Abstract
The need for efficient cell separation, an essential preparatory step in many biological and medical assays, has led to the recent development of numerous microscale separation techniques. This review describes the current state-of-the-art in microfluidics-based cell separation techniques. Microfluidics-based sorting offers numerous advantages, including reducing sample volumes, faster sample processing, high sensitivity and spatial resolution, low device cost, and increased portability. The techniques presented are broadly classified as being active or passive depending on the operating principles. The various separation principles are explained in detail along with popular examples demonstrating their application toward cell separation. Common separation metrics, including separation markers, resolution, efficiency, and throughput, of these techniques are discussed. Developing efficient microscale separation methods that offering greater control over cell population distribution will be important in realizing true point-of-care (POC) lab-on-a-chip (LOC) systems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The early development of microfludics (a.k.a lab-on-a-chip or Micro Total Analysis System (μTAS)) as a field was strongly motivated by the need to analyze biomolecules more efficiently and accurately, which was culminated by the human genome project. However, the focus of the field has now been shifting more toward addressing the need for cell biology studies [4, 8, 29, 76, 99, 110]. While the number of molecular species to be tracked keeps increasing with the progress of different “-omics’, individual cells are more and more regarded as the basic “unit” of our biological understanding. With the appropriate length scale that matches the scales of cells, microfluidics is well positioned to contribute significantly to cell biology. However, one often faces some of the same challenges in molecular analysis; cell samples are highly complex, containing many different species at widely different abundance levels. Also, often the cells with least-abundance (rare cells) are of primary importance in diagnostics. In fact, measurement at the single cell level is necessary; the natural variation of cells (even from the same lineage) confounds the studies. As a result, techniques to separate and sort cells efficiently are as important as the biosample preparation steps in molecular analysis.
The application of microfluidic systems for cell separation, which is an essential preparatory step in many biological and biomedical assays, has led to the burgeoning of various microscale separation techniques over the past two decades. Separation and isolation of rare cell populations from a heterogeneous suspension is essential for many applications, ranging from disease diagnostics and drug treatment analysis to conducting fundamental studies. For example, diagnosis and treatment of HIV disease relies on the efficient separation of human T-lymphocytes (CD4+) from whole blood [17, 83]. Similarly, malaria diagnosis and treatment relies on the separation of parasite-infected red blood cells (RBCs) from uninfected cells [37, 38]. Separation of neuronal cells has gained interest for its potential applications in cell replacement therapy of neurodegenerative disorders such as Parkinson’s disease, multiple sclerosis, and Alzheimer’s [45, 60, 112]. Cell separation methods are also needed for separation of nucleated RBCs (NRBCs) from the peripheral blood of pregnant women for monitoring maternal, fetal, and neonatal health [50, 73]. Recently, various separation techniques have been successfully applied to separate stem cells based on their intrinsic properties to conduct fundamental studies [23, 24, 57]. As the need to perform fundamental studies and understand different cell types and their functions grows, separation of these individual cells becomes critical. Thus, cell separation techniques are critical to help develop and augment our understanding of stem cell biology.
Conventional cell separation systems employ membrane-based filtering schemes, which are limited by the membrane pore size and are easily susceptible to clogging. Microfluidics presents a functional tool-set for cell separation offering numerous advantages including, (i) reduced sample and expensive reagent volumes; (ii) fast sample processing, reducing analysis time; (iii) high sensitivity and spatial resolution, increasing detection accuracy; (iv) integrated reference systems with little human intervention, reducing odds of sample contamination; (v) increased portability—potential for point-of-care (POC) diagnostic in resource poor settings lacking clinical labs and skilled personnel; and (vi) low cost [18, 36, 98, 105]. Thus, developing efficient microscale separation methods that can offer greater control over cell size distribution is becoming increasingly important for realizing many lab-on-a-chip systems. A caveat against processing of small sample volumes is the increased likelihood of the analyzed sample not being representative of the entire population, especially while analyzing rare cell types.
Traditionally, microscale cell separation techniques take advantage of the disparities in the intrinsic properties of the different cell populations to achieve separations. Mechanical and physical properties, including size, shape, density, adhesion, and deformability, are common markers for differentiation. Due to their high sensitivity and efficiency, cell separation based on polarizability and magnetic characteristics have become extremely popular in the recent years. Highly specific separations based on differences in cell affinity (surface biomarkers) have also been effectively exploited to demonstrate separation between cells with similar physical and electromagnetic properties.
In this review, we aim to present the current state-of-the-art in microfluidic cell separation technologies. Although, several recent articles extensively review various microfluidic cell separation techniques, this study attempts to categorize the various separation principles as active or passive techniques based on the separation principle [31, 59, 63, 85, 92, 104]. Similar to other microfluidic components (e.g., micromixers, micropumps, and microvalves), microfluidic cell separation techniques can also be broadly classified as active and passive separation techniques [59, 66, 81, 84]. Simply put, active techniques rely on an external force field for functionality, while passive techniques rely entirely on the channel geometry and inherent hydrodynamic forces for functionality. In lieu of this classification, this review compares various passive and active cell separation techniques developed on the microscale (Table 1).
In this article, each presented technique includes a brief description of the separation principle followed by popular examples demonstrating its application toward cell separation. Common separation metrics including resolution, specificity, efficiency, and throughput of these microfluidic techniques will also be discussed. The presented review is by no means comprehensive and readers are encouraged to read the original papers for additional details.
2 Active separation techniques
2.1 Fluorescence-activated cell sorting (FACS) or flow cytometer: the “Gold Standard”
FACS has been the cell sorting method of choice for many biologists, due to its mature engineering development, sensitivity, and high throughput. In FACS, cells are identified via fluorescence signal from individual cells, one by one but at a relatively high speed, along the flow of cell solutions streaming past a detector. Typically, the cell surface markers are identified via specific antibodies labeled with fluorescent molecules. Alternatively, immunohistochemistry (fluorescent labeling of fixed cells) or cytosolic expression of GFP-tagged proteins is used to identify the subset of cells with specific properties. The detector observes either light scattering or the presence or absence of fluorescence signal coming from each cell and records it. If separation of the initial mixture is desired, various actuation methods are available to accomplish that goal. Traditionally, FACS involves ejecting single cells in liquid droplets of around 70 μm, which is a similar process used in an inkjet printer [52]. Each liquid droplet is first analyzed by the detector and then deflected using a tangential electric field to different buckets for sorting. Specifically, the maximum sort rate is on the order of 100,000 cells/s, based on physical principles [5]. While there are several technical drawbacks associated with this method (including equipment expense, clogging, contamination, serial nature of the sorting, and cell viability after ejection, deflection, and impact) [34], FACS has been the workhorse of biologist researchers, mainly due to the following reasons: (1) Single cell level sensitivity—FACS is capable of detecting cell surface markers at the single cell level largely due to the excellent sensitivity of the fluorescence detection; (2) High throughput of the sorting/counting allows population-averaged single cell data; (3) Ability to track multiple parameters—recent advances in fluorescence dyes, coupled with the availability of low cost lasers, allows one to track multiple parameters at a reasonable equipment cost. A typical (not state-of-the-art) flow cytometry system can routinely count/sort more than 10,000 cells/s with the ability of tracking 4–5 different fluorescence wavelengths (therefore tracking 4–5 different cell surface markers/tagged cytosolic biomolecules) [11]. The equipment cost has come down significantly in recent years as well. Therefore, any new cell sorting techniques are often “benchmarked” against FACS.
