Abstract
Background
The CHAARTED and LATITUDE trials demonstrated improved outcomes with docetaxel or abiraterone plus androgen deprivation therapy in metastatic hormone sensitive prostate cancer (mHSPC) using two different prognostic scores.
Objective
The aim of our study was to assess the concordance between the two scores and if these retained their prognostic value exclusively in de novo mHSPC.
Patients and Methods
De novo mHSPC patients referring to our institution were retrospectively stratified according to the CHAARTED and LATITUDE classifications: high volume/high risk (HV/HR), low-volume/low-risk (LV/LR), and HVorHR (HV/LR and LV/HR). The Kaplan-Meier method and Cox proportional-hazard models were used to estimate hazard ratios for overall survival.
Results
The study population included 106 patients. Concordance between the CHAARTED and LATITUDE classifications was observed in 86.8% of cases (65.1% HV/HR, 21.7% LV/LR), while 13.2% of patients fulfill the criteria of only one of the two classifications (HVorHR). When analyzed independently, the CHAARTED and LATITUDE classifications maintained their prognostic value (mOS 28.2 months in HV versus 60.9 months in LV, p = 0.006; 28.2 months in HR versus 40.6 months in LR, p = 0.017). The LR/LV population showed significantly longer mOS compared to the HR/HV group (72.6 months versus 26.3 months; p = 0.005), and to HVorHR patients (35.1 months; p = 0.003). No difference in OS was observed between HV/HR and HVorHR patients. ECOG PS ≥ 1 and patient age improved the prognostic value of the two classifications with multivariate analysis.
Conclusions
Our study showed a lack of complete concordance between the CHAARTED and LATITUDE classifications. The analysis confirmed the role of these prognostic scores to stratify de novo mHSPC patients in clinical practice.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
A lack of complete concordance between the CHARTED and LATITUDE prognostic classifications was observed in a cohort of de novo mHSPC patients | |
The CHAARTED and LATITUDE classifications maintained their prognostic value in de novo mHSPC | |
ECOG PS ≥ 1 and patient age improved the prognostic value of the two prognostic classifications |
ᅟ
1 Introduction
Prostate cancer (PC) is the most frequently diagnosed solid tumour among men and the third leading cause of cancer-related deaths [1, 2]. In Europe, today only 4% of newly diagnosed patients present with metastatic disease [3]. De novo hormone sensitive metastatic PC (mHSPC) represents a biologically aggressive condition characterized by poor prognosis [4] and a 5-year OS rate of about 35% [5, 6]. Given the strict growth dependence of prostate cancer cells on androgens, androgen deprivation therapy (ADT) has been considered the standard of care for newly diagnosed mHSPC since the 1940s [7, 8]. Even though the rate of response to ADT reaches 90%, the majority of patients progress to castration-resistant prostate cancer (CRPC) in less than 2 years. The lack of a substantial improvement in survival over the last 25 years has led to the development of novel therapeutic strategies for this subset of PC patients [4]. Recently, four randomized controlled trials have redefined the first-line treatment approach of mHSPC, demonstrating to significantly prolonged OS with the addition of cytotoxic chemotherapy (docetaxel) or the second-generation endocrine agent abiraterone acetate to ADT [9,10,11,12]. The CHAARTED study revealed a statistically significant and clinically relevant OS benefit with the addition of docetaxel to ADT compared to ADT alone in mHSPC patients, 75% of which had de novo mHSPC [9]. Of note, a greater benefit was observed in patients with metastatic high-volume disease, while the OS improvement did not reach statistical significance in the subpopulation with low-volume disease [9]. Data from the STAMPEDE study confirmed this benefit, with a median OS improvement of 10 months for the subset of patients with metastatic disease at diagnosis [12]. The French GETUG-AFU 15 trial failed to demonstrate a survival advantage with docetaxel; however, reanalysis showed a trend towards an OS benefit for patients with high-volume disease [13]. In the LATITUDE trial, similar result emerged with the early use of abiraterone acetate and prednisone concurrent with ADT both in men with newly diagnosed high-risk mHSPC and mHSPC patients not selected for prognostic factors [10, 11].
The availability of several therapeutic options raises the question about adequate treatment selection for de novo mHSPC [14]. Besides the profoundly different toxicity profiles of docetaxel and abiraterone, a reliable prognostic stratification of patients is needed for guiding the decision-making process. The prognostic classifications used in the LATITUDE and CHAARTED trials are similar but not entirely corresponding, making it difficult to compare the two study populations. Therefore, concerns about the ideal prognostic classification to be used in daily clinical practice represents an unsolved issue.
