particular, we noticed, in the evaluation before and after PEA, that CT-score values ≤6 corresponded to a mPAP values closer to normal; then a cutoff = 6 could help in identifying hemodynamic values almost close to normal.
Finally, the CT-score proposed seems to be simple, fast to apply and useful in evaluating hemodynamic profile and consequently the severity of PH, using only a CT exam; in this way the new Index could be used to give cardiologists an estimate of hemodynamic changes in patients with CTEPH, either in those who carry out the intervention and in those who cannot, especially when a RHC is contraindicated or difficult to be performed.
RHC is an invasive procedure and the use of an alternative non-invasive imaging modality to reduce the need of RHC at diagnosis or follow-up is an attractive proposition.
The limitations of our study are due to the absence of ECG gating and to the fact that our proposed CT-score needs to be largely evaluated in terms of reproducibility.
Conclusion
The results of the present study confirm the fundamental role of CTPA in the evaluation of pulmonary vascular and parenchymal alterations in patients with CTEPH, highlight the usefulness of CTPA in the radiological evaluation of this patients and in the assessing the outcome of the surgical procedure when executed.
Finally, our results open a new “horizon” in the use of CTPA, through the use of the CT-score introduced, even if more studies are needed to assess the reproducibility of the method. We can affirm that CT-score here proposed could represent a quick and easy index of hemodynamic changes, not only in surgical but also in not surgical CTEPH patients, either associated with right heart catheterization or as a substitute for this invasive technique, when it is contraindicated or not possible.
Abbreviations
- AV:
-
Azygos vein
- BAH:
-
Bronchial arteries hypertrophy
- CC:
-
Collateral circles
- CPB:
-
Cardio-pulmonary bypass
- CS:
-
Coronary sinus
- CTEPH:
-
Chronic thromboembolic pulmonary hypertension
- CTPA:
-
Computed tomography pulmonary angiography
- ISB:
-
Interventricular septum bowing
- LV:
-
Left ventricle diameter
- MP:
-
Mosaic perfusion
- PEA:
-
Pulmonary endarterectomy
- PA:
-
Pulmonary artery
- PE:
-
Pulmonary embolism
- PH:
-
Pulmonary hypertension
- RA:
-
Right atrium
- RV:
-
Right ventricle
- RVFWT:
-
Right ventricle free wall thickness
- SD:
-
Standard deviation
- SVC:
-
Superior vena cava
- TR:
-
Tricuspid regurgitation
References
Reesink HJ, Marcus JT, Tulevski II et al (2007) Reverse right ventricular remodeling after pulmonary endarterectomy in patients with chronic thromboembolic pulmonary hypertension: utility of magnetic resonance imaging to demonstrate restoration of the right ventricle. J Thorac Cardiovasc Surg 133:58–64
Berman M, Gopalan D, Sharples L et al (2014) Right ventricular reverse remodeling after pulmonary endarterectomy: magnetic resonance imaging and clinical and right heart catheterization assessment. Pulm Circ 4:36–44
Marshall PS, Kerr KM, Auger WR (2013) Chronic thromboembolic pulmonary hypertension. Clin Chest Med 34:779–797
Taboada D, Pepke-Zaba J, Jenkins DP et al (2014) Outcome of pulmonary endarterectomy in symptomatic chronic thromboembolic disease. Eur Respir J 44:1635–1645
Wijesuriya S, Chandratreya L, Medford AR (2013) Chronic pulmonary emboli and radiologic mimics on CT pulmonary angiography a diagnostic challenge. Chest 143:1460–1471
Peňa E, Dennie C, Veinot J et al (2012) Pulmonary hypertension: how the radiologist can help. RadioGraphics 32:9–32
Ameli-Renani S, Rahman F, Nair A et al (2014) Dual-energy CT for imaging of pulmonary hypertension: challenges and opportunities. RadioGraphics 34:1769–1790
Castañer E, Gallardo X, Ballesteros E et al (2009) CT Diagnosis of chronic pulmonary thromboembolism. RadioGraphics 29:31–53
Schoepf UJ, Costello P (2004) CT angiography for diagnosis of pulmonary embolism: state of the art. Radiology 230:329–337
Miller GA, Sutton GC, Kerr IH et al (1971) Comparison of streptokinase and heparin in treatment of isolated acute massive pulmonary embolism. Br Med J 2:681–684
Walsh PN, Greenspan RH, Simon M et al (1973) An angiographic severity index for pulmonary embolism. Circulation 47–48(suppl 2):101–108
Qanadli SD, El Hajjam M, Vieillard-Baron A et al (2001) New CT index to quantify arterial obstruction in pulmonary embolism: comparison with angiographic index and echocardiography. Am J Roentgenol 176:1415–1420