More recently, fluorescence-based sorting of cells has been implemented in microfluidic devices. These methods often involve the flowing of cells past a microfluidic T-junction (Fig. 1). Optical forces (~100 cells/s) [109], electro-osmotic flow (~20 cells/s) [34], and hydrostatic pressure changes (~1 particle/s) [13, 62] can sufficiently influence whether each cell is deflected left or right at the T-junction. These microfluidic flow cytometers are not fully developed to match the throughput of commercial flow cytometers. However, they can still fill a niche application area of precious, low-abundance cell counting/sorting, since FACS machines require a minimum number of cells (~100,000) for operation (Fig. 1A) [109]. In addition, microfluidic cytometry eliminates the need to eject the cells in liquid droplets, a physical process that can adversely affect cell viability/signaling.
Fluorescence activated cell sorting (FACS). A top Schematic illustrating the principle of FACS. Reprinted by permission from Macmillan Publishers Ltd: Nature Biotechnology [109], copyright 2005. Cells dispersed in the input sample are focused into a single stream at the channel center using sheath flows before passing over the detection region. Based on the fluorescence detected the cells are then switched into one of the two outlets. bottom Bright-field and fluorescence images indicating the cell population in the waste and collection outlet wells. The results clearly show the majority of fluorescent GFP-positive cells collected at the collection well. B top Droplet-based FACS. Cells encapsulated in liquid droplets are sorted based on their fluorescent signatures. Also shown in the figure are individual frames indicating the sorting of fluorescent droplets. bottom Schematic representation of typical optical set-up for microfluidic based FACS. Redrawn with permission from [6], copyright 2009, The Royal Society of Chemistry
The main difference between conventional FACS and microfluidic FACS comes from the fact that in microfluidic channels, flow switching (necessary for sorting) would be subject to much higher drag, compared with droplet-based switching in conventional FACS. This has been addressed on the microscale by designing a droplet-based FACS system (Fig. 1B) [6]. One can also overcome this inherent limitation by building a parallel array of microfluidic cytometer units, the core strength of microfluidics technology. Such a strategy has been implemented, using dielectrophoresis [108] and large-scale optical-driven dielectrophoretic manipulation [19], to separate many cells in parallel, although these methods have not been explored in depth for FACS. At least one company (Innovative Micro Technology, Santa Barbara, CA) is commercializing such a parallel microfluidic cytometer system, matching the high throughput of conventional, droplet-based FACS systems.
Another interesting twist on FACS would be “parallelized cell cytometry”, where the fluorescence of cells is monitored in a highly parallel way, instead of the serial manner. The obvious advantage for this would be significant gain in cell scanning speed, both for microfluidic and standard cell cytometry. For example, Krivacic et al. [61] used fiber-optic fiber array scanning to analyze cells attached to an adhesive slide, at a speed of 300,000 cells/s. Love et al. [69] implemented the same idea in microfluidic format, to detect and isolate the particular B cell that produces antibodies toward a given target. In both examples, the impressive functionality is from the efficient manner of scanning many cells in parallel, leading to the detection/separation of even rare cells.
2.2 Magnetic sorting
In magnetic cell sorting, sample cells are first incubated with magnetic beads with the recognition molecule (antibodies), for “magnetic labeling” instead of fluorescence. Then, a magnetic field gradient is used to isolate the magnetic beads, which in turn picks out the cells. This is often also termed as Magnetically Actuated Cell Sorter (MACS®), which is a trademark of Miltenyi Biotec GmbH, a company that provides commercial solutions for magnetic cell sorting. In contrast to FACS, which is serial in nature, magnetic cell sorting can be operated in either a serial or a parallel manner, resulting in higher throughput. Up to 1011 cells can be processed in 30 min [102]. One clear advantage of using a magnetic field is that the magnetic field is largely permeable to biological tissues and cells and less likely to interfere with cell function or immunochemistry necessary for magnetic labeling. The entire process can be done in solution phase, thereby minimizing any physical damage to the cells. It was shown that the magnetic particles do not influence viability or function of the labeled cells and do not affect the results of FACS later [9, 102].
Magnetic cell sorting can be typically operated in either batch or continuous flow processing modes. Batch processing involves first the presence of a magnetic field to collect the cells attached to magnetic beads and then the absence of that magnetic field to collect the accumulated cells. A large-scale example of batch processing is the placement of magnets next to a column filled with ferromagnetic material, such as steel wool coated with plastic to prevent cell damage [75]. This technique is already commercialized and widely used for various cell sorting applications. Smaller-scale examples of batch processing involve magnets placed next to a microfluidic channel [35, 40] and local magnetic fields created by an orthogonal array of electrical wires [68]. Advantages to MACS include the possibility of collecting many cells at one point of time (sorting in parallel) and relatively low cost. However, there are several drawbacks to the method, such as low sensitivity (difficulty capturing cells in subpopulations below 1% [75]), the time-consuming and sensitive process of labeling cells with the magnetic beads, and the possible necessity for removing them following separation.
In continuous flow magnetic cell sorting, a quadrupole magnet is placed next to a liquid column [120]. A cell solution flows through the column, and cells attached to magnetic beads are deflected in one direction, while cells that do not have beads attached continue to go straight. A miniaturized, microfluidic version of this method involves parallel magnetic lines placed oblique to the direction of liquid flow in a parallel-plate flow chamber [9, 53]. Cells attached to magnetic beads follow the direction of the parallel magnetic lines, while other cells follow the direction of the liquid. Using a similar technique, recently Pamme and Wilhelm introduced continuous magnetic cell sorting based on free-flow magnetophoresis (Fig. 2A) [86]. The separation relies on the variation in the uptake of magnetic material between different cell populations. Drawbacks to this method include cell adhesion, low throughput, and low recovery (perhaps due to the variations in the number of beads attached to cells) [53].
Magnetic separation. A top Principle of free-flow magnetophoresis. The separation relies on the variation in the uptake of magnetic particles for different cell populations during a given incubation period. Applying a transverse magnetic field (y-direction) creates a field gradient across the channel resulting in cell separation based on magnetic moment. bottom Transmission and fluorescence images indicating the separation of a mixture of non-magnetic and magnetic (fluorescently labeled) macrophages. The fluorescently labeled magnetic cells are deflected toward exits 2 and 3, while the nonmagnetic macrophages exit via number 1. Reproduced with permission from [86], copyright 2006, The Royal Society of Chemistry. B right Design of a microfluidic magnetophoretic separator using a patterned ferromagnetic wire for blood separation. When a magnetic field is applied across the microchannel, the paramagnetic RBCs align along the ferromagnetic wire, while the diamagnetic WBCs are forced away, thus achieving separation. left Illustration of RBC concentration and separation at the microchannel outlet junction, indicating ~90% filtration efficiency. Reproduced with permission from [42], copyright 2006, The Royal Society of Chemistry
The use of a constant magnetic field to separate cells based on their inherent magnetic properties has been proposed, although not demonstrated. In contrast to MACS, where cells are attached to magnetic beads, this method relies on the inherent magnetism of each cell in a population. This method is applicable to several cell types, such as red blood cells (due to their high concentration of Fe ions). Zborowski et al. demonstrated magnetophoresis of red blood cells, as well as human lymphocytes labeled with iron-rich protein ferritin [119, 121]. An attempt to separate red blood cells from white blood cells has been published [42], based on the principle that white blood cells are diamagnetic and red blood cells can either be paramagnetic or diamagnetic (Fig. 2B).