The objective of our analysis was to assess the concordance between the two prognostic scores from the CHAARTED and LATITUDE trials. In particular, we evaluated how many patients with high-risk (HR) - or low-risk (LR) - according to the LATITUDE trial could also be considered as high-volume (HV) - or low-volume (LV) - based on the CHAARTED study, and vice versa. Moreover, we tested if these scores retained their prognostic value in a population with exclusively de novo metastatic PC. The ultimate goal of an adequate prognostic patient stratification will be to guide clinicians toward the most appropriate treatment selection.
2 Patients and Methods
2.1 Patients
Consecutive prostate cancer patients referred to the oncology department of the Azienda Ospedaliera Universitaria Integrata of Verona were screened to find those with mHSPC at diagnosis. The study was reviewed by the relevant IRC/CE and all patients signed an informed consent form before the start of therapy.
Patients were excluded if baseline characteristics such as histological diagnosis, extension of disease, or type of treatment were not available. Baseline characteristics required for the inclusion were: diagnosis of prostate cancer with evidence of metastatic spread at bone scan, computed tomography (CT), magnetic resonance imaging (MRI), or choline PET/CT at diagnosis, availability of ECOG performance status, the beginning of the ADT and at least one follow up visit after the initial diagnosis.
2.2 Patient Stratification
The prognosis at baseline was evaluated for each patient using the CHAARTED and LATITUDE criteria [8, 9]. The CHAARTED criteria classified patients based on the disease volume; high-volume was defined by the presence of visceral metastases or ≥ 4 bone lesions with ≥1 beyond the vertebral bodies and pelvis, while low-volume included patients that did not meet the above criteria. The LATITUDE classified high-risk disease based on the presence of at least two criteria among presence of visceral metastases, number of bone lesions ≥3, or Gleason score ≥ 8. For the purpose of this analysis, patients who did not fulfill the criteria for high-risk were defined as low-risk.
Based on the above classifications, patients were classified in three groups: high-volume/high-risk (HV/HR), low-volume/low-risk (LV/LR), and the third group that included patients with high-volume/low-risk or low-volume/high-risk (HVorHR).
The primary objective of our study was to assess the concordance between these two prognostic scores. Secondary objectives were to validate if these classification systems retained their prognostic value exclusively in the subset of de novo mHSPC patients, to report survival by risk groups, and to test if other baseline characteristic not included in the original classifications may improve the current prognostic models.
2.3 Statistics
Descriptive statistics were used to characterize patients at baseline, defined as the date of histological diagnosis. OS was evaluated from diagnosis to death or last follow-up, whichever occurred first. Survival was estimated by the Kaplan-Meier method, and compared across groups using the log-rank test. Cox proportional-hazard models, stratified according to baseline characteristics, and used to estimate hazard ratios for OS.
All the variables were considered statistically significant if p < 0.05. The PASW software (Predictive Analytics SoftWare; v 21; IBM SPSS) was used for analysis.
3 Results
3.1 Patients
A total of 559 prostate cancer patients referred to our department from January 2007 to October 20th 2017 and among these, 126 fulfilled the criteria to be included in the analysis. Baseline characteristics of these patients are reported in Table 1. All patients received ADT as first-line therapy, while only 10.3% (13/126) of patients was treated with ADT plus docetaxel.
3.2 Comparison Between Prognostic Classifications
Based on the CHAARTED classification, 126 patients were evaluable; 30 patients (23.8%) were classified as LV and 96 (76.2%) as HV of disease. When the LATITUDE classification was used, 106 patients were evaluable; 33 patients (31.1%) were classified as LR and 73 (68.9%) as HR of disease.
When the two classifications were matched, 69 patients (65.1%) had HV/HR, 23 (21.7%) had LV/LR, and the remaining 14 (13.2%) had HV or HR. Within the last group, ten patients (9.4%) had HV/LR disease and four (3.8%) had LV/HR disease (Table 2). Of note, the feature that characterized the HV/LR patients was a GS <8; while among the four LV/HR patients, one had five bone vertebral lesions, and three had only three bone lesions.
Therefore, the two classifications were concordant in 86.8% of cases, and discordant in the remaining 13.2%.
3.3 Overall Survival
At the date of analysis, after a median follow up of 35.6 months, 70 patients (66.0%) had died. The median OS was 35.1 months (95% CI, 27.8 – 42.4 months) in the overall cohort. When the CHAARTED classification was used, the median OS was 60.9 months (95% CI, 27.1 – 94.7 months) in the LV and 28.2 months (95% CI, 21.0 – 35.4 months) in the HV group (p = 0.006).
