Abstract
The present work seeks to determine if a particular non-linear analytic method is effective at quantifying uterine electromyography (EMG) data for estimating the onset of labor. Twenty-seven patients were included, and their uterine EMG was recorded non-invasively for 30 min. The patients were grouped into two sets: G1: labor, N=14; G2: antepartum, N=13. G1 patients all delivered spontaneously within 24 h of recording while G2 patients did not. The uterine electrical signals were analyzed offline by first isolating the uterine-specific frequency range and then randomly selecting “bursts” of uterine electrical activity (each associated with a uterine contraction) from every recording. Wavelet transform was subsequently applied to each of the bursts’ traces, and then the fractal dimension (FD) of the resulting transformed EMG burst-trace was calculated (Benoit 1.3, Trusoft). Average burst FD was found for each patient. FD means for G1 and G2 were calculated and compared using t test. FD was significantly higher (P<0.05) for G1: 1.27±0.03 versus G2: 1.25±0.02. The wavelet-decomposition-generated fractal dimension can be used to successfully discern between patients who will deliver spontaneously within 24 h and those who will not, and can be useful for the objective classification of antepartum versus labor patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
One of the most important health problems in obstetrics today is pre-term birth and its associated complications, which contribute to handicaps and are the cause of about 85% of all perinatal deaths [29]. Pre-term neonates, with birth-weights less than 2,500 g, represent about 10% of the total number of babies born each year. The complications of pre-term birth include significant neurological, mental, behavioral and pulmonary problems in later life. Among the pre-term survivors, the rate of neurological impairment varies from 10 to 20% and growth restriction occurs in approximately 20% of the surviving infants. The development of effective methods to prevent or reduce the incidence of pre-term birth depends upon the understanding of the mechanisms that initiate labor.
Perhaps the most difficult and important task facing medical practitioners in maternity care today is the diagnosis of labor. Knowing that true labor (which will lead to delivery) has begun, as well as predicting when it will start, is important for both normal and aberrant pregnancies. Prediction of labor in normal pregnancies is important for minimizing unnecessary hospitalizations, interventions and expenses. On the other hand, accurate prediction and diagnosis of spontaneous pre-term labor will also allow clinicians to start any necessary treatment early in women with true labor and avert unnecessary treatment and hospitalization in women who are simply having pre-term contractions, but who are not in true labor. Even noticeable dynamic cervical change may not be an accurate indicator of true labor, as a large percentage of women with established cervical change do not deliver pre-term when not treated with tocolytics [15]. To date, the only effective method for preventing pre-term labor has been constant contact and care from health care practitioners [11], a very inefficient and expensive process.
Many studies, including our own, have demonstrated that myometrial (uterine) cells are coupled together electrically by gap junctions composed of proteins [6]. The grouping of proteins provides channels of low electrical resistance between cells that facilitate pathways for the efficient conduction of action potentials. Throughout most of pregnancy, and in all species studied, these cell-to-cell channels or contacts are few, indicating poor coupling and decreased electrical conductance. This condition favors quiescence of the myometrium and the maintenance of pregnancy. At term, however, in order to properly expel the developed fetus, the cell junctions increase and form an electrical syncytium required for effective contractions [6]. As action potentials propagate over the surface of a myometrial cell, the depolarization causes voltage-dependent Ca2+ channels to open. When this occurs, Ca2+ enters the muscle cell, traveling down its electro-chemical gradient to activate the myofilaments and provokes a contraction [28] by increasing the size and/or number of actual portals for Ca2+ entry.
Previous studies established that this myometrial electrical activity governs myometrial mechanical contractions [14, 20]. Extensive studies have been done in the last 60 years to monitor uterine contractility using the electrical activity measured from electrodes placed on the uterus [4, 5, 30]. These studies also revealed that the most significant human uterine electrical activity resides somewhere in the DC−3.00 Hz. frequency range. Some studies have now indicated that uterine electromyography (EMG) activity can be monitored accurately and non-invasively from the abdominal surface [3, 7, 8]. In a most recent study, we also quantified human uterine EMG signals over a narrow band of frequencies (uterine-specific, 0.34–1.00 Hz) using power spectrum analysis, a method generally most applicable to linear phenomena [17]. The technique has proved useful for the classification of pregnant patients. However, it may be that non-linear methods are even more suitable for uterine electrical data [21].
Fractal analysis is one such non-linear analytic candidate. A fractal is an object composed of subunits (and sub-subunits) that resemble the larger-scale structure, a property known as self-similarity [10]. Fractal processes generate irregular fluctuations on multiple time scales, analogous to fractal objects that have “wrinkly” structure on different length scales. The roughness of these time series, therefore, possesses a self-similar (scale-invariant) property. Since scale-invariance appears to be a general mechanism underlying many physiological structures and functions, one can adapt new quantitative tools derived from fractal mathematics for measuring their variability.