2.3 Dielectrophoresis
Dielectrophoretic forces are caused when a non-uniform (inhomogeneous) electric field interacts with the induced electrical polarization, or dipole, of a cell. Depending on the frequency and the conductivity of cell cytosol and surrounding media, these forces tend to hold the cell in place near the high field region (positive dielectrophoresis) or push the cell away from the high field region (negative dielectrophoresis) (Fig. 3A) [30, 90]. Other factors affecting the magnitude of the dielectrophoretic force include membrane properties (permeability, capacitance, and conductivity) and size. Especially, dielectrophoretic forces are strongly dependent on size of the cell (~R 3).
Dielectrophoretic cell sorting. A top Schematic illustrating the principle of dielectrophoresis (DEP). In the presence of an inhomogeneous electric field, polarizable cells experience strong electrostatic forces directing them either toward (positive DEP) or away (negative DEP) from regions of stronger fields. bottom Photograph of a microfluidic device with patterned electrodes for separation of live and dead yeast cells. The viable yeast cells show a positive response and align along the electrode edges while dead cells show a negative response and are pushed away from the edges, thus achieving separation. Figure reprinted from [30], copyright 2005, with permission from Elsevier. B top Schematic of DEP-activated cell sorting (DACS). Dielectrophoretically responsive cells introduced in the sample flow are deflected into the collection channel by the patterned electrodes. The technique was successfully used to separate target E.coli cells tagged with streptavidin-coated polystyrene beads. bottom Sequentially captured optical micrographs showing the motion of labeled polystyrene beads under the influence of DEP deflection. Adapted from [47], copyright 2005 National Academy of Sciences, USA
Unlike magnetic field, there are many cases where biological cells’ inherent electrical characteristics (e.g., conductivity) are modified based on cell type and state. This allows for label-free (unlike FACS or magnetic sorting) cell sorting, without relying on immunochemistry. The types of cell populations that have been separated so far include bacteria from peripheral blood [15], one cell population from another cell population [48], and cultured cancer cells from normal cells [7, 16]. One of the critical challenges of dielectrophoresis separation is the strong size-dependence of dielectrophoresis. Most cell types exhibit natural size variations up to ~10%, which may override the cell-type specific differences in dielectrophoretic sorting and capturing. In addition, dielectrophoresis strongly depends on the conductivity of media (extracellular solution), and it is more difficult to achieve dielectrophoretic sorting in high conductivity, physiological buffers. Conductivity of media could also lead to Joule heating near the electrodes and bubble generation, as well as heat-related cell death. Often, the conductivity of the media should be modified (without disrupting the osmotic balance) to achieve specific sorting. One interesting recent development is isodielectric sorting [106, 107], where a gradient of media conductivity used to render the cell sorting is dependent primarily on the dielectric properties, not the size of the cell. This would be an important attribute in dealing with real cell samples.
Recently, a sorting technique based on “dielectrophoretic labeling” has been demonstrated for cell separations (Fig. 3B) [47]. Target bacterial cells tagged to polystyrene beads (using antibodies) were sorted using parallel oblique electrical lines that generated dielectrophoretic forces under alternating current conditions [47]. Cells attached to beads would be deflected into the collection channel, while unlabeled cells would be deflected into the waste channels. The throughput of this system was measured at ~10,000 cells/s.
2.4 Electrophoretic sorting
Similar to other colloids, cells have surface (membrane) charge density as well as zeta (membrane) potential, which could be reflective of the biological characteristics of the cells. This property (electrophoretic mobility of the cell) could be a basis for a non-labeled cell separation technique. Numerous studies dating back to the 1950s [2, 3, 12, 72, 74, 89] exist, demonstrating the correlation between cell states (tumor vs. normal, cell cycles, etc.) and their electrophoretic mobility. However, attempts to utilize this properties as a means to diagnose cancer have produced mixed results [46].
Electrophoresis is a powerful separation technique with much success in molecular separation, and it is certainly feasible that it can also be applied to cell separation. The ideal mode of operation would be a continuous, free-flow parallel electrophoresis, which has been demonstrated in a large-scale chamber in 1980s [43, 44, 51, 103]. However, cell separation using electrophoresis has not been drawing much attention among researchers, probably due to a number of reasons. Earlier implementations of free-flow cell electrophoresis (~10 cm dimensions) suffered a number of technical issues related to convection, which led to poor separation [1]. If so, microfluidic implementations of the same idea might address some of the technical challenges. In addition, the basic science behind this technique is complicated, both from biological (cell surface charge density, mainly provided by carbohydrates) and physical (colloid electrophoretic mobility) points of view. Still, the simplicity of the technique and the possibility of sorting cells with or without labels could make this a viable cell sorting technique.
2.5 Optical sorting
Light has been used to manipulate and separate particles depending on their optical polarizability, a technique that has been widely demonstrated by the popular optical trapping method. For sorting, demonstration of this technique using interferometric patterns of light for separations based on size (protein microcapsules) and refractive index (separating polymer from silica spheres) has been accomplished (Fig. 4A) [70], however, cell separation has not yet been provided. Guck et al. [41] used the interaction of cells (which render the cells to deform) with a focused laser beam to count and sort the cells based on deformability, in a serial manner. While throughput of this technique is still limited compared with FACS, this technique is unique in terms of tracking the cells’ deformabilty with very minimal preparation/labeling. Interaction between cells and focused optical fields can be utilized in large-scale array form [19] for cell sorting and manipulation, to increase the throughput.
A Optical separation. right Design of a microfluidic optical fractionation system for size-based and refractive index based separation. The particle mixture introduced through chamber B passes through a 3D optical lattice in the fractionation chamber (FC) where strong optical interactions selectively generate separate flow paths. left Separation of 2 μm (black crosses) and 4 μm (white crosses) diameter capsules in an optical lattice. Reprinted with permission from Macmillan Publishers Ltd: Nature [70], copyright 2003. B Acoustic separation. right Schematic illustrating the principle of acoustic separation by standing surface acoustic waves (SSAW) generated using interdigital transducers (IDT). The varying acoustic forces repositions the larger cells closer to the channel center and smaller cells farther from the center. Figure reproduced with permission from [96], copyright 2009, The Royal Society of Chemistry. left Illustration of a size-based acoustic fractionator using an ultrasonic transducers placed under the microchannel. Cells of varying sizes align at distinct positions across the microchannel cross-section based on the acoustic primary radiation force experienced. Reprinted with permission from [88], copyright 2007, American Chemical Society
2.6 Acoustic sorting
Ultrasonic acoustic resonance (standing waves) within a microchannel, induced by piezoelectric material, can produce radiation force to manipulate particles and molecules (Fig. 4B) [96]. Petersson et al. achieved free-flow acoustophoresis and demonstrated a continuous flow separation of differently sized beads (2–10 μm), and separation of blood cells (platelets, red cells, and leukocytes) (Fig. 4B) [88]. Because cell viability within the radiation force field could be a concern for this technique, Evander et al. [32] studied the force and temperature increase that are felt by the cells in the acoustic standing wave. Within the trap, neural stem cells were viable for more than 15 min. The maximum force felt by the cells is on the order of ~0.5 nN, which is comparable to other manipulation techniques such as optical trapping. In fact, acoustic standing waves have found more use in cell manipulation, trapping, and concentration. Johansson et al. [56] used acoustic cell manipulation to switch cell flow for a microfluidic FACS system. Norris et al. [82] used an acoustic standing wave within the microfluidic channel to collect sperm cells out of epithelial cell lysate for forensic DNA analysis. However, due to the nature of the system, materials for the device must be chosen carefully to transmit the acoustic power to the fluid properly [67].
