Abstract
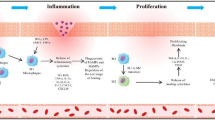
Wound healing in diabetes is a complex process, characterised by a chronic inflammation phase. The exact mechanism by which this occurs is not fully understood, and whilst several treatments for healing diabetic wounds exist, very little research has been conducted towards the causes of the extended inflammation phase. We describe a mathematical model which offers a possible explanation for diabetic wound healing in terms of the distribution of macrophage phenotypes being altered in the diabetic patient compared to normal wound repair. As a consequence of this, we put forward a suggestion for treatment based on rectifying the macrophage phenotype imbalance.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Baumgartner-Parzer, S.M., 1995. High-glucose triggered apoptosis in cultured endothelial cells. Diabetes 4411, 1323–1327.
Bellingan, G.J., 1996. In vivo fate of the inflammatory macrophage during resolution of inflammation: Inflammatory macrophages do not die locally, but emigrate to the draining lymph nodes. J. Immunol. 1576, 2577–2585.
Black, E., 2003. Decrease of collagen deposition in wound repair in type 1 diabetes independent of glycemic control. Arch. Surg. 1381, 34–40.
Bulgrin, J.P., 1995. Nitric oxide synthesis is suppressed in steroid-impaired and diabetic wounds. Wounds 7, 48–57.
Cobbold, C.A., Sherratt, J.A., 2000. Mathematical modelling of nitric oxide activity in wound healing can explain keloid and hypertrophic scarring. J. Theor. Biol. 2042, 257–288.
Darby, I.A., 1997. Apoptosis is increased in a model of diabetes-impaired wound healing in genetically diabetic mice. Int. J. Biochem. Cell Biol. 291, 191–200.
Greenhalgh, D., 2003. Wound healing and diabetes mellitus. Clin. Plast. Surg. 301, 37–45.
Hehenberger, K., 1998. Inhibited proliferation of fibroblasts derived from chronic diabetic wounds and normal fibroblasts treated with high glucose is associated with increased formation of l-lactate. Wound Repair Regen. 62, 135–141.
Lerman, O., 2003. Cellular dysfunction in the diabetic fibroblast: Impairment in migration, vascular endothelial growth factor production, and response to hypoxia. Am. J. Pathol. 1621, 303–312.
Loots, M.A., 1998. Differences in cellular infiltrate and extracellular matrix of chronic diabetic and venous ulcers versus acute wounds. J. Invest. Dermatol. 1115, 850–857.
Lorenzi, M., Cagliero, E., Toledo, S., 1985. Glucose toxicity for human endothelial cells in culture: Delayed replication, disturbed cell cycle, and accelerated death. Diabetes 347, 621–627.
Miyasaki, K., 2002. Chap. 5—Monocytes. In: Basic Immunology Course Notes (Web Document), UCLA.
Mulder, G.D., 1998. Clinician's Pocket Guide to Chronic Wound Repair. In: Wound Care Communications Network, 4th edn. Springhouse Corp., Springhouse, PA, pp. 58–67.
Murray, J.D., 2002. Mathematical Biology. Vol. 1: An Introduction, 3rd edn. In: Interdiscliplinary Applied Mathematics Series. Springer-Verlag, Berlin, pp. 7–10, 40–41.
Pettet, G., 1996. On the role of angiogenesis in wound healing. Proc. R. Soc. Lond. Part B 2631376, 1487–1493.
Riches, D.W.H., 1996. Macrophage involvement in wound repair, modelling and fibrosis. In: Clark, R.A.F. (Ed.), The Molecular and Cellular Biology of Wound Repair, 2nd edn., pp. 95–142.
Robson, M.C., Steed, D.L., Franz, M.G., 2001. Wound Healing: Biologic features and approaches to maximize healing trajectories. Curr. Probl. Surg. 382, 65–140.
Sank, A., 1994. Human endothelial cells are defective in diabetic vascular research. J. Surg. Res. 576, 647–653.
Schaeffer, M.R., 1996. Nitric oxide regulates wound healing. J. Surg. Res. 631, 237–240.
Schaeffer, M.R., 1997. Diabetes-impaired healing and reduced wound nitric oxide synthesis: A possible pathophysiologic correlation. Surgery 1215, 513–519.
Shukla, A., 1998. Differential expression of proteins during healing of cutaneous wounds in normal and chronic models. Biochem. Biophys. Res. Commun. 2442, 434–439.
Singer, A.J., Clark, R.A.F., 1999. Cutaneous wound healing. N. Engl. J. Med. 34110, 738–746.
Wahl, S.M., 1987. Transforming growth factor beta induces monocyte chemotaxis and growth factor production. Proc. Natl. Acad. Sci. U.S.A. 8416, 5788–5792.
Wetzler, C., 2000. Large and sustained induction of chemokines during impaired wound healing in the genetically diabetic mouse: Prolonged persistence of neutrophils and macrophages during the late phase of repair. J. Invest. Dermatol. 1152, 245–253.
Williams, G., Pickup, J.C., 2001. Handbook of Diabetes, 2nd edn. Blackwell Science, Oxford, 69p.
Zykova, S.N., 2000. Altered cytokine and nitric oxide secretion in vitro by macrophages from diabetic type II-like db/db mice. Diabetes 499, 1451–1458.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Waugh, H.V., Sherratt, J.A. Macrophage Dynamics in Diabetic Wound Dealing. Bltn. Mathcal. Biology 68, 197–207 (2006). https://doi.org/10.1007/s11538-005-9022-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-005-9022-3