Mastora I, Remy-Jardin M, Masson P et al (2003) Severity of acute pulmonary embolism: evaluation of a new spiral CT angiographic score in correlation with echocardiographic data. Eur Radiol 13:29–35
Ghaye B, Ghuysen A, Bruyere PJ et al (2006) Can CT pulmonary angiography allow assessment of severity and prognosis in patients presenting with pulmonary embolism? What the radiologist needs to know. RadioGraphics 26:23–40
Groves AM, Win T, Charman SC et al (2004) Semi-quantitative assessment of tricuspid regurgitation on contrast-enhanced multidetector CT. Clin Radiol 59:715–719
Jamieson SW, Kapelanski DP (2000) Pulmonary endarterectomy. Curr Probl Surg 37:165–252
Jenkins DP, Madani M, Mayer E et al (2013) Surgical treatment of chronic thromboembolic pulmonary hypertension. Eur Respir J 41:735–742
Fedullo PF, Auger WR, Kerr KM et al (2001) Chronic thromboembolic pulmonary hypertension. N Engl J Med 345:1465–1472
Iino M, Dymarkowski S, Chaothawee L et al (2008) Time course of reversed cardiac remodeling after pulmonary endarterectomy in patients with chronic pulmonary thromboembolism. Eur Radiol 18:792–799
Jamieson SW, Kapelanski DP, Sakakibara N et al (2003) Pulmonary endarterectomy: experience and lessons learned in 1500 cases. Ann Thorac Surg 76:1457–1462
Dartevelle P, Fadel E, Mussot S, Chapelier A, Herve P, de Perrot M, Cerrina J, Ladurie FL, Lehouerou D, Humbert M et al (2004) Chronic thromboembolic pulmonary htpertension. Eur Respir J 23:637–648
Heinrich M, Uder M, Tscholl D et al (2005) CT scan findings in chronic thromboembolic pulmonary hypertension predictors of hemodynamic improvement after pulmonary thromboendarterectomy. Chest 127:1606–1613
Kauczor HU, Schwickert HC, Mayer E et al (1994) Spiral CT of bronchial arteries in chronic thromboembolism. J Comput Assist Tomogr 18:855–861
Bergin CJ, Sirlin C, Deutsch R et al (2000) Predictors of patient response to pulmonary thromboendarterectomy. Am J Roentgenol 174:509–515
Oikonomou A, Dennie CJ, Müller NL et al (2004) Chronic thromboembolic pulmonary arterial hypertension correlation of postoperative results of thromboendarterectomy with preoperative helical contrast-enhanced computed tomography. J Thorac Imaging 19:67–73
Liu M, Ma Z, Guo X et al (2011) Computed tomographic pulmonary angiography in the assessment of severity of chronic thromboembolic pulmonary hypertension and right ventricular dysfunction. Eur J Radiol 80:e462–e469
Kuriyama K, Gamsu G, Stern RG et al (1984) CT-determined pulmonary artery diameters in predicting pulmonary hypertension. Invest Radiol 19:16–22
Haimovici JB, Trotman-Dickenson B, Halpern EF et al (1997) Relationship between pulmonary artery diameter at computed tomography and pulmonary artery pressures at right-sided heart catheterization. Massachusetts General Hospital Lung Transplantation Program. Acad Radiol 4:327–334
Edwards PD, Bull RK, Coulden R et al (1998) CT measurement of main pulmonary artery diameter. Br J Radiol 71:1018–1020
Mahammedi A, Oshmyansky A, Hassoun PM (2013) Pulmonary artery measurements in pulmonary hypertension. The role of computed tomography. J Thorac Imaging 28:96–103
Chan AL, Juarez MM, Shelton DK et al (2011) Novel computed tomographic chest metrics to detect pulmonary hypertension. BMC Med Imaging 11:7
Corson N, Armato SG, Labby ZE et al (2014) CT-based pulmonary artery measurements for the assessment of pulmonary hypertension. Acad Radiol 21:523–530
Schmidt HC, Kauczor HU, Schild HH et al (1996) Pulmonary hypertension in patients with chronic pulmonary thromboembolism: chest radiograph and CT evaluation before and after surgery. Eur Radiol 6:817–825
Sato M, Ando M, Kaneko K et al (2013) Respiratory and hemodynamic changes in patients with chronic thromboembolic pulmonary hypertension 1 year after pulmonary endarterectomy. Ann Vasc Dis 6:578–582
Corsico AG, D’Armini AM, Cerveri I et al (2008) Long-term outcome after pulmonary endarterectomy. Am J Respir Crit Care Med 178:419–424
Liu M, Ma Z, Guo X et al (2013) Cardiovascular parameters of computed tomographic pulmonary angiography to assess pulmonary vascular resistance in patients with chronic thromboembolic pulmonary hypertension. Int J Cardiol 164:295–300
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
No funds were used for the present study.
Ethical standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Leone, M.B., Giannotta, M., Palazzini, M. et al. A new CT-score as index of hemodynamic changes in patients with chronic thromboembolic pulmonary hypertension. Radiol med 122, 495–504 (2017). https://doi.org/10.1007/s11547-017-0750-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-017-0750-x