3 Passive separation techniques
3.1 Pillar and weir structures
The use of pillared structures is attractive for size and deformability based cell sorting. By tailoring the dimensions in between pillars, selection of cells based on their size and deformability can be achieved. Huang et al. described a method of continuous separation based on “deterministic lateral displacement” (DLD) of micron-sized particles with separation resolution less than 20 nm [49]. The design of the microfluidic device consists of arrays of micro-pillar structures within the main flow channel (Fig. 5A). The micro-pillars are suitably placed in the microchannel such that cells larger than the critical diameter d c = 20% × 2w (w is the separation gap between two adjacent pillars) follow a deterministic path while smaller cells maintain an average downward flow direction around the pillars, leading to the formation of multiple streams based on size [49]. Varying the inter pillar distances, a wide range of cell sizes can be continuously separated using this technique. The same group successfully fractionated blood components by tailoring the design of the microfluidic devices to the sizes of the blood cells [25]. DLD offers a simple and elegant way to continuously separate cells over a wide range of particle sizes with superior resolution. Also, stacking up micro-pillar arrays of varying gaps enables sorting of multiple sizes of cells in a single pass as shown in Fig. 5A for the fractionation of whole blood. However, the presence of a large number of obstructions may lead to channel clogging issues. In addition, deformability of cells could pose a mechanism that blurs the size selectivity/resolution.
A Separation using deterministic lateral displacement. Schematic illustrating the separation principle by deterministic lateral displacement technique. Cells with diameter smaller than the critical diameter stay within the flow streamlines, while the larger cells are displaced to the right at each obstacle. center Schematic of the fractionating device used for separating the blood components using DLD. right Leukocyte and RBC concentration at the three outlets. Reprinted with permission from [25], copyright 2006 National Academy of Sciences, USA. B Schematic design of a microfluidic device for cancer cell (CTCs) isolation and enumeration. The isolation regions consist of arrays of crescent-shaped isolation wells or cell traps to trap cancer cells while allowing blood cells to sieve through. right Separation results indicating >80% isolation efficiency for different cancer cells over various operating pressures. Figure reproduced from [101], copyright 2009, with kind permission from Springer Science + Business Media, LLC. C Physical based separation using weir structures. left Schematic of a weir-based filter with 3.5 μm gap between the etched silicon dam and channel top for size based cell separation. The disc-shaped RBCs flow through the dam structures while the bigger WBCs are filtered out. Figure reprinted from [14, 111], copyright 1998, with permission from Elsevier. center A cross-flow based cell separation design using weir structures as size filters. right Device picture after filtration illustrating the clear separation of white blood cells from blood. Reprinted from [14], copyright 2008, with permission from Elsevier
Micropillar arrays have also been employed to demonstrate effective isolation of circulating tumor cells (CTCs) from blood [77, 101] and also in leukapheresis [94]. Sethu et al. showed leukocyte depletion from whole blood without channel clogging, offering the possibility for leukocyte depleted blood to be returned to the donor [94]. The microchannel design features three parallel channels separated by 2.5 μm spaced pillar structures alongside the central channel region, thus permitting passage of erythrocytes into the side channels while acting as barriers to leukocytes migration. Using a similar approach, CTCs, which are generally larger and stiffer than blood constituents, are isolated with different gap sizes [77] or through the use of an array of crescent-shaped isolation wells or cell traps to trap CTCs, and then subsequently release and retrieve them for further analysis (Fig. 5B) [101]. The simplicity of employing pillar arrays makes the technique attractive for various cell sorting applications.
Using weir structures is another popular cell sorting technique exploiting mechanical disparities between cell samples. Gaps are created in between weirs and the top cover, which act as a filter to permit smaller cells to go through while impeding larger cells, thus separating the heterogeneous cell sample. Wildings et al. used a series of such weir structures to isolate leukocytes from blood [111] as shown in Fig. 5C. The weir-type filter features a 3.5-μm gap between the silicon structure and the Pyrex top cover, effectively allowing the passage of biconcave-shaped erythrocytes of ~2 μm thickness. The major drawback of a direct filtration system is the clogging issues that may interfere with the separation process due to the processing of large sample volumes. Weir filters that are perpendicular to the flow cause cell buildup over time, leading to obstructions. In order to address this limitation, Chen et al. designed microfluidic devices with the weirs structures placed parallel to the main flow (Fig. 5C) [14]. The device consists of three parallel micro channels, separated by weir filtration blocks along the main flow direction. With a 3.5-μm gap, the device allows erythrocytes and other smaller cells to cross from the main channel into the side flow, while effectively restricting the leukocytes in the main channel (Fig. 5C). However, such mechanical filtration systems that separate cells by geometrical differences would require variations in design for different samples. Furthermore, reported studies show limited efficiency in the target cell isolation [111], although it is sufficient for carrying out further downstream assays.
3.2 Pinched flow fractionation
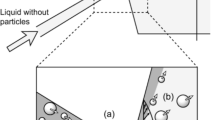
Pinched flow fractionation (PFF) is a novel hydrodynamic chromatographic technique for size-based separations in microchannels introduced by Yamada et al. [116]. In PFF, a dilute sample stream is pumped along with a sheath buffer by means of two inlets. The microchannel design consists of a pinched segment which opens into a broadened segment; the width of the pinched segment is comparable to the diameter of the cells and significantly smaller than the broadened segment (Fig. 6A). The operating principle of this technique relies on controlling the flow rate of the sheath buffer such that the cells in the sample flow are aligned to one channel sidewall in the pinched segment. This results in the center of inertia of the smaller sized cells being aligned closer to the channel wall as compared to the bigger cells. The parabolic velocity profile in Poiseuille flow significantly amplifies this difference in the lateral positions of the center of inertia between cells of varying sizes in the broadened segment, directing cells of different sizes into distinct outlets to achieve separation. The technique was employed to achieve separation of a mixture of 15 and 30 μm polystyrene particles [116]. The group also applied the technique to successfully filter erythrocytes from solution [100] (Fig. 6A), and to sort emulsion droplets based on size [71]. Recently, PFF was used to identify point mutations in the HBB gene with functionalized polystyrene microspheres of different sizes [65]. As a separation technique, PFF offers many advantages over other microfluidic cell separation methods. As the separation efficiency is based solely on the laminar flow profile in the pinched and broadened segment, cells of varying sizes can be separated effectively by tuning the ratio of the sample and sheath buffer flow rates in the same device. Separation efficiencies >90% with throughput in the order of ~4,000 particles/min have been demonstrated [100]. Higher flow rates are limited as inertial forces acting on the particles tend to affect particle motion, affecting separation efficiency.
