Abstract
We develop a multiphasic hydrodynamic theory for biofilms taking into account interactions among various bacterial phenotypes, extracellular polymeric substance (EPS), quorum sensing (QS) molecules, solvent, and antibiotics. In the model, bacteria are classified into down-regulated QS, up-regulated QS, and non-QS cells based on their QS ability. The model is first benchmarked against an experiment yielding an excellent fit to experimental measurements on the concentration of QS molecules and the cell density during biofilm development. It is then applied to study development of heterogeneous structures in biofilms due to interactions of QS regulation, hydrodynamics, and antimicrobial treatment. Our 3D numerical simulations have confirmed that (i). QS is beneficial for biofilm development in a long run by building a robust EPS population to protect the biofilm; (ii). biofilms located upstream can induce QS downstream when the colonies are close enough spatially; (iii). QS induction may not be fully operational and can even be compromised in strong laminar flows; (v). the hydrodynamic stress alters the biofilm morphology. Through further numerical investigations, our model suggests that (i). QS-regulated EPS production contributes to the structural formation of heterogeneous biofilms; (ii) QS down-regulated cells tend to grow at the surface of the biofilm while QS up-regulated ones tend to grow in the bulk; (iii) when nutrient supply is sufficient, QS induction might be more effective upstream than downstream; (iv) QS may be of little benefit in a short timescale in term of fighting against invading strain/species; (v) the material properties of biomass (bacteria and EPS) have strong impact on the dilution of QS molecules under strong shear flow. In addition, with this modeling framework, hydrodynamic details and rheological quantities associated with biofilm formation under QS regulation can be resolved.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Bacteria are ubiquitous in nature as well as in our daily life. In general, bacteria usually do not exist as isolated cells but rather live in organized communities. These bacterial communities, where bacteria are less motile and are glued together by extracellular polymeric substance (EPS), are called biofilms. It is commonly perceived by the medical community that biofilms are responsible for many diseases or ailments associated with chronic infections, supported by the survey data that biofilms are present on the removed tissue of 80% of patients undergoing surgery for chronic sinusitis (Sanclement et al. 2005). Unlike a planktonic (free-swimming) bacterium, bacteria in biofilms appear to be more adaptive to the stressful environment; for instance, they are more tolerable to antimicrobial agents and hydrodynamic stress. As the result, biofilms are always hard to be eradicated by standard antimicrobial treatment (Lewis 2010), which perhaps explains the tendency for relapse of chronic diseases or ailments caused by biofilms.
One of the main features of biofilms, which makes bacteria in biofilms functioning quite different from planktonic bacteria, is that bacteria living in biofilms are observed to communicate, and furthermore, cooperate with each other by secreting solution-soluble signaling molecules known as autoinducers (Miller and Bassler 2001). For gram-negative bacteria, the particular molecules are acyl homoserine lactones (AHLs) (Whitehead et al. 2001). Experimental results have shown that bacteria can sense the concentration of the signaling molecules secreted by bacteria in the surrounding and function cooperatively to accomplish certain tasks when the concentration of autoinducers reaches a threshold value (Waters and Basseler 2005). This phenomenon is commonly known as quorum sensing (QS). The signaling molecule or the autoinducer is therefore called the quorum sensing molecule. This cell–cell communication mechanism via quorum sensing has been observed in many bacterial systems, such as vibrio fischeri (Nealson et al. 1970), where quorum sensing was first observed, Pseudomonas aeruginosa, Escherichia coli and so on. This mechanism allows unicellular microorganism to perform behavior like multicellular organism.
Quorum sensing has been shown to be responsible for mediating a variety of social activities in biofilms, which include the secretion of diverse by-products, biofilm growth (Chopp et al. 2002, 2003; Garcia-Aljaro and Melado-Rovira 2012), swarming motility and virulence gene expressions (Quinones et al. 2005), biofilm dispersion (Cárcamo-Oyarce et al. 2015; Solano et al. 2014), and antimicrobial resistance (Thompson et al. 2015). In particular, the phenomenon of quorum sensing regulating EPS production during biofilm formation has been widely reported, for instance in Marketon et al. (2003), Vuong et al. (2003), Waters and Basseler (2005).
EPS can be treated as the so-called public good since its a shared resource that each individual bacterium can take advantage of, even if it is produced by other bacteria. However, how quorum sensing regulation affect the EPS production is not clearly resolved. Besides, in some bacterial systems, like Vibrio cholerae, the EPS production is suppressed when the density of autoinducer reaches its threshold value (Heithoff and Mahan 2004), whereas in Pseudomonas aeruginosa, the EPS production would be facilitated once the quorum sensing is activated (Sakuragi and Kolter 2007). In addition, as the phenomenon involves transport of signaling molecules, the heterogenous structure in biofilm colonies and the solvent–biomass interaction cannot be ignored, as they can affect the convection and diffusion rates of the signaling molecules transported as well as the morphology of biofilm colonies. This motivates us to develop a full 3D mathematical model to study how quorum sensing regulates biofilm formation and development as well as the pros and cons of quorum sensing in an aqueous environment where hydrodynamic interaction between the various biofilm components and the ambient fluid flow is important.
The induction of quorum sensing is usually observed at high bacterial cell volume fractions, where the concentration of QS molecules is accumulated to a certain threshold value. However, a high bacterial cell density does not necessarily induce quorum sensing since many other factors have been observed to contribute to this induction such as nutrient supplies, hydrodynamic shear stress (Kirisits et al. 2007,) and the PH environment. Intuitively, a hydrodynamic flow may have a diluting effect on the signaling molecules, which then lead to a delay or even failure of QS induction by preventing QS molecules, like AHLs, from reaching a certain threshold value. For instance, in Kirisits et al. (2007), the authors claim that the amount of biomass required for the full QS induction of the population increases as the flow rate increases. On the other hand, coming with the inflow solvent, more essential substances, such as nutrient which enhances the production of QS molecules by bacteria, would be fresh in; thus hydrodynamic flow may lead to higher production of the signaling molecules and thereby facilitate QS induction. Little work has been done to analyze the correlations, not to mention taking the heterogenous structure factor of biofilms into account. This adequately serves as another motivation for us to develop a full 3D hydrodynamic biofilm model to study the hydrodynamic effect on quorum sensing.
In the literature, related with quorum sensing, some other factors have also been singled out for possible impact to biofilm development. For instance, recently, it has been verified that, except for cooperation, there probably exist cheaters in the bacterial population which exploit the signaling molecules produced by others (Popat et al. 2012). In Fauvart et al. (2012), the authors observed that quorum sensing coordinated secretion of rhamnolipid acts as a surfactant, which leads to finger pattern formation in swimming bacteria aggregates. With the ever-increasing knowledge about quorum sensing, researchers are working on developing potential applications by taking advantage of this cell–cell communication mechanism. For an overview of applications of quorum sensing in biotechnology, readers are referred to Choudhary and Schmidt-Dannert (2010). One promising application is that it is probably more efficient to use anti-quorum sensing treatment than antibiotic agents in treating some biofilm infections, which is likely to pose a selective pressure for the development of resistant mutants (Hentzer and Givskov 2003).
Inspired by experimental findings, researchers have come up with several mathematical models to study biofilm formation and function, as well as related issues. Concerning coupling biofilm growth with quorum sensing features, several mathematical models have been developed. Ward et al. (2001) proposed an ODE system modeling quorum sensing in bacteria and later extended by Koerber et al. (2002) into a 1D PDE system in space and time. The dependence of quorum sensing on the depth of a growing biofilm is discussed in Chopp et al. (2003). In Nadell et al. (2008), the evolution of quorum sensing in bacterial biofilms is simulated by an individual-based model. A delay model (Barbarossa et al. 2010) is proposed by assuming a (not yet detected) enzyme, which degrades the autoinducer into an active form. In Perez-Velazquez et al. (2015), quorum sensing regulation and its heterogeneity in Pseudomonas syringae on leaves is studied by treating it as a nonnegative stochastic process. Recently, Matur et al. (2015) proposed a single-cell spatial model for quorum sensing. The inhibition of quorum sensing is modeled by Fozard et al. (2012) as a stochastic process on the level of individual cells, claiming the time at which treatment is initiated is crucial for the effective prevention of quorum sensing. Jabbari et al. (2012a) models it by reactive dynamics; the study is further extended in Jabbari et al. (2012b) with spatial diffusion. The hydrodynamic effects on quorum sensing induction have also been investigated theoretically. A 2D partial differential equation model coupled with the Stokes flow is proposed in Frederick et al. (2010) and later the authors extended this model (Frederick et al. 2011) and proved its well posedness in Sonner et al. (2011). Another 2D continuum model with Stokes background flow is proposed in Vaughan et al. (2010), where the shape of biofilms at the onset of hydrodynamic shear is studied. Recently, authors in Uecke et al. (2014) model quorum sensing with a background flow by treating each bacteria as an individual. In Emerenini et al. (2015), the authors study the quorum-sensing-induced biofilm detachment and later the model is further analyzed in Emerenini et al. (2017). In Langebrake et al. (2014), the authors investigate QS inside colonies by studying the propagation of autoinducer waves, and some spatial–temporal patterns of autoinducers are also studied in Dilanji et al. (2012).
Despite the advances documented in the literature, there is very little work on 3D hydrodynamics of biofilms in the ambient fluid environment available, which takes into account the spatial–temporal heterogeneous structure of biofilms as well as the hydrodynamic flow effects. This is perhaps mainly because of computational challenges in resolving biofilms which are a truly 3D microorganism with highly heterogeneous spatial–temporal structures and complex dynamics.
In this paper, we develop a new, full 3D hydrodynamic model for the mixture system consisting of biofilms and the ambient fluid, extending our previous phase-field models for biofilms of two and three effective components (Lindley et al. 2011; Zhang et al. 2008a). This model couples hydrodynamics of the biofilm–solvent mixture to the quorum sensing activity, as well as dynamics of nutrient and antimicrobial agents. Using this model, we aim to investigate biofilm formation and development regulated by quorum sensing in an aqueous environment under hydrodynamical flows and possibly antimicrobial treatment. A second-order numerical scheme in both time and space based on finite difference methods is devised to solve the governing system of partial differential equations in the model. The numerical solver for the discretized equations is then implemented on graphical processing units (GPUs) in a 3D space and time.
The 3D numerical simulation tool is employed to investigate the interaction among QS molecules and various biomass components under a hydrodynamic environment. Given the complexity of the problem, we select a couple of important case studies to present in this paper. First of all, we benchmark the reactive kinetics with experimental results on the concentration of QS molecules and the cell density during biofilm development. Then, we apply the full 3D simulation tool to investigate the interaction between hydrodynamics and quorum sensing regulation. Finally, we examine the impact of hydrodynamic stresses on the biofilm morphology and biofilm material properties on QS molecule dilution under strong shear. The model and the accompanying computation tool are general enough to allow a wide spectrum of case studies for biofilm related issues, beyond what we report in this paper.
