Abstract
Purpose
The aim of this study was to review some prognostic factors for survival after radiofrequency ablation (RFA) of metastases from colorectal cancer (CRC).
Materials and methods
From 1996 to 2009, 262 patients with metastases from CRC were treated with RFA. Fourteen were lost to follow-up. The following predictors were analysed in the remaining 248: synchronous/metachronous metastases, single/multiple metastases, diameter of largest metastasis and absence/presence of extrahepatic metastases. Survival was measured from the date of metastasis diagnosis and from the date of RFA.
Results
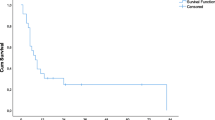
Survival at 1, 2, 3 and 5 years was 93%, 78%, 62% and 35% from metastasis diagnosis, and 84%, 59%, 43% and 23% from the date of RFA. Median survival was 41 months in patients with largest metastasis ≤3 cm and 21.7 months for those with metastases >3 cm (p=0.0001); survival increased to 45.2 months in patients with largest metastasis ≤2.5 cm and fell to 18.5 months in those with metastasis >3.5 cm. Median survival of patients with extrahepatic metastases was significantly lower than that of patients without extrahepatic disease (23.3 vs. 32.6 months, p=0.018).
Conclusions
In light of our long-term results obtained with commonly used equipment, small lesion size (diameter of largest lesion ≤3 or 2.5 cm) proved to be the most favourable prognostic factor for survival in patients with CRC metastases to the liver treated with RFA. This conclusion is probably related to the possibility of obtaining radical ablation and points to the usefulness of devices allowing ablation of larger volumes. In the presence of extrahepatic metastases, RFA has less impact on survival, even though it is potentially useful in patients at a higher risk of death due to hepatic rather than extrahepatic metastases.
Riassunto
Obiettivo
Scopo del nostro lavoro è stato rivalutare alcuni predittori di sopravvivenza nella termoablazione con radiofrequenze (RFA) delle metastasi da carcinoma colorettale (CRC).
Materiali e metodi
Tra il 1996 e il 2009 abbiamo trattato con RFA 262 pazienti con metastasi da CRC. Quattordici sono stati persi al follow-up; in 248 sono stati analizzati i seguenti predittori: metastasi sincrone/metacrone, metastasi unica/multiple, diametro della metastasi principale, assenza/presenza di metastasi extraepatiche. Le sopravvivenze sono state calcolate dalla data di diagnosi di metastasi e da quella della RFA.
Risultati
La sopravvivenza a 1, 2, 3, 5 anni è stata 93%, 78%, 62%, 35% dalla diagnosi di metastasi, e 84%, 59%, 43%, 23% dalla RFA. Nei pazienti con metastasi principale ≤3 cm la sopravvivenza mediana è stata 41 mesi vs. 21,7 di quelli con >3 cm (p=0,0001), ma in quelli ≤2,5 cm è salita a 45,2 mesi e in quelli >3,5 cm è scesa a 18,5. La sopravvivenza mediana dei pazienti con metastasi extraepatiche è stata significativamente inferiore a quelli senza (23,3 vs. 32,6 mesi, p=0,018).
Conclusioni
Alla luce dei nostri risultati a lungo termine, ottenuti con le apparecchiature comunemente utilizzate negli ultimi anni, le piccole dimensioni (diametro della lesione principale ≤3 o 2,5 cm) si confermano il fattore prognostico più favorevole per la sopravvivenza dei pazienti con metastasi epatiche da CRC sottoposti a RFA. Questa conclusione è molto probabilmente riconducibile alla possibilità di un’ablazione radicale e induce a prospettare l’utilità di apparecchiature in grado di aumentare il volume di ablazione. La RFA in presenza di metastasi extraepatiche è meno efficace in termini di sopravvivenza, ma potenzialmente utile nei pazienti a minor rischio di decesso per le metastasi extra-epatiche rispetto a quelle epatiche.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References/Bibliografia
Parkin DM, Bray F, Ferlay J et al (2005) Global cancer statistics, 2002. CA Cancer J Clin 55:74–108
Jemal A, Siegel R, Ward E et al (2007) Cancer statistics, 2007. CA Cancer J Clin 57:43–66
Bentrem DJ, DeMatteo RP, Blumgart LH (2005) Surgical therapy for metastatic disease to the liver. Annu Rev Med 56:139–156
Livraghi T, Solbiati L, Meloni MF et al (2003) Treatment of focal liver tumors with percutaneous radiofrequency ablation complications encountered in a multicenter study. Radiology 226:441–451
Ruan DT, Warren RS (2005) Liverdirected therapies in colorectal cancer. Semin Oncol 32:85–94
Van de Velde CJH (2005) Treatment of liver metastases of colorectal cancer. Ann Oncol 16:144–149
Meyers MO, Sasson AR, Sigurdson ER (2003) Locoregional strategies for colorectal hepatic metastases. Clin Colorectal Cancer 3:34–44
Bilchik AJ, Rose DM, Allegra DP et al (1999) Radiofrequency ablation: a minimally invasive technique with multiple applications. Cancer J Sci Am 5:356–361
Gillams AR (2005) The use of radiofrequency in cancer. Br J Cancer 92:1825–1829