Aleft Schematic illustrating the principle of pinched flow fractionation. Suspended particles are aligned against a microchannel sidewall in the pinched segment and are separated according to their sizes as they flow into the broadened segment as a result of expansion of the laminar flow sheet. Reprinted with permission from [116], copyright 2004, American Chemical Society. right Photograph and schematic illustration of erythrocyte separation (indicated by arrows) employing PFF in a microfluidic device is also shown. Figure reproduced with permission from [100], copyright 2005, The Royal Society of Chemistry. B Principle of hydrodynamic filtration. (a–c) Schematic diagrams showing particle positions at a branch point at; a low, b medium, and c high relative flow rates. d Schematic diagram showing particle concentration and separation into multiple side channels. Figure reproduced with permission from [115], copyright 2005, The Royal Society of Chemistry
3.3 Hydrodynamic filtration
Hydrodynamic filtration is another passive separation technique introduced by Yamada et al. for size-based separation in a microfluidic device [115]. In this technique, particle laden flow is pumped through a channel having multiple side branching outlets. These side channels drain liquid from the main channel continuously, thereby aligning all particles along the sidewalls of the main microchannel (Fig. 6B). The difference in size positions the smaller particles closer to the sidewall than larger particles, thus resulting in them being filtered out earlier than the larger sized particles. The technique was initially used to demonstrate ~29 fold enrichment of leukocytes in blood [115]. Recently, the group demonstrated size-based separation of liver cells with throughputs >2 × 105 cells/min [117]. As the separation mechanism is based purely on the flow profile and not microchannel geometry, channels significantly larger than the cell diameters can be employed, minimizing clogging and further increasing throughput.
3.4 Inertial forces
Inertial lift forces inherent to cell motion in microchannel flows can be exploited to precisely manipulate the cell position within these flows. Although, the first experimental study on such inertial migration was demonstrated in the 1960s [93], it was not until very recently that such phenomenon has been employed for microfluidic-based cell separation [27]. Lateral migration of cells in microchannels occurs due to the superposition of two inertial lift forces: the shear-induced lift force and the wall-induced lift force [87]. These two forces equilibrate the cells at distinct positions within the microchannel cross-section based on their size relative to the microchannel dimensions, thus achieving separation (Fig. 7A). A recent review highlights the applications of this technique toward high-throughput cell separations [26]. Inertial migration of cells was employed for demonstrating a 100-fold platelet enrichment in blood using asymmetrical serpentine microchannels (Fig. 7B) [28], and for the separation of neuroblastoma cells and glioma cells with 80% efficiency and high cell viability (>90%) (Fig. 7C) in spiral microchannels [64]. The device reported a throughput of ~106 cells/min, significantly higher than many microfluidic cell separation techniques. Recently, using inertial lift force-induced migration, separation of bacteria from human blood cells with greater than 99% efficiency and an exceedingly high throughput of ~4 × 106 cells/min (Fig. 7D) was demonstrated [113]. Since this technique relies entirely on intrinsic hydrodynamic forces, the design of these systems is very simple and can be easily integrated with existing microfluidic modules. Also, the channel dimensions are comparably larger than the sample cell sizes, leading to both high volume throughput and eliminating problems arising from channel clogging. As these systems can function in a continuous fashion, cell separation throughputs of 106 cells/min are achievable. Finally, cell–cell interactions can deteriorate the efficiency of this separation technique, limiting its application to only dilute samples, thus requiring prior sample preparation procedures.
Inertial lift force-induced cell separation. A Inertial lift forces exerted on cells suspended in a Poiseuille flow. The combined effect of the shear-induced lift force and the wall-induced lift force align cells at distinct equilibrium positions based on their size. Reproduced with permission from [87], copyright 2009, The Royal Society of Chemistry. B Schematic showing the inertial lift force-based self ordering in asymmetrical serpentine microchannel (inset). Adapted from [27], copyright 2007 National Academy of Sciences, USA. Cells of varying sizes focus differentially resulting in their separation as demonstrated by 100-fold enrichment of platelets from blood. Figure reprinted with permission from [28], copyright 2008, American Chemical Society. C Differential inertial microfluidic based cell separation in spiral microchannels demonstrating separation of SH-SY5Y neuroblastoma cells (pseudo-colored green) from smaller C6 glioma cells. Redrawn with permission from [64], copyright 2009, The Royal Society of Chemistry. D Separation of blood cells (8 μm) from bacteria (1 μm) using soft inertial microfluidics employing just a single curved sample flow segment. Reproduced with permission from [113], copyright 2009, The Royal Society of Chemistry
3.5 Affinity-based separation
The use of affinity-based separation exploits the dependence of certain molecules recognizable on the cells of interest. Primarily, sorting is achieved in a heterogeneous sample by the binding of cells to the molecules immobilized onto the microfluidic device. Unwanted cells are permitted to flow through without interaction and are removed. The technique is highly specific, targeting only cells that express the complementary group of molecules and has the ability to separate cells of similar physical characteristics (size and density). Molecules commonly used in affinity-based cell separation include proteins as well as antibodies, and immobilization techniques are either by covalent [122] bonds or absorption [10]. The sensitivity of the technique is enhanced by the use of microscale devices whose surface area to volume ratio is significantly larger than macroscale adhesion assays. Such technologies have been demonstrated for separating mixed suspensions of T and B lymphocytes [79] which can aid in the study of lymphocyte subpopulations in immune responses during diseases or injury. Anti-CD5 and anti-CD19 antibodies are used to recognize different cell subpopulations, which show the ability to obtain high purity (>97%) in separating the mixed suspension. A linear shear stress flow chamber is used (Fig. 8A), which determines the binding effect at various shear rates. Purity of captured subpopulations remains high when the concentration of target cells in the suspension is low, demonstrating the high sensitivity of the system. Nagrath et al. have also demonstrated the methodology for isolating circulating tumor cells (CTCs) from peripheral blood (Fig. 8B) [80]. CTCs are reported to be a good measure of cancer prognosis but the rarity of cells in blood poses the technical difficulty of enumerating them. Using anti-EpCAM antibodies coated on microposts, effective isolation of CTCs from whole blood is achieved in a single processing step (Fig. 8B). The specificity of the biomarkers is the major limitation of this technique and the selection of a suitable molecule is crucial for an effective separation. For instance, Siewarts et al. show low expression levels of EpCAM on various breast cancer cell lines [97], which is routinely used for enumeration of CTCs in breast cancer patients. Furthermore, optimization of operating parameters needs to be considered for different binding molecules so that sufficient interaction time is given for the binding of targeted cells to occur. These problems can be overcome by having a clear understanding of the intended applications so that a suitable marker can be selected and the binding kinetics of the molecules can be known. Most affinity-based separation techniques are based on “capture and release” methods, while continuous flow sorting would be more desirable in terms of sorting efficiency and selectivity. Karnik et al. [58] implemented a slanted affinity pattern, on which interacting cells are allowed to roll along the edges of the pattern, to separate cells continuously based on the surface affinity.
Affinity-based separation. A Linear shear stress chamber used by Murthy et al. to determine the effect of shear stress on the binding and separation efficiency of T and B lymphocytes. Figure reproduced with permission from [79], copyright 2004, American Chemical Society. B Layout of the CTC chip designed by Nagrath et al. consisting of micropost array. Inset showing a scanning electron micrograph of a lung cancer cell between two chemically functionalized microposts. Reprinted with permission from Macmillan Publishers Ltd: Nature [80], copyright 2007
3.6 Biomimetic separation
In blood vessels with luminal diameter less than 300 μm, red blood cells (RBCs), which are smaller in size and more deformable than leukocytes, tend to migrate to the axial center of the vessel as a result of the parabolic velocity profile in the vessel [91]. This results in the formation of a reduced hematocrit plasma layer adjacent to the vessel wall, the Fahraeus–Lindqvist effect [39]. As the RBCs migrate radially toward the axial center, mechanical collisions between RBCs and leukocytes displace the leukocytes to the vessel wall; a process aptly termed as margination. This unique hemodynamic microcirculation effect has been extensively studied using microfluidic systems and has been applied for leukocyte enrichment using asymmetrical bifurcations [55, 78, 95] and the separation of plasma from whole blood [33, 54, 118], and (Fig. 9). This biomimetic separation technique uses whole blood samples eliminating additional sample preparation steps.