The rest of this paper is organized into four sections. In the second section, we present the detail of the hydrodynamic phase-field model for biofilms. Then, we design a linear numerical solver for the governing partial differential equations in the model using semi-implicit finite difference schemes and briefly discuss the implementation issue in the third section. In the fourth section, the numerical results and discussions are presented. In the last section, we give a concluding remark for the study.
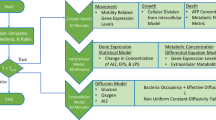
2 3D Hydrodynamic Model for Biofilms
In this section, we extend a previously developed two phase biofilm model (Zhang et al. 2008a) to include the effect of quorum sensing among the various bacterial phenotypes classified based on their quorum sensing abilities. The biofilm, consisting of bacteria, EPS, and solvent, is coarse-grained into a single fluid model with multi-components, in which different types of bacteria, EPS, as well as solvent are modeled effectively as separate fluid components. Quorum sensing molecules, nutrients, and antibiotic agents are treated as phantom materials which exhibit chemical reactive effects without their mass and volume being considered. Treating substances of such small molecules as phantom materials is an effective approximation to the complex mixture system given the little contribution from these materials to the transport of the total mass and volume of the entire material system.
2.1 Basic Notations
We study biofilms in a cubic domain \(\Omega =[0, \quad L_x] \times [0, \quad L_y] \times [0, \quad L_z]\), with \(L_x,L_y,L_z\) the length in x, y, z directions, respectively. Following our previous notational approaches (Zhang et al. 2008a), we denote the volume fraction of the effective solvent and biomass as \(\phi _s\) and \(\phi _n\), respectively. In biomass, we further divide it into EPS and bacteria, whose volume fractions are denoted by \(\phi _p\) and \(\phi _b\), respectively, i.e., \(\phi _n = \phi _p +\phi _b\). The incompressibility condition of the mixture system requires
Let \({\rho }_{b}, \mathbf {v}_{b}\), \({\rho }_{p}, \mathbf {v}_{p}\) and \({\rho }_{s}, \mathbf {v}_{s}\) be the density and velocity of the bacteria, EPS and solvent, respectively. Then the volume-averaged density and velocity for the mixture are denoted as
In the biofilm model, we classify the bacteria into four different phenotypes according to their responses to quorum sensing (QS) molecules (Frederick et al. 2011; Nadell et al. 2008): QS down-regulated bacteria (QS\(^{-}\)), QS up-regulated bacteria (QS\(^{+}\)), non-QS bacteria (non-QS), as well as the dead bacteria, whose volume fractions are denoted by \(\phi _{b1}, \phi _{b2}, \phi _{b3}\) and \(\phi _{b4}\), respectively. Hence, the volume fraction of the bacteria could be written as
We note that since the bacteria in biofilms are less motile (Guttenplan and Kearns 2013), we treat all bacteria having the same kinematics. In biofilms, QS\(^{-}\) are the bacteria, which can convert into QS\(^{+}\) once the concentration of QS molecules reaches a threshold value (Frederick et al. 2011; Nadell et al. 2008) whereas QS\(^{+}\) are the bacteria, which can convert into QS\(^{-}\) when the concentration of QS molecules is lower than the threshold. In another word, QS\(^{-}\) bacteria belong to the wild phenotype of the bacteria and QS\(^{+}\) bacteria represent the phenotype which has bonded QS molecules and certain activated gene transcription for enhanced EPS production and therefore reacts differently from its original phenotype. Non-QS bacteria represent those bacteria which are not affected by the concentration of quorum sensing molecules but may be regulated by other proteins in the biofilm. These non-quorum sensing bacteria can be regarded a phenotype, which do not prefer a social relation with other bacteria, or they can also be treated as a different type of bacteria, such as invaders. Dead bacteria are those which lose their vitality but are still attached in the biofilm.
In addition to the above bacterial phenotypes, we also keep track of the functional components whose volume are omitted in the model, but their chemical effects are retained as their mass fractions are much smaller than those of biomass and solvent. These include (i) nutrients, (ii) antimicrobial agents and (iii) quorum sensing molecules, whose concentrations are denoted by c, d and A, respectively.
We make a simplifying assumption here that all bacterial cells and EPS that they produce interact with solvent equally in the context of thermodynamics (i.e., entropic motion and mixing). Thus the effective free energy density functional of the biological system is proposed as follows using the extended Flory–Huggin’s mixing free energy density with an extra conformational entropy term (Cahn and Hilliard 1958; Cogan and James 2004; Cogan and Keener 2005; Zhang et al. 2008a; Zhao et al. 2016a, b)
where N is an effective polymerization index for the non-solvent biomass components, \(\gamma _1\) parametrizes the strength of the conformational entropy, \(\gamma _2\) parameterizes the strength of the bulk interaction potential, \(\chi \) is the mixing coefficient. We remark the mixing coefficient \(\chi \) may depend on \(\phi _b\) and \(\phi _p\). Here we treat it as constant, until further experiment data are accessible.
2.2 Transport Equations for Biomass Components
With the free energy, the transport equation for each bacterial type is assumed to be governed by a reactive Cahn–Hilliard-type equation (Cahn and Hilliard 1958; Cahn 1959)
where \(\lambda \) is the mobility parameter, \(\mu _j = \frac{\delta f}{\delta \phi _{bj}}\) is the chemical potential with respect to bacterial type j, and \(g_{bj}\) is the reactive rate for cell type j.
The reactive kinetics of these cell types include regulated growth due to nutrient, the phenotype conversion between the QS down-regulated and QS up-regulated cells, and decay due to antimicrobial agents as well as natural causes (death). We consider both mechanisms of logistic growth dynamics for the bacteria (Almeida et al. 2012; Perez-Velazquez et al. 2015) and the Monod growth kinetics due to nutrient supply (Frederick et al. 2011). The inclusion of the logistic growth in this model is based on our assumption that the bacterial growth slows down when the spacing among bacteria is squeezed, especially in a confined geometry. Mathematically, this assumption eliminates the potentially unbounded growth locally without the mechanism. Should we control the carrying capacity \(\phi _b^*\) as a large number, it would not qualitatively change the model prediction compared with the linear growth model in most cases. The QS down-regulated cells will produce the same phenotype, while the QS up-regulated cells will produce \(\beta \) fraction of QS down-regulated cells and \((1-\beta )\) fraction of QS up-regulated cells (Ward et al. 2001). For each phenotype, we consider their death rates due to natural causes, in which the decay rate for the dead cells is interpreted as the dissolving or conversion rate of the cell into solvent. Between the quorum sensing cells, there exists the up and down regulation coordinated by the concentration of the quorum sensing molecule AHL (Frederick et al. 2010, 2011; Nadell et al. 2008; Ward et al. 2001). The reactive kinetics for all cell types are given by the following
where the \(C_{bi}\) are the growth rates, \(\beta \) represents the percentage of QS\(^{+}\) bacteria that produces QS\(^{-}\) bacteria (Ward et al. 2001), \(C_{di}\) is the decay rate due to the antimicrobial treatment (Cogan 2012), \(r_i\) is the natural death rate (Frederick et al. 2010), \(k_{bi}, k_{di}\) are the half saturation constants in the respective Monod model for each bacterial phenotype, respectively, where \(i=1,2,3\). Here \(r_4\) is the natural conversion rate from dead bacteria into solvent due to cell lysis (Bayles 2007). And \(\phi _{b}^*\) is the carrying capacity of the bacterial volume fraction. In addition, the conversion rate between QS\(^{+}\) and QS\(^{-}\) are modeled by \(\frac{r_{12} A^n}{\tau ^n + A^n}\) and \(\frac{r_{21} \tau ^n}{\tau ^n + A^n}\), with \(\tau \) the threshold AHL concentration locally required for quorum sensing induction to occur (Frederick et al. 2011) and the exponent n the degree of polymerization in the synthesis of AHL (Frederick et al. 2011). While the concentration of AHL gets bigger, quorum sensing induction gradually takes place to increase the up-regulated bacterial cell population and decrease the down-regulated bacterial cell population. The non-QS bacterial cells are not affected by the quorum sensing molecules by definition. In this paper, we assume \(\beta =1\) to hypothesize that QS-regulated cells are born all as QS down-regulated.
The transport equation for EPS (\(\phi _p\)), which is the product of the live bacteria, is proposed as a reactive Cahn–Hilliard-type equation as well and is given by,
where \(\mu _p = \frac{\delta f}{\delta \phi _p}\) is the chemical potential with respect to \(\phi _p\), and \(g_p\) is the reactive term proposed as follows
Here \(C_{pj}\) is the EPS production rate with respect to the jth type cell, \(j=1,2,3\), facilitated by the nutrient (Zhang et al. 2008a). \(\phi _p^*\) gives the upper bound for the EPS production. We assume that some EPS can be dissolved into solvent naturally with rate \(r_p\) due to cell lysis (Bayles 2007) and accelerated by drugs with the rate given by \(C_{dp}. k_{pi}\) and \(k_{dp}\) are the half saturation constants for the Monod models used.
2.3 Transport Equations for the Functional Components
There are three functional components in our biofilm model, namely, nutrient, antimicrobial agents, and quorum sensing molecules, whose concentrations are denoted by c, d and A, respectively. These molecules all have small molecular weight compared with the components of the biomass, instead of tracking their volumes, we track their concentrations, which are governed by convection–diffusion–reaction equations. These molecules are transported with solvent, as they are dissolved in it.
Specifically, the transport equation for the concentration of the quorum sensing molecule AHL (A) is given by
where \(D_a\) is the diffusion coefficient and \(g_a\) is the reactive term given by
Here \(r_a\) is the decay rate of the AHL concentration due to binding with QS bacterial cells (QS receptors) (Waters and Basseler 2005), \(r_{a1}\) and \(r_{a2}\) are the background growth rate due to the down-regulated and up-regulated quorum sensing cells, respectively. \(C_{a1}\) and \(C_{a2}\) are growth rates, taking into account the effect that the concentration of AHL would affect the productivity of AHL by the bacteria positively (Fekete et al. 2010). Here \(A^*\) gives the upper bound of solubility for AHL (Abraham 2006). \(C_a\) is the decay rate of AHL due to antimicrobial agents and \(k_\mathrm{da}\) is the half saturation constant. The diffusion coefficient is given by following the Hinson model (Hinson and Kocher 1996),
where the first term on the right-hand side represents the reduction effect due to the presence of bacteria while the second term is an empirical fitting accounting for the reduction effect due to the presence of EPS. Here \(D_\mathrm{pr}\) is a model parameter fitted experimentally.