Buscarini E, Savoia A, Brambilla G et al (2005) Radiofrequency thermal ablation of liver tumors. Eur Rad 15:884–894
Stang A, Fischbach R, Teichmann W et al (2009) A systematic review on the clinical benefit and role of radiofrequency ablation as treatment of colorectal liver metastases. Eur J Cancer 45:1748–1756
Adam R, Pascal G, Castaing D et al (2004) Tumor progression while on chemotherapy. A contraindication to liver resection for multiple colorectal metastases? Ann Surg 240:1052–1064
Siperstein A, Berber E, Ballem N et al (2007) Survival after radiofrequency ablation of colorectal liver metastases: 10-year experience. Ann Surg 246:559–567
Jaskolka JD, Asch MR, Kachura JR et al (2005) Needle tract seeding after radiofrequency ablation of hepatic tumors. J Vasc Interv Radiol 16:485–491
Shibata T, Yamamoto Y, Yamamoto N et al (2003) Cholangitis and liver abscess after percutaneous ablation therapy for liver tumors: incidence and risk factors. J Vasc Interv Radiol 14:1535–1542
Choi D, Lim HK, Kim MJ et al (2005) Liver abscess after percutaneous ra diofrequency ablation for hepatocellular carcinomas: frequency and risk factors. AJR Am J Roetgenol 184:1860–1867
Livraghi T, Lazzaroni S, Meloni F, Solbiati L (2005) Risk of tumour seeding after percutaneous radiofrequency ablation for hepatocellular carcinoma. Br J Surg 92:856–858
Giorgio A, Tarantino L, de Stefano G et al (2005) Complications after percutaneous saline-enhanced radiofrequency ablation of liver tumors: 3-year experience with 336 patients at a single center. AJR Am J Roentgenol 184:207–211
De Meijer VE, Verhoef C, Kuiper JW et al (2006) Radiofrequency ablation in patients with primary and secondary hepatic malignancies. J Gastrointest Surg 10:960–973
Oshowo A, Gillams A, Harrison E et al (2003) Comparison of resection and radiofrequency ablation for treatment of solitary colorectal liver metastases. Br J Surg 90:1240–1243
Gillams AR, Lees WR (2005) Radiofrequency ablation of colorectal liver metastases. Abdom Imaging 30:419–426
Sorensen SM, Mortensen FV, Nielsen DT (2007) Radiofrequency ablation of colorectal liver metastases: long-term survival. Acta Radiol 48:253–258
Stangl R, Altendorf-Hofmann A, Charnley RM, Scheele J (1994) Factors influencing the natural history of colorectal liver metastases. Lancet 343:1405–1410
Solbiati L, Livraghi T, Goldberg SN et al (2001) Percutaneous radio-frequency ablation of hepatic metastases from colorectal cancer: long-term results in 117 patients. Radiology 221:159–166
Wolpin BM, Mayer RJ (2008) Systemic treatment of colorectal cancer. Gastroenterology 134:1296–1310
Cunningham D, Humblet Y, Siena S et al (2004) Cetuximab monotherapy and cetuximab plus irinotecan in irinotecanrefractory metastatic colorectal cancer. N Engl J Med 351:337–345
Yuste AL, Aparicio J, Segura A et al (2003) Analysis of clinical prognostic factors for survival and time to progression in patients with metastatic colorectal cancer treated with 5-fluorouracil-based chemotherapy. Clin Colorectal Cancer 2:231–234
Hurwitz H, Fehrenbacher L, Novotny W et al (2004) Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med 350:2335–2342
Fuchs CS, Marshall J, Mitchell E et al (2007) Randomized, controlled trial of irinotecan plus infusional, bolus, or oral fluoropyrimidines in first-line treatment of metastatic colorectal cancer: results from the BICC-C study. J Clin Oncol 25:4779–4786
Goldberg RM, Sargent DJ, Morton RF et al (2004) A randomized controlled trial of fluorouracil plus leucovorin, irinotecan, and oxaliplatin combinations in patients with previously untreated metastatic colorectal cancer. J Clin Oncol 22:23–30
Fong Y, Cohen AM, Fortner JG et al (1997) Liver resection for colorectal metastases. J Clin Onc 15:938–946
Gillams AR, Lees WR (2008) Fiveyear survival following radiofrequency ablation of small, solitary, hepatic colorectal metastases. J Vasc Interv Radiol 19:712–717
Livraghi T, Solbiati L, Meloni F et al (2003) Percutaneous radiofrequency ablation of liver metastases in potential candidates for resection the “test-of time” approach. Cancer 97:3027–3035
Machi J, Oishi AJ, Sumida K et al (2006) Long-term outcome of radiofrequency ablation for unresectable liver metastases from colorectal cancer: evaluation of prognosic factors and effectiveness in first- and second-line management. Cancer J 12:318–326
Rees M, Tekkis PP, Welsh FK et al (2008) Evaluation of long-term survival after hepatic resection for metastatic colorectal cancer: A multifactorial model of 929 patients. Ann Surg 247:125–135
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Veltri, A., Guarnieri, T., Gazzera, C. et al. Long-term outcome of radiofrequency thermal ablation (RFA) of liver metastases from colorectal cancer (CRC): size as the leading prognostic factor for survival. Radiol med 117, 1139–1151 (2012). https://doi.org/10.1007/s11547-012-0803-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-012-0803-3