Fahraeus effect based cell separation. A A 34-fold leukocyte enrichment was achieved by employing a 5.5 mm long rectangular microchannel based on leukocyte margination (WBCs indicated by arrows). The larger WBCs are displaced to the channel sidewalls and extracted using an asymmetrically bifurcated outlet. Figure redrawn with permission from [95], copyright 2005, American Chemical Society. B Microfluidic device with a series of parallel side channels used for plasma skimming from whole blood. The figure also shows a high-speed snapshot confirming the concentration of RBCs at the microchannel center with pure plasma collected at the plasma outlet. Figure reproduced with permission from [118], copyright 2006, The Royal Society of Chemistry
3.7 Other mechanisms
In microfluidic channels, various other passive cell sorting mechanisms have been implemented, aided by low dispersion and tight control on individual cells by pressure fields. Recently, Choi et al. [21, 22] introduced a novel size-based separation technique based on hydrophoretic sizing. The technique relies on the generation of a pressure field gradient induced using slanted obstacles placed in the flow path. This principle was used to demonstrate enrichment of WBCs from RBCs with a 210-fold enrichment ratio at a throughput of 4 × 103 cells/s. Another mechanism is using the inherent motility of cells (either by diffusion or active motility) as a biomarker for cell separation. For example, Cho et al. [20] used a microfluidic laminar flow junction of two streams, one of which contains sperm cells, to separate motile, healthy sperm from unhealthy ones.
4 Conclusions and outlook
In this review, we presented current state-of-the-art microfluidic cell separation technologies and categorized them as active or passive depending on their separation principles. Here, active means the microfluidic devices will need to rely on an external force field to function while passive means that they will rely predominantly on channel geometry and/or inherent hydrodynamic forces.
The development of microfluidic cell separation technology and its use in the biomedical and clinical sciences will continue to grow and expand for several reasons. First, the use of microfluidics will result in significant reduction in the samples and reagents needed. This not only saves cost, but also means less invasive extraction of bodily fluids such as blood from patients. Second, small sample size requires less processing time and hence less waiting time to realize the outcome. Third, advancement in micro and nanotechnology has also resulted in higher sensitivity and spatial resolution when sensing samples, and this will inevitably increase detection accuracy. Fourth, a self-contained and self-automated microfluidics system will not only prevent contamination and hence false results but also require little or no human intervention. Thus, a simple to use device can be easily created. Finally, there will be increased portability, and this is especially important when used in regions where there is serious lack of well-equipped clinical labs and skilled personnel.
Current microfluidics technology has already demonstrated that it can be employed to separate and characterize individual diseased or rare cells from a population of cells more rapidly, with enhanced sensitivity and accuracy, and in a controlled and reproducible way. In the near future, an integrated approach can be developed to perform a series of more complex tasks apart from just cell separation. These can include accurately manipulating and handling even smaller sample volumes of bodily fluids and cells, providing the necessary microenvironment and controlled stimuli to perform further testing on the separated cells of interest, and implementing high-resolution observation techniques to analyze the biological functions or gene expression in these cells. Ultimately, the aim is to develop a portable and inexpensive diagnostic microfluidic device that can integrate all the necessary complex processes on a chip. Such microfluidic devices can then be made available to regions where clinical facilities and support are scarce and can also be used to detect the onset or progression of diseases or even assess the efficacy of certain drugs to combat a disease.
References
Akagi T, Ichiki T (2008) Cell electrophoresis on a chip: what can we know from the changes in electrophoretic mobility? Anal Bioanal Chem 391:2433–2441
Akagi T, Suzuki M, Ichiki T (2006) Application of on-chip electrophoresis of cell to evaluation of cell cycle stages of HL-60 cells. Jpn J Appl Phys Part II Lett 45:L1106–L1109
Ambrose EJ, James AM, Lowick JHB (1956) Differences between the electrical charge carried by normal and homologous tumour cells. Nature 177:576–577
Andersson H, van den Berg A (2003) Microfluidic devices for cellomics: a review. Sens Actuators B Chem 92(3):315–325
Ashcoroft RG, Lopez PA (2000) Commercial high speed machines open new opportunities in high throughput flow cytometry. J Immunol Methods 243:13–24
Baret JC et al (2009) Fluorescence-activated droplet sorting (FADS): efficient microfluidic cell sorting based on enzymatic activity. Lab Chip 9(13):1850–1858
Becker FF et al (1995) Separation of human breast cancer cells from blood by differential dielectric affinity. Proc Natl Acad Sci 92:860–864
Beebe DJ, Mensing GA, Walker GM (2002) Physics and applications of microfluidics in biology. Annu Rev Biomed Eng 4:261–286
Berger M et al (2001) Design of a microfabricated magnetic cell separator. Electrophoresis 22:3883–3892
Bernard A, Michel B, Delamarche E (2001) Micromosaic immunoassays. Anal Chem 73(1):8–12
Bonetta L (2005) Flow cytometry smaller and better. Nature Methods 2:785–795
Brent TP, Forrester JA (1967) Changes in surface charge of HeLa cells during the cell cycle. Nature 215:92–93
Chen C et al (2003) Design and operation of a microfluidic sorter for Drosophila embryos. Sens Acuators B 102:59–66
Chen X et al (2008) Microfluidic chip for blood cell separation and collection based on crossflow filtration. Sens Actuators B Chem 130(1):216–221
Cheng J et al (1998) Preparation and hybridization analysis of DNA/RNA from E. coli on microfabricated bioelectronic chips. Nat Biotechnol 16:541–546
Cheng J et al (1998) Isolation of cultured cervical carcinoma cells mixed with peripheral blood cells on a bioelectronic chip. Anal Chem 70:2321–2326
Cheng XH et al (2007) A microfluidic device for practical label-free CD4 + T cell counting of HIV-infected subjects. Lab Chip 7(2):170–178
Chin CD, Linder V, Sia SK (2007) Lab-on-a-chip devices for global health: past studies and future opportunities. Lab Chip 7(1):41–57
Chiou PY, Ohta AT, Wu MC (2005) Massively parallel manipulation of single cells and microparticles using optical images. Nature 436:370–372
Cho BS et al (2003) Passively driven integrated microfluidic system for separation of motile sperm. Anal Chem 75(7):1671–1675