The transport equation for nutrient is given as follows
where \(D_c\) is the diffusion coefficient for nutrient transport and the reactive rate is given by
which represents the nutrient consumption rate due to bacterial metabolism (Frederick et al. 2010; Zhao et al. 2016a), representing the nutrient consumption due to both bacterial cell duplications and EPS production. For the diffusion rate of the nutrient (\(D_c\)), which is primarily oxygen in this model, Stewart (Stewart 1996) suggests it to be around \(75\%\) than that in the pure solvent. Since the molecular weight of oxygen is relatively small, it is supposed to penetrate EPS and the membrane of cells equally. So we set the diffusion rate of oxygen in biofilms as follows
where the second term in the expression represents the reduction in diffusion due to the presence of the biomass.
Finally, the transport equation for antibiotic agents is proposed as follows
where \(D_d\) is the diffusion rate of antimicrobial agents and
is the reactive term. Antibiotics (or antimicrobial agents) are assumed to be consumed by all bacterial cells and EPS at the same rate. Here \(r_d\) represents the natural decay rate of antimicrobial agents (Shen et al. 2016). For the diffusion rate \(D_d\), we also adopt a Hinson type model (Hinson and Kocher 1996)
We remark that, in the development of these transport equations, we try to include as many biological and physical effects as possible, which result in a large set of model parameters. Some of them can be measured or found in the literature, while others are obtained either using our best guesses or calibrated against available experiments. Nevertheless, all these effects included in the model are present in the biofilm system.
2.4 Momentum and Continuity Equation
In order to couple the biofilm components to hydrodynamics, we need the governing equation for the averaged velocity \(\mathbf {v}\). We assume the fluid mixture as solenoidal and impose the balance of linear momentum as follows
where \(\rho \) is the averaged density, p is the hydrostatic pressure, \(\tau \) is the extra stress, and the last term is the interfacial force due to the inhomogeneity of biomass distribution, derived from a variational principle (Chen 2002; Zhao et al. 2016d). In this paper, we propose the mixture as an extended Newtonian fluid, then the extra stress tensor is given by
where \(\eta _b\), \(\eta _p\) and \(\eta _s\) are the viscosity of bacteria, EPS and solvent. and \(\mathbf {D}= \frac{1}{2}(\nabla \mathbf {v}+ \nabla \mathbf {v}^T)\) is the rate of strain tensor. EPS is viscoelastic, whose relaxation time ranges from few seconds to over 10 min. When we study biofilm growth, the timescale is much larger than the EPS relaxation time. Therefore, treating the biomass in biofilms as an extended Newtonian is a reasonable approximation.
2.5 Boundary Conditions
In order to simulate biofilms in a fixed domain, we need to impose boundary conditions for the governing partial differential equations, which mimic common situations for biofilms being cultured and observed in vitro. In this paper, we focus on simulating biofilm flows in two special geometries: in a petri dish or a flow cell in 3D space and time.
To mimic the biofilm development in a petri dish, both x and z directions are assumed periodic. At boundaries in the y direction, no-flux boundary conditions are imposed for the biomass and functional components and no-slip boundary conditions are imposed for the velocity,
where \(\mathbf {n}\) is the unit outer normal. We also impose a nutrient feeding condition \(c|_{y=L_y} = c_0\) in place of the zero-flux condition wherever there is a steady supply of nutrient through the boundary at \(y=L_y\).
For the case of biofilms in a finitely long water channel, which we call it a flow cell, periodic boundary conditions are imposed in the z direction, and y direction is bounded by solid walls, as Eq. (21). The channel is finite in the x direction. So, there exist the inlet–outlet boundary conditions. The inlet velocity at \(x=0\) is given by
where \(p_0\) is a prescribed pressure gradient. Suppose that solvent has already reached equilibrium when flowing out of the cell at \(x=L_x\), we impose \(\mathbf {v}_x = 0\) at the outlet boundary. For the nutrient concentration c, we prescribe boundary condition
For QS molecules A, we prescribe boundary condition
Here we use the Dirichlet boundary condition for nutrient concentration c at the inflow, because fresh nutrient supply is provided with the inflow. For QS molecules, we did not consider any situation such as injecting A into the channel. Thus, we propose the flux-free boundary condition for A at the inflow, assuming that there are no biofilms upstream.
For the biomass components, we impose no-flux boundary conditions in the x direction, which applies to the situation where no biomass is flown in and out of the flow cell,
2.6 Non-dimensionalization
We denote \(t_0\) as the reference timescale, h the reference length scale, and \(c_0\), \(d_0\) and \(A^\star \) the characteristic concentration of the nutrient, antimicrobial agents and quorum sensing molecules, respectively. Then, we non-dimensionalize the variables as follows,
The following dimensionless parameters then emerge,
For simplicity, we drop the symbol \(\tilde{}\), and the non-dimentionalized equations in the hydrodynamic model for biofilms are summarized as follows:
where the reactive terms are given by
2.7 Numerical Methods
For the coupled PDEs in the governing system of equations, we devise a second-order numerical method to solve them. We note that, in the following notation, any variable with overline represents the second-order extrapolation. In the following, we denote the extrapolated data using over-lines; for instance, \(\overline{\mathbf {v}}^{n+1} = 2\mathbf {v}^n - \mathbf {v}^{n-1}\).
Given the initial condition (\(\mathbf {v}^0 = 0, s^0 = p^0=0, \phi _{bj}^0, \phi _p^0, c^0, d^0, A^0\)), we first compute (\(\mathbf {v}^1, s^1, p^1, \phi _{bj}^1, \phi _{p}^1, c^1, d^1, A^1\)) by a first-order scheme. Having computed (\(\mathbf {v}^{n-1}, s^{n-1}, p^{n-1}, \phi _{bj}^{n-1}, \phi _{p}^{n-1}, c^{n-1}, d^{n-1}, A^{n-1}\)) and (\(\mathbf {v}^{n}, s^{n}, p^{n}, \phi _{bj}^{n}, \phi _p^{n}, c^{n}, d^{n}. A^n\)), where \(n \ge 2\), we calculate (\(\mathbf {v}^{n+1}, s^{n+1}, p^{n+1}, \phi _{bj}^{n+1}, \phi _p^{n+1}, c^{n+1}, d^{n+1}, A^{n+1}\)) in the following steps,
-
1.
Prediction:
$$\begin{aligned} \left\{ \begin{array}{lll} &{}\rho ^{n+1} \left[ \frac{3 \mathbf {u}^{n+1}-4\mathbf {v}^n + \mathbf {v}^{n-1}}{2 \delta t}\right] +\rho ^{n+1} \overline{\mathbf {v}}^{n+1} \cdot \nabla \overline{\mathbf {v}}^{n+1} + \frac{1}{2} (\nabla \cdot (\rho ^{n+1} \overline{\mathbf {v}}^{n+1})) \overline{\mathbf {v}}^{n+1} , \\ &{}+ \frac{1}{R_{e_s}} \nabla s^n+ \nabla p^n - \frac{1}{R_{e_a}} \nabla ^2 \mathbf {u}^{n+1} = \overline{\mathbf {R}}^{n+1} - \frac{1}{R_{e_a}} \nabla ^2 \overline{\mathbf {v}}^{n+1}, \\ &{} \mathbf {u}^{n+1} \cdot \mathbf {n}|_{y=0,L_y} = 0, \\ &{}\mathbf {u}^{n+1}|_{x=0} = \mathbf {v}_0, \quad \mathbf {u}^{n+1}_x|_{x=L_x} = 0, \end{array} \right. \end{aligned}$$ -
2.
Projection:
$$\begin{aligned} \left\{ \begin{array}{lll} - \nabla \cdot \left( \frac{1}{\rho ^{n+1}}\nabla \psi ^{n+1}\right) =&{} \nabla \cdot \mathbf {u}^{n+1}, \\ \frac{\partial \psi ^{n+1}}{\partial n}|_{y=0,L_y} =&{} 0 , \\ \frac{\partial \psi ^{n+1}}{\partial n}|_{x=0,L_x} =&{} 0, \end{array} \right. \end{aligned}$$ -
3.
Correction:
$$\begin{aligned} \left\{ \begin{array}{lll} \mathbf {v}^{n+1} = &{} \mathbf {u}^{n+1} + \frac{1}{\rho ^{n+1}} \nabla \psi ^{n+1} ,\\ s^{n+1} = &{} s^n - \nabla \cdot \mathbf {u}^{n+1} ,\\ p^{n+1} = &{} p^n - \frac{3 \psi ^{n+1}}{2\delta t} + \frac{1}{R_{e_a}} s^{n+1}, \end{array} \right. \end{aligned}$$with
$$\begin{aligned} \overline{\mathbf {R}}^{n+1} = - \Gamma _1 \nabla ^2 \overline{\phi }^{n+1}_n \nabla \overline{\phi }^{n+1}_n + \nabla \cdot \left( \overline{\phi }^{n+1}_n \overline{\tau }^{n+1}_n + \overline{\phi }^{n+1}_s \overline{\tau }^{n+1}_{s}\right) , \end{aligned}$$and
$$\begin{aligned} \frac{1}{R_{e_a}} = \frac{\phi _{b,\max }}{R_{e_b}} + \frac{\phi _{p,\max }}{R_{e_p}} +\frac{1-\phi _{p,\max }-\phi _{b,\max }}{R_{e_s}}, \end{aligned}$$where \(R_{e_b}\), \(R_{e_p}\) and \(R_{e_s}\) are the Reynolds numbers for bacteria, EPS and solvent, respectively.
-
4.