When the LATITUDE classification was used, the median OS was 40.6 months (95% CI, 24.7 – 56.4 months) in the LR and 28.2 months (95% CI, 22.8 – 33.6 months) in the HR group (p = 0.017).
The median OS in the HV/HR group was 26.3 months (95% CI, 20.0 – 32.7 months), 72.6 months (95% CI, 25.5 – 119.6 months) in the LV/LR group, and 35.1 months (95% CI, 31.4 – 38.8 months) in the HV or HR group. Significant differences were found when the LV/LR group was compared to the HV/HR group (p = 0.005) or to the HV or HR group (p = 0.003), while no significant difference was found when the HV or HR group was compared with the HV/HR group (p = 0.7).
3.4 Univariate and Multivariate Analysis
ECOG performance status ≥1, baseline PSA, first-line treatment with docetaxel plus ADT, and the patients’ age were tested for their predictive value at univariate analysis. The prognostic variables associated with poor OS were ECOG PS ≥ 1 and patient age (Table 3).
The prognostic variables associated with poor OS according to CHAARTED or LATITUDE classifications at multivariate analysis were ECOG PS and patient age (Table 3).
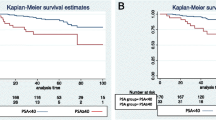
OS for the CHAARTED and LATITUDE classifications, adjusted for age and ECOG are reported in Fig. 1A and B.
4 Discussion
De novo metastatic prostate cancer represents a small but particularly interesting subset of PC. Compared to primary progressive metastatic patients, the worse prognosis of newly diagnosed mHSPC reflects a peculiar aggressiveness of this condition [15, 16]. However, the cohort of men presenting with metastatic PC at the time of diagnosis comprises a highly heterogeneous disease that differs in terms of clinical presentation, tumour biology, and prognosis [17, 18]. In fact, besides extremely aggressive disease (symptomatic patients with undifferentiated tumors, visceral metastases, extensive bone involvement), more indolent forms exist (asymptomatic, oligometastatic patients).
In the last few years, management paradigms for mHSPC have been developing. Chemotherapy with docetaxel and anti-hormonal therapy with abiraterone acetate, both in combination with ADT represent two possible therapeutic options for some mHSPC patients, challenging the approach to newly diagnosed mPC. In particular, the CHAARTED trial demonstrated the greatest benefit with concurrent ADT plus docetaxel in the subgroup of men with high-volume disease – defined as the presence of visceral metastases and/or ≥ 4 bone lesions with ≥1 outside the axial skeleton –, showing a 17-month OS improvement (49.2 versus 32.2 months; HR 0.60, p = 0.0006) [9]. The LATITUDE study showed a 38% reduction in the risk of death in hormone-naïve high-risk metastatic PC, defined if at least two of the following factors were met: Gleason score ≥ 8, presence of ≥3 bone lesions or visceral metastasis [10]. However, a direct comparison between these two strategies is still lacking. Moreover, a validated prognostic score that helps clinicians with an adequate treatment selection for de novo mHSPC has to be identified. The CHAARTED and LATITUDE trials used two different prognostic classifications, based on the presence of phenotypic features retrospectively associated with worse cancer-specific survival [19,20,21,22]. Both classifications identify the presence of visceral disease as a predictor of poor prognosis. Conversely, the CHAARTED trial acknowledged sites and number of bone metastases, whereas the LATITUDE classification stratified patients based on the number of skeletal metastases and Gleason score. When analyzed independently, the CHAARTED and LATITUDE risk classifications maintained their prognostic value in our cohort of de novo mHSPC, with a statistically significant difference in OS between HV and LV patients (mOS of 28.2 vs 60.9 months; p = 0.006), and between HR and LR patients (mOS of 28.2 vs 40.6 months; p = 0.017). The median survivals are comparable to those reported in the literature [23].