Choi S, Park JK (2007) Continuous hydrophoretic separation and sizing of microparticles using slanted obstacles in a microchannel. Lab Chip 7(7):890–897
Choi S et al (2007) Continuous blood cell separation by hydrophoretic filtration. Lab Chip 7(11):1532–1538
Colter DC, Sekiya I, Prockop DJ (2001) Identification of a subpopulation of rapidly self-renewing and multipotential adult stem cells in colonies of human marrow stromal cells. Proc Natl Acad Sci USA 98(14):7841–7845
David R, Groebner M, Franz WM (2005) Magnetic cell sorting purification of differentiated embryonic stem cells stably expressing truncated human CD4 as surface marker. Stem Cells 23(4):477–482
Davis JA et al (2006) Deterministic hydrodynamics: taking blood apart. Proc Natl Acad Sci USA 103(40):14779–14784
Di Carlo D (2009) Inertial microfluidics. Lab Chip 9(21):3038–3046
Di Carlo D et al (2007) Continuous inertial focusing, ordering, and separation of particles in microchannels. Proc Natl Acad Sci USA 104(48):18892–18897
Di Carlo D et al (2008) Equilibrium separation and filtration of particles using differential inertial focusing. Anal Chem 80(6):2204–2211
Dittrich PS, Manz A (2006) Lab-on-a-chip: microfluidics in drug discovery. Nat Rev Drug Discov 5(3):210–218
Doh I, Cho YH (2005) A continuous cell separation chip using hydrodynamic dielectrophoresis (DEP) process. Sens Actuators A Phys 121(1):59–65
Eijkel JCT, van den Berg A (2006) Nanotechnology for membranes, filters and sieves. Lab Chip 6(1):19–23
Evander M et al (2007) Noninvasive acoustic cell trapping in a microfluidic perfusion system for online bioassays. Anal Chem 79(7):2984–2991
Faivre M et al (2006) Geometrical focusing of cells in a microfluidic device: an approach to separate blood plasma. Biorheology 43(2):147–159
Fu AY et al (1999) A microfabricated fluorescence-activated cell sorter. Nat Biotechnol 17:1109–1111
Furdui VI, Harrison DJ (2004) Immunomagnetic T cell capture from blood for PCR analysis using microfluidic systems. Lab Chip 4(6):614–618
Gardeniers JGE, van den Berg A (2004) Lab-on-a-chip systems for biomedical and environmental monitoring. Anal Bioanal Chem 378(7):1700–1703
Gascoyne P et al (2002) Microsample preparation by dielectrophoresis: isolation of malaria. Lab Chip 2(2):70–75
Gascoyne P, Satayavivad J, Ruchirawat M (2004) Microfluidic approaches to malaria detection. Acta Tropica 89(3):357–369
Goldsmith HL, Cokelet GR, Gaehtgens P (1989) Robin Fahraeus: evolution of his concepts in cardiovascular physiology. Am J Physiol 257(3):H1005–H1015
Grodzinski P et al (2003) A modular microfluidic system for cell pre-concentration and genetic sample preparation. Biomed Microdevices 5(4):303–310
Guck J et al (2005) Optical deformability as an inherent cell marker for testing malignant transformation and metastatic competence. Biophys J 88:3689–3698
Han KH, Frazier AB (2006) Paramagnetic capture mode magnetophoretic microseparator for high efficiency blood cell separations. Lab Chip 6(2):265–273
Hannig K (1982) New aspects in preparative and analytical continuous free-flow Cell electrophoresis. Electrophoresis 3:235–243
Hansen E, Hannig K (1984) Electrophoretic separation of lymphoid cells. Methods Enzymol 108:180–197
Hedlund E et al (2007) Selection of embryonic stem cell-derived enhanced green fluorescent protein-positive dopamine neurons using the tyrosine hydroxylase promoter is confounded by reporter gene expression in immature cell populations. Stem Cells 25(5):1126–1135
Hoffmann W et al (1981) Cell electrophoresis for diagnostic purposes. I. Diagnostic value of the electrophoretic mobility test (EMT) for the detection of gynaecological malignancies. J Cancer 43(5):588–597
Hu X et al (2005) Marker-specific sorting of rare cells using dielectrophoresis. Proc Natl Acad Sci 104(44):15757–15761
Huang Y et al (2002) Dielectrophoretic cell separation and gene expression profiling on microelectronic chip arrays. Anal Chem 74:3362–3371
Huang LR et al (2004) Continuous particle separation through deterministic lateral displacement. Science 304(5673):987–990
Huang R et al (2008) A microfluidics approach for the isolation of nucleated red blood cells (NRBCs) from the peripheral blood of pregnant women. Prenat Diagn 28(10):892–899
Hymer W et al (1987) Continuous flow electrophoretic separation of proteins and cells from mammalian tissues. Cell Biochem Biophys 10(1):61–85
Ibrahim S, van den Engh G (2003) High-speed cell sorting: fundamentals and recent advances. Curr Opin 14:5–12
Inglis DW et al (2004) Continuous microfluidic immunomagnetic cell separation. Appl Phys Lett 85(21):5093–5095
Jaggi RD, Sandoz R, Effenhauser CS (2007) Microfluidic depletion of red blood cells from whole blood in high-aspect-ratio microchannels. Microfluid Nanofluid 3(1):47–53
Jain A, Munn LL (2009) Determinants of leukocyte margination in rectangular microchannels. PLoS ONE 4(9):e7104
Johansson L et al (2009) On-chip fluorescence-activated cell sorting by an integrated miniaturized ultrasonic transducer. Anal Chem 81(13):5188–5196
Jones EA et al (2002) Isolation and characterization of bone marrow multipotential mesenchymal progenitor cells. Arthritis Rheum 46(12):3349–3360
Karnik R et al (2008) Nanomechanical control of cell rolling in two dimensions through surface Patterning of receptors. Nano Lett 8(4):1153–1158
Kersaudy-Kerhoas M, Dhariwal R, Desmulliez MPY (2008) Recent advances in microparticle continuous separation. IET Nanobiotechnol 2(1):1–13
Korecka JA, Verhaagen J, Hol EM (2007) Cell-replacement and gene-therapy strategies for Parkinson’s and Alzheimer’s disease. Regen Med 2(4):425–446
Krivacic RT et al (2004) A rare-cell detector for cancer. Proc Natl Acad Sci 101(29):10501–10504
Kruger J et al (2002) Development of a microfluidic device for fluorescence activated cell sorting. J Micromech Microeng 12:486–494
Kulrattanarak T et al (2008) Classification and evaluation of microfluidic devices for continuous suspension fractionation. Adv Colloid Interface Sci 142(1–2):53–66
Kuntaegowdanahalli SS et al (2009) Inertial microfluidics for continuous particle separation in spiral microchannels. Lab Chip 9(20):2973–2980
Larsen AV et al (2008) Pinched flow fractionation devices for detection of single nucleotide polymorphisms. Lab Chip 8(5):818–821
Laser DJ, Santiago JG (2004) A review of micropumps. J Micromech Microeng 14(6):R35–R64
Laurell T, Petersson F, Nilsson A (2007) Chip integrated strategies for acoustic separation and manipulation of cells and particles. Chem Soc Rev 36:492–506
Lee H, Purdon AM, Westervelt RM (2004) Manipulation of biological cells using a microelectromagnet matrix. Appl Phys Lett 85(6):1063–1065
Love JC et al (2006) A microengraving method for rapid selection of single cells producing antigen-specific antibodies. Nat Biotechnol 24(6):703–707
MacDonald M, Spalding G, Dholakia K (2003) Microfluidic sorting in an optical lattice. Nature 426:401–404