Update biomass (\(\phi _{bj}, \phi _p\))
$$\begin{aligned} \left\{ \begin{array}{lll} \frac{3\phi _{bj}^{n+1} - 4\phi _{bj}^n + \phi _{bj}^{n-1}}{2\delta t} + \nabla \cdot ( \overline{\phi }_{bj}^{n+1} \mathbf {v}^{n+1}) &{}= &{} \nabla \cdot \left[ \Lambda \overline{\phi }^{n+1}_{bj} \mathcal {F}\left( \sum _{i=1}^4 \phi _{bi}^{n+1} +\phi _p^{n+1} \right) \right] + g_{bi}^{n+1}, \\ \frac{3\phi _p^{n+1} - 4\phi _p^n + \phi _p^{n-1}}{2\delta t} + \nabla \cdot ( \overline{\phi }_p^{n+1}\mathbf {v}^{n+1}) &{} = &{} \nabla \cdot \left[ \Lambda \overline{\phi }^{n+1}_{p} \mathcal {F}\left( \sum _{i=1}^4 \phi _{bi}^{n+1} +\phi _p^{n+1} \right) \right] + g_p^{n+1}, \end{array} \right. \!\!\!\!\nonumber \\ \end{aligned}$$(28)where \(j=1,2,3,4\) and
$$\begin{aligned} g^{n+1}_{b1}= & {} \left[ \frac{C_{b1} \overline{c}^{n+1}}{k_{b} + \overline{c}^{n+1}} \left( 1- \frac{\overline{\phi _b}^{n+1}}{\phi _{b,\max }}\right) -r_1 - \frac{r_{12} (\overline{A}^{n+1})^2}{\tau ^2 + (\overline{A}^{n+1})^2} - \frac{C_{d1} \overline{d}^{n+1}}{k_{d1} + \overline{d}^{n+1}}\right] \phi _{b1}^{n+1}\nonumber \\&+ \frac{r_{21} \tau ^2}{\tau ^2 + (\overline{A}^{n+1})^2} \overline{\phi _{b2}}^{n+1} + \beta C_{b2} \frac{\overline{c}^{n+1}}{k_{b}+\overline{c}^{n+1}}\left( 1-\frac{\overline{\phi _b}^{n+1}}{\phi _{b,\max }}\right) \overline{\phi _{b2}}^{n+1} \nonumber \\ g^{n+1}_{b2}= & {} \left[ \frac{ (1-\beta ) C_{b2} \overline{c}^{n+1}}{k_{b} + \overline{c}^{n+1}} \left( 1- \frac{\overline{\phi _b}^{n+1}}{\phi _{b,max}}\right) -r_2 -\frac{r_{21} \tau ^2}{\tau ^2 + (\overline{A}^{n+1})^2} - \frac{ C_{d2} \overline{d}^{n+1}}{k_{d2} + \overline{d}^{n+1}} \right] \phi _{b2}^{n+1}\nonumber \\&+ \frac{r_{12} (\overline{A}^{n+1})^2}{\tau ^2 + (\overline{A}^{n+1})^2} \overline{\phi _{b1}}^{n+1}, \nonumber \\ g^{n+1}_{b3}= & {} \left[ \frac{C_{b3} \overline{c}^{n+1}}{k_{b} + \overline{c}^{n+1}} \left( 1- \frac{\overline{\phi _b}^{n+1}}{\phi _{b,max}}\right) -r_3 - \frac{ C_{d3} \overline{d}^{n+1}}{k_{d3} + \overline{d}^{n+1}} \right] \phi _{b3}^{n+1}, \nonumber \\ g^{n+1}_{b4}= & {} \sum _{i=1}^3 \left[ r_i + \frac{C_{di} \overline{d}^{n+1}}{k_{d3}+\overline{d}^{n+1}} \right] \overline{\phi }_{bi}^{n+1} - r_4 \phi _{b4}^{n+1}, \nonumber \\ g_p^{n+1}= & {} \sum _{i=1}^3 \frac{C_{pi} \overline{c}^{n+1}}{k_{pi}+ \overline{c}^{n+1}} \overline{\phi }_{bi}^{n+1} \left( 1- \frac{\phi _p^{n+1}}{\phi _{p,\max }}\right) +\alpha r_4 \overline{\phi }_{b4}^{n+1} - r_p \phi _p^{n+1} \nonumber \\&- \frac{C_{dp} \overline{d}^{n+1}}{k_{dp}+\overline{d}^{n+1}} \phi _p^{n+1}, \end{aligned}$$(29)where
$$\begin{aligned} {\mathcal {F}}(\phi ) =\Gamma _2 \left( \frac{1}{N} \frac{1}{\overline{\phi }^{n+1}_n+\varepsilon } + \frac{1}{1-\overline{\phi }_n^{n+1}} - 2\chi \right) \nabla \phi - \Gamma _1 \nabla \nabla ^2 \phi . \end{aligned}$$ -
5.
Update functional components:
$$\begin{aligned} \left\{ \begin{array}{lll} \frac{3 \phi _s^{n+1}c^{n+1}- 4 \phi _s^n c^n + \phi _s^{n-1} c^{n-1}}{2 \delta t} + \mathbf {v}^{n+1} \cdot \nabla \left( c^{n+1}\phi _s^{n+1}\right) = \nabla \cdot \left( D_s^{n+1} \phi _s^{n+1} \nabla c^{n+1}\right) + \overline{g}_c^{n+1} , \\ \frac{3 \phi _s^{n+1}d^{n+1}- 4 \phi _s^n d^n + \phi _s^{n-1} d^{n-1}}{2 \delta t} + \mathbf {v}^{n+1} \cdot \nabla \left( d^{n+1}\phi _s^{n+1}\right) = \nabla \cdot \left( D_e^{n+1} \phi _s^{n+1} \nabla d^{n+1}\right) + \overline{g}_d^{n+1} ,\\ \frac{3 \phi _s^{n+1}A^{n+1}- 4 \phi _s^n A^n + \phi _s^{n-1} A^{n-1}}{2 \delta t} + \mathbf {v}^{n+1} \cdot \nabla \left( A^{n+1}\phi _s^{n+1}\right) = \nabla \cdot \left( D_a^{n+1} \phi _s^{n+1} \nabla a^{n+1}\right) + \overline{g}_a^{n+1}. \end{array} \right. \!\!\!\! \nonumber \\ \end{aligned}$$(30)where the discrete schemes for reactive terms are summarized below
$$\begin{aligned} g_a^{n+1}= & {} - r_a A^{n+1} + \left( \sum \limits _{i=1}^2 \left( r_{ai} + \frac{C_{ai} A^n }{k_{ai}+A^n}\right) \phi ^{n+1}_{bi} \right) \left( 1 - \frac{\overline{A}^{n+1}}{A_{\max }}\right) \nonumber \\&-\frac{C_a\overline{d}^{n+1}}{k_\mathrm{da} + \overline{d}^{n+1}} A^{n+1},\nonumber \\ g_c^{n+1}= & {} -\sum \limits _{i=1}^3 \frac{r_{bi} c^n}{k_{b}+c^n}\phi ^{n+1}_{bi} - \sum \limits _{i=1}^3 \frac{r_{pi} c^n}{k_{pi}+c^n} \phi ^{n+1}_{bi}, \nonumber \\ g_d^{n+1}= & {} - \frac{d^n}{k_d + d^n} \left( \sum \limits _{i=1}^4 r_{di} \phi _{bi}^{n+1}+ r_{dp} \phi _p^{n+1}\right) - r_d d^{n+1} . \end{aligned}$$(31)
The schemes above are presented as semi-discrete. The spatial discretization is carried out using central finite differences (Zhao et al. 2016c, e, 2017). The boundary conditions are discretized using the biased differencing at the solid boundaries. This scheme is implementation on GPUs using CUDA. We use a preconditioned BiCG solver (using the package CUSP 2012) to solve the sparse linear system generated from the full discretization using finite difference methods, where the preconditioner is constructed as the inverse of a good approximation of the implicit operator by approximating the variable coefficient with a constant coefficient. The preconditioner can then be computed using the fast Fourier transform. The data are saved as HDF5 format, which can be visualized using VisIt software (Childs et al. 2012). Currently, the solver can deal with up to \(256^3\) meshes in space, limited by the global memory size of a single GPU. The scheme is theoretically second order in space and time. Numerical convergence tests have been conducted and the convergence rate is confirmed.
3 Results and Discussion
The hydrodynamic model of biofilms developed is applied to study dynamic problems involving biofilms immersed in a viscous solvent. In the following, we apply it to investigate a few specific cases related to quorum sensing in biofilm formation and development, which include the effect of the threshold for quorum sensing induction and its influence on biofilm formation, and the hydrodynamical effect on the distribution of autoinducers and its consequence to biofilm development.
As alluded to earlier, the model has many parameters, and some of them can be calibrated via experiments, while others are either obtained from the literature or from our best guesses. For instance, the washout rates in the model, \(r_1, r_2, r_3, r_4, r_p, r_a\) and \(r_d\) are normally small; so, we set them into zero in the following case studies. For the monod constants (the k’s), we choose a common value \(3.5 \times 10^{-4}\) kg/m\(^3\) as used in Alpkvist et al. (2006). All the other parameters used in this paper are summarized in Table 1, unless mentioned otherwise.
3.1 Reactive Kinetics
We begin with the study on reactive kinetics in this model to elucidate bulk reactions for each component on two important mechanisms during biofilm development. By neglecting hydrodynamics, diffusive effects, and the drug effect in the model, and assuming nutrient is sufficiently supplied (setting \(c=1\)), we arrive at the governing equations for reactive kinetics in biofilms,
We fit the model parameters to the experiment in Quinones et al. (2004). The result is shown in Fig. 1. It agrees very well with the experiment, demonstrating the usefulness of the simplified kinetic model.
Fitting of the model to an experiment (Quinones et al. 2004). This demonstrates that the reactive kinetic model (or homogeneous model) predicts the experimental data on the AHL concentration and cell number density very well. The initial condition of the homogeneous model is \((\phi _{b1},\phi _{b2},\phi _{b3},\phi _p,A)=(3 \times 10^{-5},0,0,0,0)\), which is equivalent to \(10^6\) CFU/ml QS\(^{-}\) bacteria, according to Table 1. Here \(r_{a1}=r_{a2}=3.1 \times 10^{-8}\), \(C_{a1}=C_{a2}=3.1 \times 10^{-7}\) and \(C_{b1}=C_{b2}=1.11 \times 10^{-4}\)
A complete parameter study of this spatially homogeneous reaction model perhaps would warrant another paper, which will not be pursued in this paper.
We next examine the full hydrodynamic model for two quorum sensing related mechanisms that affect heterogeneous biofilm formation and functioning.
3.2 Biofilm Formation Coordinated by Quorum Sensing
Biofilm formation is a process in which bacteria aggregate in space and secrete glue-like exopopolysacharrides (EPS) to form a polymer-network around the bacteria, which creates a barrier between the bacteria and the surrounding media. It has been observed that quorum sensing controls biofilm formation in vibrio cholerae (Hammer and Bassler 2003) by regulating transcription of genes involved in expopolysacharride production. Other observations on quorum sensing regulated EPS production can also be found in Pseudomonas aeruginos (Sakuragi and Kolter 2007), sinorhizobium meliloti (Marketon et al. 2003), as well as in some other bacterial systems (Vuong et al. 2003). And biofilms adapt their architecture in order to cope with the hydrodynamic conditions and nutrient availability in Teodosio et al. (2011).
However, there has not been a consensus on what is the role of quorum sensing during biofilm formation. In this section, we will focus on simulating the mechanism of quorum-sensing-regulated EPS production using the model in a heterogeneous biofilm colony and studying pros and cons of QS regulation during biofilm formation in 3 dimensional space.
3.2.1 Biofilm Formation
First of all, we investigate heterogeneous biofilm formation in an aqueous environment where laminar flows are present outside the biofilm. The scenarios include (i) a culture petri dish filled with water where nutrient is supplied from the top surface; or (ii) a long water channel where nutrient is constantly supplied through the flowing water. For simplicity, we suppress the non-QS-regulated bacteria in the study of the first case since they are not important in this case, i.e., we consider a biofilm system where only QS-regulated bacteria are present initially. We set the initial value of \(\phi _{b3}\) (the volume faction of non-QS-regulated bacteria) to zero and assume that the dead bacteria are all dissolved in the solvent instantly. This way, we can examine how quorum sensing induction takes place and how biofilm dynamics are coordinated by the distribution of QS molecules.