Moreover, despite the different parameters used, it is of considerable importance to understand if the two prognostic systems overlap, that is, if de novo mHSPC patients with high-risk (or low-risk) disease according to the LATITUDE trial can also be considered as high-volume (or low-volume) based on the CHAARTED study, and vice versa. Our analysis found the absence of a complete concordance between these two classifications in a cohort of 106 de novo mHSPC patients. We have therefore categorized our patients into three different subgroups: HV/HR, LV/LR, and HV or HR (which includes patients with HV/LR or LV/HR). In particular, about 13% of patients matched with only one of the two classifications (HV or HR), suggesting caution when adopting only one prognostic system for treatment selection. Therefore, albeit apparently small, a 13% lack of the concordance between the two risk scores is of utmost importance for its consequences in clinical practice. The potential risk is indeed to exclude patients who are considered unsuitable for docetaxel from being treated with abiraterone, or vice versa. Where a concordance between the CHAARTED and LATITUDE systems was observed (in about 87% of cases), two opposite clinical disease patterns can be delineated: patients at very good prognosis (LV/LR group) that displayed a mOS of 72.6 months, and patients at poor prognosis (HV/HR disease), whose median survival was more than halved (26.3 months). Of note, the prognosis of patients belonging to only one of the two risk classifications (HV or HR) was significantly shorter compared to the LR/LV population (p = 0.003), but did not differ from the HV/HR group (p = 0.7). However, the small number of HV or HR patients did not allow drawing definitive conclusions.
Prognostic stratification is a fundamental step in the management of cancer patients. It not only makes clinical trial populations more homogeneous (resulting in evidence reproducible for daily clinical practice), but also allows predicting patients’ life expectancy and thus leads clinicians towards a more conscious therapeutic planning.
A critical analysis of PC patients, by matching the CHAARTED and LATITUDE prognostic classifications, can help determine which patients are more likely to benefit from ADT alone or combined with early docetaxel or abiraterone. Although an OS benefit of both combinations compared to ADT monotherapy was demonstrated in the multi-group STAMPEDE trial in the overall mHSPC population regardless of prognostic factors [10, 11], the strength of the CHAARTED and LATITUDE studies concerns the better patient selection and characterization. Indeed, given the absence of a direct comparison between these treatment options, and lacking biomarkers with predictive value, to date the disease characteristic (albeit still incompletely defined) are the only factors to take into account for treatment selection. Therefore, from the results of our study we can argue that LV/LR patients, at very good prognosis, could benefit from ADT alone. The LR population indeed was not included in the LATITUDE trial; and conclusive data about the benefit from docetaxel in the LV patients are still lacking. The subgroup of HV/LR patients, not eligible for abiraterone, could be treated with chemotherapy given their poor prognosis. Analogously, LV/HR patients could receive abiraterone acetate, considering the unclear benefit of docetaxel for patients with low-volume disease. Concerns related to the treatment selection (chemotherapy or second-generation hormonal therapy) for the subgroup of HV/HR remain high. The poor prognosis of HV/HR patients requires a better understanding, and possibly additional stratification of this subset of de novo metastatic PC to improve treatment outcomes.
Our study confirms patient age as a negative prognostic factor in PC [24, 25]. The age and the ECOG performance status could be added to the CHAARTED and LATITUDE classifications as independent prognostic factors to better predict patients prognosis. However, although prognostic, the role of these two clinical factors in the selection between docetaxel and abiraterone is still unclear.
Our study has several limitations. Given the retrospective design, all analyses are subject to selection biases and imbalances in variables not quantifiable. Moreover, the small sample size, the single center cohort with confounders and no consistent follow-up period might limit the reproducibility of our results. Data regarding specific systemic therapies, duration of systemic therapy, and disease progression endpoints, which could have affected survival outcomes, were not evaluated. Finally, the prognostic value of ECOG PS and patient age has to be confirmed in mHSPC patients treated with docetaxel or abiraterone.
In conclusion, our analysis showed a lack of complete concordance between the CHAARTED and LATITUDE classifications, highlighting the importance of both of them for patient stratification. Two main patterns can be delineated: patients with a very good prognosis (LV/LR disease), and patients with a very poor prognosis (HV/HR group). Moreover, we supported the prognostic value of the CHAARTED and LATITUDE risk groups in a de novo mHSPC population. Adding patient age and ECOG PS to these classifications could improve the current prognostic models.
Further clinical and molecular prognostic factors are needed to guide the choice of a specific therapy for mHSPC patients.
References
Center MM. International variation in prostate cancer incidence and mortality rates. Eur Urol. 2012;61:1079–92.
Malvezzi G. European cancer mortality predictions for the year 2017, with focus on lung cancer. Ann Oncol. 2017;5:1117–23.
Buzzoni C. Metastatic prostate cancer incidence and prostate-specific antigen testing: new insights from the European randomized study of screening for prostate cancer. Eur Urol. 2015;68(5):885–90.
Wu JN. No improvement noted in overall or cause-specific survival for men presenting with metastatic prostate cancer over a 20-year period. Cancer. 2014;120(6):818–23.