Maenaka H et al (2008) Continuous and size-dependent sorting of emulsion droplets using hydrodynamics in pinched microchannels. Langmuir 24(8):4405–4410
Makino K et al (1993) Measurements and analyses of electrophoretic mobilities of RAW 117 lymphosarcoma cells and their variant cells. Biophys Chem 47(3):261–265
Mavrou A et al (2007) Identification of nucleated red blood cells in maternal circulation: a second step in screening for fetal aneuploidies and pregnancy complications. Prenat Diagn 27(2):150–153
Mayhew E, O’GRADY EA (1965) Electrophoretic mobilities of tissue culture cells in exponential and parasynchronous growth. Nature 207:86–87
Miltenyi S et al (1990) High gradient magnetic cell separation with MACS. Cytometry 11:231–238
Mitchell P (2001) Microfluidics—downsizing large-scale biology. Nat Biotechnol 19(8):717–721
Mohamed H et al (2009) Isolation of tumor cells using size and deformation. J Chromatogr A 1216(47):8289–8295
Munn LL, Dupin MM (2008) Blood cell interactions and segregation in flow. Ann Biomed Eng 36(4):534–544
Murthy SK et al (2004) Effect of flow and surface conditions on human lymphocyte isolation using microfluidic chambers. Langmuir 20(26):11649–11655
Nagrath S et al (2007) Isolation of rare circulating tumour cells in cancer patients by microchip technology. Nature 450(7173):1235-U10
Nguyen NT, Wu ZG (2005) Micromixers—a review. J Micromech Microeng 15(2):R1–R16
Norris JV et al (2009) Acoustic differential extraction for forensic analysis of sexual assault evidence. Anal Chem 81(15):6089–6095
Obrien WA et al (1997) Changes in plasma HIV RNA levels and CD4(+) lymphocyte counts predict both response to antiretroviral therapy and therapeutic failure. Ann Intern Med 126(12):939–945
Oh KW, Ahn CH (2006) A review of microvalves. J Micromech Microeng 16(5):R13–R39
Pamme N (2007) Continuous flow separations in microfluidic devices. Lab Chip 7(12):1644–1659
Pamme N, Wilhelm C (2006) Continuous sorting of magnetic cells via on-chip free-flow magnetophoresis. Lab Chip 6(8):974–980
Park JS, Song SH, Jung HI (2009) Continuous focusing of microparticles using inertial lift force and vorticity via multi-orifice microfluidic channels. Lab Chip 9(7):939–948
Petersson F et al (2007) Free flow acoustophoresis: microfluidic-based mode of particle and cell separation. Anal Chem 79(14):5117–5123
Petty HR, Ware BR, Wasserman SI (1980) Alterations of the electrophoretic mobility distribution of rat mast cells after immunologic activation. Biophys J 30(1):41–50
Pohl HA (1978) Dielectrophoresis. Cambridge University Press, London
Pries AR, Secomb TW, Gaehtgens P (1996) Biophysical aspects of blood flow in the microvasculature. Cardiovasc Res 32(4):654–667
Radisic M, Iyer RK, Murthy SK (2006) Micro- and nanotechnology in cell separation. Int J Nanomed 1(1):3–14
Segre G, Silberberg A (1961) Radial particle displacements in poiseuille flow of suspensions. Nature 189:209–210
Sethu P, Sin A, Toner M (2006) Microfluidic diffusive filter for apheresis (leukapheresis). Lab Chip 6(1):83–89
Shevkoplyas SS et al (2005) Biomimetic autoseparation of leukocytes from whole blood in a microfluidic device. Anal Chem 77(3):933–937
Shi JJ et al (2009) Continuous particle separation in a microfluidic channel via standing surface acoustic waves (SSAW). Lab Chip 9(23):3354–3359
Sieuwerts AM et al (2009) Anti-epithelial cell adhesion molecule antibodies and the detection of circulating normal-like breast tumor cells. J Natl Cancer Inst 101(1):61–66
Soper SA et al (2006) Point-of-care biosensor systems for cancer diagnostics/prognostics. Biosens Bioelectron 21(10):1932–1942
Stone HA, Kim S (2001) Microfluidics: basic issues applications, and challenges. Aiche J 47(6):1250–1254
Takagi J et al (2005) Continuous particle separation in a microchannel having asymmetrically arranged multiple branches. Lab Chip 5(7):778–784
Tan SJ et al (2009) Microdevice for the isolation and enumeration of cancer cells from blood. Biomed Microdevices 11(4):883–892
Thiel A, Scheffold A, Radbruch A (1998) Immunomagnetic cell sorting—pushing the limits. Immunotechnology 4:89–96
Todd P et al (1986) Electrophoretic separation and analysis of living cells from solid tissues by several methods: human embryonic kidney cell cultures as a model. J Chromatogr A 364:11–24
Tsutsui H, Ho CM (2009) Cell separation by non-inertial force fields in microfluidic systems. Mech Res Commun 36(1):92–103
Tudos AJ, Besselink GAJ, Schasfoort RBM (2001) Trends in miniaturized total analysis systems for point-of-care testing in clinical chemistry. Lab Chip 1(2):83–95
Vahey MD, Voldman J (2008) An equilibrium method for continuous-flow cell sorting using dielectrophoresis. Anal Chem 80(9):3135–3143
Vahey MD, Voldman J (2009) High-throughput cell and particle characterization using isodielectric separation. Anal Chem 81(7):2446–2455
Voldman J et al (2002) A microfabrication-based dynamic array cytometer. Anal Chem 74:3984–3990
Wang MM et al (2005) Microfluidic sorting of mammalian cells by optical force switching. Nat Biotechnol 23(1):83–87
Whitesides GM (2006) The origins and the future of microfluidics. Nature 442(7101):368–373
Wilding P et al (1998) Integrated cell isolation and polymerase chain reaction analysis using silicon microfilter chambers. Anal Biochem 257(2):95–100
Wu ZG et al (2008) Microfluidic high viability neural cell separation using viscoelastically tuned hydrodynamic spreading. Biomed Microdevices 10(5):631–638
Wu ZG et al (2009) Soft inertial microfluidics for high throughput separation of bacteria from human blood cells. Lab Chip 9(9):1193–1199
Xia N et al (2006) Combined microfluidic-micromagnetic separation of living cells in continuous flow. Biomed Microdevices 8(4):299–308
Yamada M, Seki M (2005) Hydrodynamic filtration for on-chip particle concentration and classification utilizing microfluidics. Lab Chip 5(11):1233–1239
Yamada M, Nakashima M, Seki M (2004) Pinched flow fractionation: continuous size separation of particles utilizing a laminar flow profile in a pinched microchannel. Anal Chem 76(18):5465–5471
Yamada M et al (2007) Microfluidic devices for size-dependent separation of liver cells. Biomed Microdevices 9(5):637–645
Yang S, Undar A, Zahn JD (2006) A microfluidic device for continuous, real time blood plasma separation. Lab Chip 6(7):871–880
Zborowski M et al (1995) Analytical magnetapheresis of ferritin-labeled lymphocytes. Anal Chem 67:3702–3712
Zborowski M et al (1999) Continuous cell separation using novel magnetic quadrupole flow sorter. J Magn Magn Mater 194:224–230
Zborowski M et al (2003) Red blood cell magnetophoresis. Biophys J 84:2638–2645
Zhang ZL et al (2005) In situ bio-functionalization and cell adhesion in microfluidic devices. Microelectron Eng 78-79:556–562
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Bhagat, A.A.S., Bow, H., Hou, H.W. et al. Microfluidics for cell separation. Med Biol Eng Comput 48, 999–1014 (2010). https://doi.org/10.1007/s11517-010-0611-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11517-010-0611-4