Figure 2 depicts a simulation using the boundary condition (21), where initial conditions and solutions at \(t=300\) are shown. In this simulation, some QS down-regulated bacteria (QS\(^{-}\)) are assumed to be attached to the substrate surface initially, and the bacterial cell reproduction ensues as soon as the simulation begins. As a result, the QS molecule is produced and its concentration keeps growing. As the concentration of the quorum sensing molecules approaches the threshold value \(\tau \), QS\(^{-}\) start to bind with QS molecules and undergo a phenotype change into QS up-regulated bacteria (QS\(^{+}\)), for which the production for EPS is switched on. The biofilm colony starts growing exponentially by reproducing bacteria as well as secreting EPS. With more QS molecules produced, QS\(^{+}\) eventually outgrow QS\(^{-}\) and populate the bulk part of the biofilm colony.
Our 3D model predicts that QS-regulated EPS production contributes to the formation of heterogeneous biofilm structures. As shown in Fig. 2, this mushroom-shaped biofilm morphology qualitatively agrees with the published result on the morphological pattern in Pseudomonas aeruginosa biofilms (Parsek and Tolker-Nielsen 2008). The 3D results for volume fractions of total bacteria, QS down-regulated and QS up-regulated bacteria, EPS, and the total biomass are depicted in Fig. 2, respectively.
In addition, our model also suggests the heterogenous distribution of live bacteria and QS molecules. As shown in Fig. 2b, the distribution of live bacteria is highly heterogeneous, QS\(^{-}\) are mainly located at the surface of the biofilm, where they can access nutrients more easily, and QS\(^{+}\) are located more in the bulk of the biofilm colony. In order to show details of this process, plots of three slices at \(x=0.16,y=0.04,z=0.16\) for different biofilm components are shown in Figs. 2 and 3, respectively. In Fig. 3c, where the average velocity is small, the distribution of the QS molecules (AHLs) is dominated mainly by spatial diffusion. We observe that QS molecules are mainly distributed within the biofilm colony although small amount can diffuse into the solvent, illustrated by a three-slice plot in Fig. 3b. The biomass flux at \(z=0.5\) is plotted in Fig. 3d. It shows that it is mainly distributed at the top surface between the biomass the surrounding solvent, where there is more nutrient available near the surface than in other places so that the biomass tends to grow the fastest in these places. A pressure profile is also plotted in a 2D slice, which shows that high pressure is observed in the interior while low one is present near the surface of the biofilm.
This numerical simulation explains the growth process of the biofilm due to QS-regulated bacteria and QS-regulated EPS production together with some hydrodynamic signatures. Of course, the biomass can’t grow without a sufficient nutrient supply. But, in this case study, we assume its role is secondary.
(Color figure online) Biofilm formation with EPS production coordinated by quorum sensing in a biofilm in which non-QS bacteria are neglected. This simulation shows several clusters of QS\(^{-}\) in a developing heterogeneous biofilm colony, where EPS production during biofilm formation is regulated by quorum sensing. The initial profile is shown in (a). The characteristic timescale is \(t_0=10^3\) s and the simulation is conducted from \(t=0\) to \(t=300\), which represents roughly 3.4 days in real time. b A time \(t=300\), the bacterial distribution in terms of volume fractions is shown, where the red represents QS\(^{-}\) and the blue represents QS\(^{+}\); they are mixed. In the result, more QS\(^+\) are observed in the bulk of the biofilm while more QS\(^-\) show up near the biofilm surface. In c–f, three 2D slices of QS\(^{-}\), QS\(^{+}\), EPS and the total biomass at \(t=300\) are depicted, respectively, from which more interior details of the biofilm colony are on display
(Color figure online) Velocity, pressure and functional components from the simulation in Fig. 2. a–b 2D slices for the distribution of nutrient and QS molecules, AHL. c–e 2D slice at \(z=0.5\) of the average velocity \(\mathbf {v}\), the biomass flux and the hydrostatic pressure at \(t=300\), respectively. Low pressures occur in the interfacial region where the biofilm tends to grow. d Depicts explicitly the biomass flux given by \(\phi _n\mathbf{v}_n\). This shows that QS molecules (AHL) are mainly concentrated in the biofilm colony, but small amount is diffused into the surrounding aqueous environment
3.2.2 Benefits and Detriments of Quorum Sensing Regulation
With biofilm formation regulated by quorum sensing, a straight forward question is whether quorum sensing is always beneficial? Experimental findings seem to suggest the answer is ‘it depends.’ As the production of QS molecules is energetically costly, i.e., consuming ATP (Keller and Surette 2006), bacteria probably would switch on the quorum sensing mechanism to start producing QS molecules whenever they are in the ‘right time’ and ‘right place.’ In this subsection, we would like to further investigate the so-called right time and right place such that QS regulation for EPS production can be in favor of the biofilm development.
Our numerical study confirms (Frederick et al. 2011) that quorum sensing regulation is not beneficial to biofilm development in a short time, especially, in terms of gaining biomass. To explore the feature, we conduct another numerical simulation, where the quorum sensing mechanism is switched off by setting \(r_{21}=r_{12} = 0\) while the other model parameters are retained as in the previous numerical experiment. It is shown in Fig. 4 that the volume fraction of the total live bacteria without quorum sensing regulation is higher than that with quorum sensing regulation switched on. However, the total biomass in the system without QS regulation, including EPS and bacteria, is much less than that with QS regulation. This indicates that QS regulation is not a way to reproduce bacteria, but rather a way to increase the amount of EPS so as to expand biofilm colony, especially, in a short time period. The result agrees qualitatively with the experimental observation reported in Xavier and Foster (2007).
(Color figure online) Comparison between bacterial volume fractions during biofilm development with and without quorum sensing. Here we set \(r_{12}=r_{21}=0\) for simplicity. a Volume fractions of total biomass. b Volume fractions of QS down-regulated bacteria. c Volume fractions of QS up-regulated bacteria. d Volume fractions of total live bacteria. The initial conditions are the same as the one used in Fig. 2. With quorum sensing, the total biomass is higher than without, but the total live bacteria is not necessarily higher
In the long time period, our numerical studies confirm that (Frederick et al. 2011) QS regulation is of benefits to stabilizing biofilm colony and settling on the host, as well as protecting bacteria from environmental stress. Given the protective nature of EPS, the biofilm colony can become more stable and resistant to external stress while more EPS is produced. EPS can protect bacteria from being attacked by antimicrobial agents or immune systems of the host. To illustrate the beneficial effect of quorum sensing, we conduct a numerical experiment with the drug/disinfectant effect included. The result is shown in Fig. 5. In this simulation (see Fig. 5f), non-QS-regulated bacteria are killed dramatically while QS-regulated bacteria are killed in a much slower pace. Our model and simulation confirm that EPS acts as a barrier to protect bacteria from being attacked by antimicrobial agents. In Fig. 5a, b, we notice that bacteria staying within the environment filled with EPS lives longer while being treated by antimicrobial agents. In Fig. 5e, we see that antimicrobial agents penetrate less in the place with more EPS than in the place with less EPS. This numerical result agrees qualitatively with the claim made in Frederick et al. (2011) that biofilms can benefit from quorum sensing induced EPS production if bacteria cells have the objective of acquiring a thick, protective layer of EPS.
(Color figure online) Biofilm development subject to both quorum sensing and antibiotic (drug) treatment. The initial biofilm profile is a grown biofilm at \(t=252\) from Fig. 6. Antimicrobial agents are dosed through the top boundary. Here the characteristic time is \(t_0=10\) s. a–c The distribution of the live bacteria, EPS, dead bacteria in their volume fractions at \(t=94\), respectively. d, e Three slices of dead bacteria and antibiotics at \(t=94\), respectively. f shows total volume fractions of QS bacteria and non-QS bacteria from \(t=0\) to \(t=600\), respectively. After a long period of antimicrobial treatment, the bacterial population is reduced to a negligible level in this simulation
In addition, via numerical simulations, our model predicts that quorum sensing regulation is not beneficial to biofilm formation in a short time period when competing with invading strains of bacteria. In other words, given a highly reproductive invasion strain (like non-QS-regulated bacteria), QS-regulated bacteria can lose the competition such that the non-QS-regulated bacteria can take over and eventually populate the whole biofilm colony, which is mentioned in the case study of bulk reactive kinetics. To illustrate the point, let’s consider a biofilm system with three different phenotypes of QS-regulated bacteria: QS\(^{-}\), QS\(^{+}\) and non-QS. In this system, we suppose that non-QS-regulated bacteria have the same reproductive rate as that of QS\(^{-}\), but do not bind QS molecules and convert themselves into QS\(^{+}\). We conduct a numerical simulation, taking into account all these three phenotypes to study the detail on how the interaction between these bacteria can affect biofilm development via different self-production and EPS production rates. In this simulation, initially some QS\(^{-}\) and non-QS-regulated bacteria with the same volume fraction are assumed to be attached to the substrate surface. We assume \(C_{b1}=C_{b3}\) and the growth rate for QS up-regulated bacteria \(C_{b2}\) is smaller. A 3D prediction of the biofilm growth process is shown in Fig. 6. In Fig. 6b, non-QS-regulated bacteria outgrow the QS-regulated bacteria and take over the whole biofilm colony eventually. This is because the QS-regulated bacteria undergo mutual conversion which slows down the reproduction of QS\(^+\) bacteria as well as QS\(^-\). The total volume fraction for each type of bacteria is plotted in Fig. 6c, where non-QS bacteria grow exponentially, while QS-regulated bacteria grows only slightly. Since non-QS-regulated bacteria have a higher effective reproduction rate than those of QS-regulated ones combined, non-QS-regulated bacteria eventually grow faster to gain more access to nutrient. Hence, the density of non-QS-regulated bacteria eventually surpasses that of QS-regulated bacteria in the majority part of the biofilm colony due exclusively to the quorum sensing effect.
(Color figure online) Biofilm development with both QS bacteria and Non-QS bacteria in the biofilm. Randomly distributed biofilm colonies are given initially. Here, the characteristic time is \(t_0=10^3\) s. a Initial bacterial volume fraction. b Bacterial volume fractions at time \(t=252\), where purple represents non-QS bacteria and red represents QS bacteria. c The volume fractions for each bacterial phenotype in the entire biofilm colony, where non-QS-regulated bacteria outgrow QS-regulated bacteria. d The volume fraction of non-QS bacteria. e The volume fraction of QS bacteria at \(t=252\). f–i 2D slices at \(z=0.5\) for non-QS, QS bacteria, hydrostatic pressure, and biomass flux \(\phi _n \mathbf {v}_n\) at \(t=252\), respectively. The non-QS-regulated bacteria outgrow the QS-regulated bacteria in most part of the biofilm except in regions near the bottom of the domain. The hydrostatic pressure is again small near the interface and large in the interior of the biofilm. Here \(C_{b1}=C_{b3}=4.0 \times 10^{-4}\) and \(C_{b2}=10^{-4}\)
We remark that these findings perhaps also explain why antimicrobial treatment is more effective for young biofilms, where EPS layer tends to be thinner. When biofilms have grown for weeks, the effect of antimicrobial agents drops significantly. When the biofilm is young, there exists less EPS surrounding it; when the biofilm is “old,” however, the QS-induced EPS has already accumulated around bacteria to prevent drugs from penetrating deeper into the biofilm. This agrees qualitatively well with the observation reported in Shen et al. (2011) that biofilms at an earlier age is more susceptible to antimicrobial treatment than those in an older age.