James ND. Survival with newly diagnosed metastatic prostate cancer in the "docetaxel era": data from 917 patients in the control arm of the STAMPEDE trial (MRC PR08, CRUK/06/019). Eur Urol. 2015;67(6):1028–38.
Berg KD. Improved survival for patients with de novo metastatic prostate cancer in the last 20 years. Eur J Cancer. 2017;72:20–7.
Huggins C. Studies on prostatic cancer. II. The effects of castration on advanced carcinoma of the prostate cancer. Arch Surg. 1941;43:209–23.
Seidenfeld J. Single-therapy androgen suppression in men with advanced prostate cancer: a systematic review and meta-analysis. Ann Intern Med. 2000;132:566–77.
Sweeney CJ. Chemohormonal therapy in metastatic hormone-sensitive prostate cancer. N Engl J Med. 2015;373(8):737–46.
Fizazi K. Abiraterone plus prednisone in metastatic, castration-sensitive prostate cancer. N Engl J Med. 2017;377(4):352–60.
James ND. Abiraterone for prostate cancer not previously treated with hormone therapy. N Engl J Med. 2017;377(4):338–51.
James ND. Addition of docetaxel, zoledronic acid, or both to first-line long-term hormone therapy in prostate cancer (STAMPEDE): survival results from an adaptive, multiarm, multistage, platform randomised controlled trial. Lancet. 2016;387(10024):1163–77.
Gravis G. Androgen deprivation therapy (ADT) plus docetaxel versus ADT alone in metastatic non castrate prostate cancer: impact of metastatic burden and long-term survival analysis of the randomized phase 3 GETUG-AFU15 trial. Eur Urol. 2016;70(2):256–62.
Fizazi K. Should docetaxel be standard of care for patients with metastatic hormone-sensitive prostate cancer? Pro and contra. Ann Oncol. 2015;26(8):1660–7.
Finianos A. Characterization of differences between prostate cancer patients presenting with De novo versus primary progressive metastatic disease. Clin Genitourin Cancer. 2017;S1558-7673(17):30247–51.
Mosillo C. De novo metastatic castration sensitive prostate cancer: state of art and future perspectives. Cancer Treat Rev. 2018.
Tannock IF. Many men with castrate-sensitive metastatic prostate cancer should not receive chemotherapy. Ann Oncol. 2016;27(3):545–6.
Iacovelli R. De novo, progressed, and neglected metastatic castration-sensitive prostate cancer: is one therapy fit for all? Clin Genitourin Cancer. 2018.
Tait C. Quantification of skeletal metastases in castrate-resistant prostate cancer predicts progression-free and overall survival. BJU Int. 2014;114(6b):E70–3.
Glass TR. Metastatic carcinoma of the prostate: identifying prognostic groups using recursive partitioning. J Urol. 2003;169(1):164–9.
Tangen CM. Ten-year survival in patients with metastatic prostate cancer. Clin Prostate Cancer. 2003;2(1):41–5.
Yossepowitch O. The natural history of noncastrate metastatic prostate cancer after radical prostatectomy. Eur Urol. 2007;51(4):940–7.
Strock J. Characterization of patients who present with de novo metastatic prostate cancer: single-institution database analysis. J Clin Oncol. 2013;31(6_supplement):33.
Halabi S. Clinical outcomes by age in men with hormone refractory prostate cancer: a pooled analysis of 8 cancer and leukemia group B (CALGB) studies. J Urol. 2006;176(1):81–6.
Pettersson A. Age at diagnosis and prostate cancer treatment and prognosis: a population-based cohort study. Ann Oncol. 2018;29(2):377–85.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No external funding was used in the preparation of this manuscript.
Conflict of Interest
Roberto Iacovelli, Chiara Ciccarese, Claudia Mosillo, Davide Bimbatti, Emanuela Fantinel, Lisa Stefani, Michele Simbolo, Mario Romano, Renzo Mazzarotto, Matteo Brunelli, Emilio Bria, Aldo Scarpa, Rita T. Lawlor, Walter Artibani, and Giampaolo Tortora declare that they have no conflicts of interest that might be relevant to the contents of this manuscript.
Additional information
Roberto Iacovelli and Chiara Ciccarese share first co-authorship
Rights and permissions
About this article
Cite this article
Iacovelli, R., Ciccarese, C., Mosillo, C. et al. Comparison Between Prognostic Classifications in De Novo Metastatic Hormone Sensitive Prostate Cancer. Targ Oncol 13, 649–655 (2018). https://doi.org/10.1007/s11523-018-0588-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11523-018-0588-8