Next, we turn to discuss the interaction between hydrodynamics and quorum sensing during the development of biofilms in an aqueous environment.
3.3 Role of Hydrodynamics on Quorum Sensing
3.3.1 Quorum Sensing in an Aqueous Environment
Many biofilms grow in an aqueous environment encompassing laminar flows, such as river bank or pipes, or quiescent aqueous environment. It is observed that a flow environment can impact on QS induction during biofilm formation (Kirisits et al. 2007). In return, the hydrodynamically altered QS induction can affect biofilm structures and functions. Recently, in Kim et al. (2016), authors found flows assist long-distance QS by enhancing autoinducer accumulation.
Here, we use the hydrodynamic model along with the computational tool to investigate the effect in detail. For simplicity, we consider the case where there are only QS-regulated bacteria in the biofilm system. The boundary conditions used in the simulation are set up to mimic biofilm development in a flow cell, where the fluid (solvent) flows through the biofilm domain continuously. We conduct numerical simulations of the biofilm system subject to varying inlet fluid velocities and focus on biofilm development under hydrodynamic shear. The result is summarized in Fig. 7. Initially, some QS\(^{-}\) bacteria are attached at the center of the flow cell. Solvent containing nutrient keeps flowing through the flow cell while the bacteria grow into a biofilm colony. As shown in Fig. 7c, a higher hydrodynamic shear can actually dilute the concentration of QS molecules, which leads to a delay in QS induction. 2D slices (at \(z=0.5\)) of QS molecules and QS\(^{+}\) bacteria at time \(t=25\) are shown in Fig. 8. This numerical experiment demonstrates that the maximum concentration of QS molecules and the volume fraction of QS\(^{+}\) are higher in the flow cell when subject to a slower inlet speed and than subject to a higher inlet speed.
(Color figure online) Biofilm development subject to varying inlet flow velocities. This gives a comparative study on how some QS\(^{-}\) bacteria develop into a biofilm colony under different inlet velocities \(v_0 = p_0 y(1-y)\), where \(p_0 = 1, 10, 20\). a The initial distribution of QS\(^{-}\) bacteria in volume fractions. b 2D slice (\(z=0.5\)) of the initial velocity profile. c The total volume of QS\(^{-}\) bacteria, QS\(^{+}\) bacteria, total bacteria, as well as the concentration of AHL, at three selected inlet velocities. The volume fraction of QS\(^-\) bacteria and the total bacteria scales with the inlet speed, i.e., the higher the inlet speed is, the larger the volume fractions are, whereas the volume fraction of QS\(^+\) bacteria and the AHL concentration scales inversely with the inlet speed, i.e., the larger the inlet speed is, the smaller the volume fraction and the concentration are
(Color figure online) The volume fraction of QS\(^+\) and AHL concentration at \(t=15\) with respect to three selected inlet speeds. This figure shows 2D slices at \(z=0.5\) for the simulation depicted in Fig. 7. a–c AHL distribution at \(t=15\) for inlet velocity \(v_0=p_0y(1-y)\) with \(p_0=1,10,20\), respectively. d–f 2D slices of volume fraction of QS\(^+\) (\(\phi _{b2}\) ) at \(t=15\) for inlet velocity \(v_0=p_0y(1-y)\) with \(p_0=1,10,20\), respectively
Our numerical studies confirm that hydrodynamics have a pretty strong influence on quorum sensing in that a higher inlet flow dilutes the concentration of the QS signaling molecules (Frederick et al. 2010; Uecke et al. 2014; Vaughan et al. 2010), which then requires a higher cell density and delayed time for quorum sensing induction to take place (Kim et al. 2016; Vaughan et al. 2010). This means that QS may not be fully operational at high flow rates and thus indicates that the role of QS in biofilm formation can be compromised in a flowing environment.
In addition, through numerical study, our model suggests, with the diluted QS molecular concentration, the biofilm in the flow cell with a higher inlet velocity has less QS\(^{+}\) bacteria. Note however that a biofilm with a higher inlet velocity results in more total bacteria. This is because the nutrient supply at a higher inlet velocity can bring in more nutrient and thus facilitates the growth of all bacteria. A robust biofilm colony needs not only the growth of bacteria, but also the build-up of EPS. Since the EPS growth by QS\(^+\) is more than that by QS\(^-\), a reduced QS\(^{+}\) production leads to the reduced EPS production in the biofilm. Even though the total bacteria is not reduced, the EPS production is reduced by the higher inlet velocity. We note that how quickly QS molecules are produced, how quickly the QS molecules diffuse away from the biofilms and the critical concentration of QS molecules are three main factors affecting biofilm formation. When taking into account of the effect of nutrient, especially, when the growth due to nutrient dominates, the diffusion rate and the consumption rate of nutrient can be essential for the biofilm growth as well. We hope the theoretical result can be quantitatively confirmed by experiments in the future.
3.3.2 Effect of Nutrient
Compared with a quiescent aqueous environment, the salient difference of biofilms developed in a flow cell is their structural heterogeneity in the flow field, due to the hydrodynamic stress and inhomogeneous nutrient distribution. Mass transfer in the fluid mixture can be affected by hydrodynamics of the bulk fluid and the morphology of the biofilm colony. In a flow, one interesting question is which part of biofilms, upstream or downstream, is benefited more from quorum sensing regulation.
In Vaughan et al. (2010), the author suggests that biofilms upstream can induce downstream biofilms to quorum sense in the flow if they are close enough spatially without considering the heterogeneity of biofilms. Here we conduct a numerical simulation with a distribution of biofilm colonies in the flow channel. Our 3D numerical simulations agrees with this prediction. In details, in Fig. 9, it confirms (or qualitatively agreeable) with the findings in Vaughan et al. (2010) that quorum sensing takes place at downstream first, for instance quorum sensing induction takes place at the last four colonies instead of the first one located upstream. It also shows that the downstream region tends to have a higher concentration of QS molecules than at the location upstream in Fig. 9g, which we believe is an amplification of the convection and diffusion of QS molecules.
(Color figure online) Quorum sensing induction under the hydrodynamic stress with a weak nutrient supply. This figure shows quorum sensing induction in a flow cell with an inlet flow velocity. In this simulation, \(c_0=0.001\), representing a scenario with a weak nutrient supply. Initially some QS\(^{-}\) bacteria are attached to the substrate in the middle of the flow cell. a The initial profile of QS\(^-\) bacteria. b AHL concentration at \(x=0.5\), \(y=0.0625\) and \(t=25\). c–f 2D slices at \(z=0.5\) for AHL, \(\phi _{b1}\), \(\phi _{b2}\), as well as nutrient at time \(t=200\), respectively. When nutrient supply is weak in the entire flow cell, QS induction is stronger downstream than upstream. Consequently, there exist more QS\(^+\) bacteria downstream than upstream
However, this may not be always true, especially when nutrient supply is strong. A comparison with different nutrient supply rates is shown in Fig. 10. We observe that, at an early stage, bacteria downstream can be benefited since QS molecules are more diluted upstream (see Fig. 10c). QS induction is more effective downstream since there exist more QS\(^{+}\) bacteria there (see Fig. 10b). But when time passes by and the nutrient supply is kept at a sufficiently high level, it seems that quorum sensing induction benefits from a higher flow velocity since it sustains a sufficient nutrient supply that facilitates the growth of all bacteria as well the production of QS molecules. As a result, the overall production of QS molecules upstream increases such that both the concentration of QS molecules and the volume fraction of bacteria are higher upstream. In Fig. 10e, we see there are more QS\(^{+}\) bacteria upstream than downstream in this case. Figure 10h depicts the EPS production in the flow cell, which is also the result of the excessive accessibility to nutrient.
(Color figure online) Quorum sensing induction in a flow with a strong nutrient supply. This figure shows quorum sensing induction in a flow cell with inlet velocity \(v_0= 10y(1-y)\) and a strong nutrient supply (\(c_0=1.0\)). In this simulation, we use the same initial distribution of QS\(^-\) as used in Fig. 9 and characteristic time \(t_0=10^3\) s. a–c 2D slices of the volume fraction of QS\(^-\) bacteria, QS\(^+\) bacteria and the concentration of AHL at \(z=0.5\) and \(t=1.25\), respectively. d–i Depict the volume fraction of QS\(^-\) bacteria, QS\(^+\) bacteria, the concentration of AHL, the concentration of nutrient, the volume fraction of EPS, and the velocity field at \(z=0.5\) and \(t=25\), respectively. In this case, the strong nutrient supply dominates the growth of bacteria so that QS\(^+\) grows the fastest near the upstream location than the downstream one
(Color figure online) Flow effects on biofilm morphology and the distribution of QS molecules. This figure shows two grown biofilm colonies under shear in a flow cell, where the characteristic time \(t_0=1\) s. a The initial profile of the two biofilm colonies. b–e show the data at \(t = 31\). b The two colonies in the shear flow. c Distribution of QS molecules. d A 2D slice of the biofilm colonies at \(z=0.35\). e A 2D slice of the AHL distribution at \(z=0.35\)
(Color figure online) An anatomy of the flow-induced effect on biofilm morphology. a–c show the first biofilm colony in Fig. (11) at time \(t=0,18,42\), respectively. We denote the negative value by red and the positive one by blue in the rest of the subfigures. The data shown for d–h are the solutions at \(t=18\). d The second normal stress difference \(N_1=\phi _n(\tau _{yy} - \tau _{zz})\). e The first normal stress difference \(N_2=\phi _n(\tau _{xx} - \tau _{yy})\). f The shear stress in the xy plane \(\phi _n \tau _{xy}\). g The shear stress in the xz plane \(\phi _n \tau _{xz}\). h The shear stress in the yz plane \(\phi _n \tau _{yz}\). The first and second normal stress difference are of opposite signs in most part of the colony. The primary shear stress \(\phi _n \tau _{xy}\) is always positive while the two secondary shear stress components can take on either signs
3.3.3 Biofilm Morphology Under a Strong Shear Flow
In previous discussions, we focus on interactions between hydrodynamics and quorum sensing induction under the assumption that biofilms grow under weak flow fields. When the flow is strong, however, biofilms can undergo significant morphological changes and most of the biofilms can perhaps be flushed out of the flow cell. To better illustrate the hydrodynamic impact on biofilm structures and QS induction in strong flows, we conduct several numerical simulations where a grown biofilm colony is subject to a strong shear in a flow cell.
Here, we place two biofilm colonies in a \(4 \times 1 \times 1\) domain, representing a flow cell with inlet and outlet boundaries in the x direction. The initial biofilm profile is shown in Fig. 11a and the reference timescale is \(t_0=1\). From 11b, we notice that the biofilm undergoes a significant morphological change under the shear. The colonies are tilted under the shear. The one near the inlet boundary is about to detach from the bottom in Fig. 11d. In addition, a fast flow can dilute QS molecules as they are readily convected out of the flow cell shown in Fig. 11c, e.
The advantage of our full 3D hydrodynamic model, compared with those simplified models (without considering hydrodynamics) , is its capability to resolve the hydrodynamic detail as well as significant rheological functions such as the normal stress differences and shear stress components. Here, these stress components related to the simulation above are depicted in Fig. 12. In Fig. 12, we only show the sign of \(\phi _n \tau \), the effective stress for the biomass component, where we use red to represent negative and blue for positive values of the stress. It is seen through Fig. 12e that the first normal stress difference is positive at the top of the biofilm region while negative around the neck facing the inlet boundary, which means that the normal stress \(\tau _{xx}\) is larger than \(\tau _{yy}\) at the top of the biofilm, providing a drag force along the x direction. This is confirmed by Fig. 12c, where the top of the biofilm is stretched toward the x direction. In addition, through Fig. 12d, the second normal stress difference is positive at the neck facing the inlet boundary, where \(\tau _{yy}\) is larger than \(\tau _{zz}\) resulting in a stretching of the biofilm colony toward the y direction. This is supported by the fact that the neck becomes thinner due to this stretch, shown in Fig. 12c. In addition, the other three components of shear stress tensor are also depicted in Fig. 12f–g. On the plane \(y = a\) (\(0< a < 1\)), we see the shear stress \(\tau _{xy}\) is positive, which forces the biofilm to move toward the x direction (shown in the comparison of Fig. 12b, c.) The effect of \(\tau _{xz}\) and \(\tau _{yz}\) can also be observed on a z-plane, i.e., \( z = a\) (\(0< a < 1\)), shown in Fig. 12g, where \(\tau _{xz}\) provides a shear stress toward the x direction on the top of the biofilm mushroom-shaped colony and \(\tau _{yz}\) provides a shear stress toward the y direction on the top of the mushroom.
It is known that QS will be repressed under strong hydrodynamic flow as AHL will be advected (Kim et al. 2016). Here, in our model, we carried out a qualitative study by tuning the material parameter of biofilms, i.e., the viscosity of biomass. Given the same initial condition and parameters as shown in Fig. 11, we compare the volume-averaged AHL concentration by changing the biomass viscosity. Numerical results are shown in Fig. 13. As we observe, at the beginning, the volume-averaged concentration of QS molecules in the domain does not change, as they are still in the flow cell even though they may be flushed out. As time goes by, under same hydrodynamic conditions, it turns out that biofilms with a smaller viscosity (softer) can better trap QS molecules inside than biofilms with a larger viscosity (harder). This can be explained since biofilms with lager viscosity resists deformation such that the AHL would be flush away. But biofilms with a smaller viscosity tends to deform more, such that, instead of being flushed away into solvent and out of flow cell, more AHL would be trapped in the biofilm. We remark a more realistic model accounting EPS viscoelasticity is needed to better understand biofilm deformation since in such short timescale biofilms behave like viscoelastic materials.
(Color figure online) Effect of biofilm viscosity on QS molecules accumulation. This figure shows lower biofilm viscosity would help to trap AHL in biofilms in short time period. Here A, B, C represent the case: \(\eta _p=\eta _b=10^2\); \(\eta _p=\eta _b=10\);\(\eta _p=\eta _b=1\), respectively. Here the Y axis is the total volume-averaged concentration of QS molecules in flow cell, i.e., \(\frac{1}{|\Omega |} \int _{\Omega } A \hbox {d}\mathbf {x}\)
4 Conclusion
In this paper, a multiphasic hydrodynamic model for biofilms is developed to study biofilm formation and development regulated by quorum sensing, extending our previous 3D biofilm models (Zhang et al. 2008a). By identifying appropriate bacterial types based on their function in quorum sensing in the biofilm and proposing the corresponding reactive kinetics, we are able to describe several bulk phenomena related to quorum sensing induction using the new model. We then develop a numerical scheme to solve the governing system of equations in the model and implement it in 3D space and time on GPUs. With the numerical solver, we are able to simulate and predict heterogeneous spatial–temporal structures of a biofilm in terms of its volume fractions for each bacterial component, EPS and solvent. The solver is used to study a set of selected mechanisms in quorum-sensing-regulated biofilm development.
Numerical simulations indicate that QS-regulated EPS production competes for both physical space and nutrient since a higher EPS production rate can expand biofilm colonies spatially without reaching a higher local density. In addition, numerical simulations confirm the hypothesis that QS-regulated EPS production promotes biofilm development as well as protects biofilms from the environment stress, such as antimicrobial treatment and hydrodynamical stress. However, quorum sensing regulation is not universally beneficial to biofilm formation and development in all situations evidenced by what we have shown that an invasion strain of bacteria with a higher production rate can outgrow QS-regulated bacteria, where QS induction retards the proliferation of bacteria and thereby the development of biofilms in a short timescale. Our 3D simulations show that QS down-regulated cells tend to grow at the surface of the biofilm while QS up-regulated ones tend to grow in the bulk.
By simulating biofilms in a flow cell, hydrodynamical effects on quorum sensing are investigated in detail. The model captures the competition between the dynamic diluting effect and the nutrient supply associated with QS induction well. Our simulations predict that downstream bacteria can be benefited from higher inlet flow speed to initiate QS induction earlier. However, the model also shows that this only takes place in a short timescale, when nutrient supply is weak. When nutrient supply is strong, it is a competition between the flow-induced dilution of QS molecules and the nutrient supply for the production of QS molecules that ultimately determines which parts of biofilms in a flow cell would benefit more.
This 3D hydrodynamic model provides a powerful simulation and predicative tool to allow us to investigate quorum-sensing-regulated biofilm development, some factors that correlate with quorum sensing and their interaction with hydrodynamics. In addition to the results that we numerically confirmed, we hope new results predicted here can be verified in experiments in the future. In our subsequent research, other quorum-sensing-regulated factors such as virulence, growth factors, and resistance to antimicrobial agents will be investigated.
References
Abraham W (2006) Controlling biofilms of gram-positive pathogenic bacteria. Curr Med Chem 13(13):1509–1524
Almeida A, Amado I, Reynolds J, Berges J, Lythe G, Molina-Paris C, Freitas AA (2012) Quorum sensing in cd4+ t cell homeostasis: a hypothesis and a model. Front Microbiol 3:125
Alpkvist E, Picioreanu C, van Loosdrecht M, Heyden A (2006) Three-dimensional biofilm model with individual cells and continuum eps matrix. Biotechnol Bioeng 94(5):961–979
Barbarossa MV, Kuttler C, Fekete A, Rothballer M (2010) A delay model for quorum sensing of pseudomonas putida. BioSystems 102:148–156
Bayles KW (2007) The biological role of death and lysis in biofilm development. Nat Rev Microbiol 5(9):721–726
Bell N, Garland M (2012) Cusp: generic parallel algorithms for sparse matrix and graph computations (preprint)
Cahn JW, Hilliard JE (1958) Free energy of a nonuniform system. I. Interfatial free energy. J Chem Phys 28(2):258–267
Cahn JW (1959) Free energy of a nonuniform system. II. Thermodynamic basis. J Chem Phys 30(5):1121
Cárcamo-Oyarce G, Lumjiaktase P, Kümmerli R, Eberl L (2015) Quorum sensing triggers the stochastic escape of individual cells from pseudomonas putida biofilms. Nat Commun 6:5945
Chen LQ (2002) Phase-field models for microstructure evolution. Ann Rev Mater Res 32(1):113–140
Childs H, Brugger E, Whitlock B, Meredith J, Ahern S, Pugmire D, Biagas K, Miller M, Harrison C, Weber GH, Krishnan H, Fogal T, Sanderson A, Garth C, Bethel EW, Camp D, Rübel O, Durant M, Favre JM, Navrátil P (2012) Visit: an end-user tool for visualizing and analyzing very large data. High performance visualization-enabling extreme-scale scientific. Insight, pp 357–372
Chopp DL, Kirisits MJ, Moran B, Parsek MR (2002) A mathematical model of quorum sensing in a growing bacterial biofilm. J Ind Microbiol Biotechnol 29:339–346
Chopp DL, Kirisits MJ, Moran B, Parsek MR (2003) The dependence of quorum sensing on the depth of a growing biofilm. Bull Math Biol 65:1053–1079
Choudhary S, Schmidt-Dannert C (2010) Applications of quorum sensing in biotechnology. Appl Microbiol Biotechnol 86:1267–1279
Cogan NG (2006) Effects of persister formation on bacterial response to dosing. J Theor Biol 238:694–703
Cogan NG (2011) Computational exploration of disinfection of bacterial biofilms in partially blocked channels. Int J Numer Methods Biomed Eng 27:1982–1995
Cogan NG, Keener JP (2004) The role of the biofilm matrix in structural development. Math Med Biol 21(2):147–166
Cogan NG, Keener JP (2005) Channel formation in gels. SIAM J Appl Math 65(6):1839–1854
Cogan NG, Brown J, Darres K, Petty K (2012) Optimal control strategies for disinfection of bacterial populations with persister and susceptible dynamics. Antimicrob Agents Chemother 56(9):4816–4826
Dilanji G, Langebrake J, Leenheer P, Hagen S (2012) Quorum activation at a distance: spatiotemporal patterns of gene regulation from diffusion of an autoinducer signal. J Am Chem Soc 134:5618–5626
Donlan RM (2002) Biofilms microbial life on surfaces. Emerg Infect Dis 8(9):881–890
Emerenini B, Hense B, Kuttler C, Eberl H (2015) A mathematical model of quorum sensing induced biofilm detachment. PLOS ONE 10(7):e0132385
Emerenini B, Sonner S, Eberl H (2017) Mathematical analysis of a quorum sensing induced biofilm dispersal model and numerical simulation of hollowing effects. Math Biosci Eng 14(3):625–653
Fauvart M, Phillips P, Bachaspatimayum D, Verstraeten N, Fransaer J, Michiels J, Vermant J (2012) Surface tension gradient control of bacterial swarming in colonies of pseudomonas aeruginosa. Soft Matter 8:70–76
Fekete A, Kuttler C, Rothballer M, Hense B, Fischer D, Buddrus-Schiemann K, Lucio M, Muller J, Schmitt-Kopplin P (2010) Dynamic regulation of \(n\)-acyl-homoserine lactone production and degradation in pseudomonas putidalsof. FEMS Microbiol Ecol 72:22–34
Fozard JA, Lees M, King JR, Logan BS (2012) Inhibition of quorum sensing in a computational biofilm simulation. BioSystems 109:105–114
Frederick MR, Kuttler C, Hense BA, Muller J, Eberl HJ (2010) A mathematical model of quorum sensing in patchy biofilm communities with slow background flow. Can Appl Math Q 18(3):267–298
Frederick MR, Kuttler C, Hense BA, Eberl HJ (2011) A mathematical model of quorum sensing regulated eps production in biofilm communities. Theor Biol Med Model 8(1):8
Garcia-Aljaro C, Melado-Rovira S, Milton DL, Blanch AR (2012) Quorum-sensing regulates biofilm formation in virio schophthalmi. BMC Microbiol 12:287
Guttenplan SB, Kearns DB (2013) Regulation of flagellar motility during biofilm formation. FEMS Microbiol Rev 37(6):849–871
Hammer BK, Bassler BL (2003) Quorum sensing controls biofilm formation in vibrio chelerae. Mol Microbiol 50(1):101–114
Heithoff DM, Mahan MJ (2004) Vibrio cholerae biofilms: stuck between a rock and a hard place. J Bacteriol 186(15):4835–4837
Hentzer M, Givskov M (2003) Pharmacological inhibition of quorum sensing for the treatment of chronic bacterial infections. J Clin Investig 112(9):1300–1307
Hinson R, Kocher W (1996) Model for effective diffusivities in aerobic biofilms. J Environ Eng 122(11):1023–1030
Jabbari S, King JR, Williams P (2012a) Cross-strain quorum sensing inhibition by staphylococcus aureus. Part 1: a spatially homogeneous model. Bull Math Biol 74(6):1292–3251
Jabbari S, King JR, Williams P (2012b) Cross-strain quorum sensing inhibition by staphylococcus aureus. Part 2: a spatially inhomogeneous model. Bull Math Biol 74(6):1326–1353
Keller L, Surette MG (2006) Communication in bacteria: an ecological and evolutionary perspective. Nat Rev Microbiol 4:249–258
Kim MK, Ingremeau F, Zhao A, Basseler BL, Stone HA (2016) Local and global consequences of flow on bacterial quorum sensing. Nat Microbiol 1:15005
Kirisits MJ, Margolis JJ, Purevdorj-Gage BL, Vaughan B, Chopp DL, Stoodley P, Parsek MR (2007) Influence of the hydrodynamic environment on quorum sensing in pseudomonas aeruginosa biofilms. J Bacteriol 189:8357–8360
Koerber AJ, King JR, Ward JP (2002) A mathematical model of partial-thickness burn-wound infection by pseudomonas aeruginosa: quorum sensing and the build-up to invasion. Bull Math Biol 64(239–259):239–259
Kommedal R, Bakke R, Dockery J, Stoodley P (2001) Modeling production of extracellular polymeric substances in a pseudomonas aeruginosa chemostat culture. Water Sci Technol 43(6):129–134
Langebrake J, Dilanji G, Hagen S, Leenheer P (2014) Traveling waves in response to a diffusing quorum sensing signal in spatially-extended bacterial colonies. J Theor Biol 363:53–61
Lewis K (2010) Persister cells. Ann Rev Microbiol 64(1):357–372
Lindley B, Wang Q, Zhang T (2011) A multicomponent model for biofilm-drug interaction. Discrete Contin Dyn Syst Ser B 15:417–456
Marketon MM, Glenn SA, Eberhard A, Gonzalez JE (2003) Quorum sensing controls exopolysaccharide production in sinorhizobium melioti. J Bacteriol 185(1):325–331
Matur MG, Muller J, Kuttler C, Hense RA (2015) An approximative approach for single cell spatial modeling of quorum sensing. J Comput Biol 22(3):227–235
Miller M, Bassler B (2001) Quorum sensing in bacteria. Ann Rev Microbiol 55:165–199
Nadell CD, Xavier JB, Levin SA, Foster KR (2008) The evolution of quorum sensing in bacterial biofilms. PLoS Biol 6(1):0171–0179
Nealson K, Platt T, Hastings JW (1970) The cellular control of the synthesis and activity of the bacterial luminescent system. J Bacteriol 104(1):313–322
Parsek MR, Tolker-Nielsen T (2008) Pattern formation in pseudomonas aeruginosa biofilms. Curr Opin Biotechnol 11:560–566
Perez-Velazquez J, Quinones B, Hense B, Kuttler C (2015) A mathematical model to investigate quorum sensing regulation and its heterogeneity in pseudomonas syringae on leaves. Ecol Complex 21:128–141
Picioreanu C, van Loosdrecht MCM, Heijnen JJ (1998) Mathematical modeling of biofilm structure with a hybrid differential discrete cellular automaton approach. Biotechnol Bioeng 58(1):101–116
Popat R, Crusz S, Messina M, Williams P, West S, Diggle SP (2012) Quorum-sensing and cheating in bacterial biofilms. Proc R Soc B Biol Sci 279(1748):4765–4771
Quinones B, Pujol CJ, Lindow SE (2004) Regulation of ahl production and its contribution to epiphytic fitness in pseudomonas syringae. MPMI 17(5):521–531
Quinones B, Dulla G, Lindow SE (2005) Quorum sensing regulates exopolysaccharide production, motility, and virulence in pseudomonas syringae. Mol Plant Microbe Interact 18(7):682–693
Roberts ME, Stewart PS (2005) Modeling protection from antimicrobial agents in biofilms through the formation of persister cells. Microbiology 151:75–80
Sakuragi Y, Kolter R (2007) Quorum-sensing regulation of the biofilm matrix genes (pel) of pseudomonas aeruginosa. J Bacteriol 189(14):5383–5386
Sanclement JA, Webster P, Thomas J, Ramadan HH (2005) Bacterial biofilms in surgical specimens of patients with crhonic rhinosinusitis. Laryngoscope 115(4):578–582
Shen Y, Stojic S, Haapasalo M (2011) Antimicrobial efficacy of chlorhexidine against bacteria in biofilms at different stages of development. J Endod 37(5):657–661
Shen Y, Zhao J, de la Fuente-Núñez C, Wang Z, Hancock RE, Roberts CR, Ma J, Li J, Haapasalo M, Wang Q (2016) Experimental and theoretical investigation of multispecies oral biofilm resistance to chlorhexidine treatment. Sci Rep 6:27537. doi:10.1038/srep27537
Solano C, Echeverz M, Lasa I (2014) Biofilm dispersion and quorum sensing. Curr Opin Microbiol 18:96–104
Sonner S, Efendiev MA, Eberl HJ (2011) On the well-posedness of a mathematical model of quorum-sensing in patchy biofilm communities. Math Methods Appl Sci 34:1667–1684
Stewart PS (1996) Theoretical aspects of antibiotic diffusion into microbial biofilms. Antimicrob Agents Chemother 40(11):2517–2522
Stewart PS (2003) Diffusion in biofilms. J Bacteriol 185(5):1485–1491
Teodosio JS, Simões M, Melo LF, Mergulhao FJ (2011) Flow cell hydrodynamics and their effects on E. coli biofilm formation under different nutrient conditions and turbulent flow. Biofouling 27(1):1–11
Thompson JA, Oliveira RA, Djukovic A, Ubeda C, Xavier KB (2015) Manipulation of the quorum sensing signal ai-2 affects the antibiotic-treated gut microbiota. Cell Rep 10:1–11
Uecke H, Müller J, Hense B (2014) Individual-based model for quorum sensing with background flow. Bull Math Biol 76(7):1727–1746
Vaughan B, Smith B, Chopp D (2010) The influence of fluid flow on modeling quorum sensing in bacterial biofilms. Bull Math Biol 72(5):1143–1165
Vuong C, Gerke C, Somerville G, Fischer E, Otto M (2003) Quorum-sensing control of biofilm factors in staphylococcus epidermidis. J Infect Dis 188(5):706–718
Ward JP, King JR, Koerber A, Williams P, Croft J, Sockett R (2001) Mathematical modelling of quorum sensing in bacteria. Math Med Biol 18(3):263–292
Waters CM, Basseler BL (2005) Quorum sensing: cell-to-cell communication in bacteria. Ann Rev Cell Dev Biol 21(1):319–346
Whitehead NA, Barnard A, Slater H, Simpson N, Salmond G (2001) Quorum sensing in gram-negative bacteria. FEMS Microbiol Rev 25:365–404
Xavier JB, Foster KR (2007) Cooperation and conflict in microbial biofilms. PNAS 104(3):876–881
Zhang T, Cogan NG, Wang Q (2008a) Phase-field models for biofilms i. Theory and simulations. SIAM J Appl Math 69:641–669
Zhang T, Cogan NG, Wang Q (2008b) Phase-field models for biofilms ii. 2-d numerical simulations of biofilm-flow interaction. Commun Comput Phys 4(1):72–101
Zhao J, Seeluangsawat P, Wang Q (2016a) Modeling antimicrobial tolerance and treatment of heterogeneous biofilms. Math Biosci 282:1–15
Zhao J, Shen Y, Happasalo M, Wang ZJ, Wang Q (2016b) A 3d numerical study of antimicrobial persistence in heterogeneous multi-species biofilms. J Theor Biol 392:83–98
Zhao J, Wang Q, Yang X (2016c) Numerical approximations to a new phase field model for two phase flows of complex fluids. Comput Methods Appl Mech Eng 310:77–97
Zhao J, Yang X, Shen J, Wang Q (2016d) A decoupled energy stable scheme for a hydrodynamic phase field model of mixtures of nematic liquid crystals and viscous fluids. J Comput Phys 305:539–556
Zhao J, Yang X, Wang Q (2016e) Energy stable numerical schemes for a hydrodynamic model of nematic liquid crystals. SIAM J Sci Comput 38(5):3264–3290
Zhao J, Li H, Wang Q, Yang X (2017) Decoupled energy stable schemes for a phase field model of three-phase incompressible viscous fluid flow. J Sci Comput 70(3):1367–1389
Acknowledgements
Qi Wang is partially supported by NIH and NSF through awards DMS-1200487, DMS-1517347, R01GM078994-05A1, and NSFC awards 11571032 and 91630207. Jia Zhao is partially supported by an ASPIRE grant from the Office of the Vice President for Research at the University of South Carolina.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, J., Wang, Q. Three-Dimensional Numerical Simulations of Biofilm Dynamics with Quorum Sensing in a Flow Cell. Bull Math Biol 79, 884–919 (2017). https://doi.org/10.1007/s11538-017-0259-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-017-0259-4