Complex fluctuations with the statistical properties of fractals have been described for heart rate variability [13], fluctuations in respiration, [27] systemic blood pressure [19], white blood cell counts [9] and certain ion channel kinetics [2]. If uterine electrical traces (recorded during contractions) possess fractal properties, then they also could be characterized by this method. The present work seeks to determine if this particular non-linear analytic technique is effective at quantifying uterine EMG data for discerning between labor and non-labor patients.
2 Materials and methods
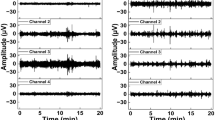
In this proof-of-concept study, 27 pregnant patients, with known gestational ages ranging from 25–39+ weeks, were retrospectively included, after having been admitted to the labor and delivery area of the University of Texas Medical Branch in the Department of Obstetrics and Gynecology and upon signing an IRB-approved informed consent to participate. Patient ages ranged from 17 to 35 years old. Along with standard tocodynamometer contraction data acquisition, their uterine EMG was simultaneously recorded non-invasively for 30 min, using bipolar sets of abdominal surface electrodes, and by sampling at 100 Hz, with analog band-pass filtering from 0.05 to 4.00 Hz (Fig. 1). Patients were required to lay supine or on their side, and to remain still during recordings in order to reduce movement artifact. There is always some degree of overlap in electrical frequencies from various signal sources in the body, or from external movements. However, we isolated what we have determined to be a uterine-specific range (again, 0.34–1.00 Hz for humans) using digital filtering, in order to remove unwanted respiratory influences (generally <0.34 Hz for the patients studied) and cardiac influences (generally >1.00 Hz for the patients studied), as well as most remaining movement artifact [17]. The recordings were then saved and grouped into two sets: G1: labor, with N=14; G2: antepartum (i.e., non-labor), with N=13.
G1 and G2 patients were distinguished by a clinical determination, which considered the number of contractions per unit time, the strength of uterine contractions observed, and the extent of cervical dilation. Patients were excluded for whom a clear clinical determination of labor could not be made. Patients exhibiting distress or abnormal pregnancy were excluded. All G1 patients ultimately term-delivered (i.e., delivered at 37+ weeks gestation) spontaneously within 24 h of EMG recording, while G2 patients term-delivered spontaneously more than 24 h from EMG measurement. The patients were retrospectively grouped in this way in order to improve the likelihood that the uterine EMG signals would have different fractal properties between G1 and G2. Thirty-three such term patients were recruited over a 20-month period of time. However, six of these patients were excluded due to having cesarean section rather than undergoing spontaneous vaginal delivery.
The uterine electrical signals acquired were analyzed offline by first randomly selecting “bursts” of uterine electrical activity (each burst was a period of elevated electrical activity, and each was associated with a uterine contraction) from every recording. Wavelet transform was subsequently applied (Benoit 1.3, Trusoft) to each of the individual bursts’ traces, thereby decomposing the uterine burst signal into the first three wavelet “scales” (the particular wavelet used was a step-function). The wavelet method of signal decomposition is seemingly ideal for uterine EMG data, which may involve non-stationary traces, i.e., the variance of the trace does not remain constant with increasing length of the data set.
The wavelet method of determining fractal dimension is based on the property that the wavelet transform of a self-affine trace also has self-affine properties. One measure of the fractality of a trace is the fractal dimension (FD), decreasing from two to one, which represents from “more-” to “less-wavy” lines drawn in two dimensions. Such concepts can be applied directly to uterine electrical activity traces, specifically those occurring during electrical bursts associated with uterine contractions, for labor versus non-labor patients.
Consider n wavelet transforms each with a different scaling coefficient a i , where S 1, S 2,...,S n are the standard deviations from zero of the respective scaling coefficients a i.
Define the ratio of the standard deviations G 1, G 2,...,G n−1 as:
Estimate the average value of G i as:
The Hurst exponent (H) is H=f(G avg), where f is a heuristic function that approximates the Hurst exponent by G avg for stochastic self-affine traces. Benoit fractal analysis software (Trusoft) sets n=4 and a i =2 i for i=0, 1, 2, 3. By definition, the fractal dimension FD=2−H.
Statistics An average FD was found for the electrical bursts of each given patient. The FD group means for both G1 and G2 were found and compared using Student’s t test. Also, average FD was compared to both gestational age and measurement-to-delivery (MTD) time interval for each patient in order to search for correlation using Pearson product moment. Receiver–operator characteristic (ROC) curves were generated. In all statistical tests, P<0.05 was considered significant.
3 Results
Bursts of uterine electrical activity corresponded to contractions as measured by tocodynamometer (Fig. 2a, b), but this can be clearly seen only when the uterine EMG signals are properly filtered to remove noise. The average gestational age of G1 was significantly higher than that of G2 (P<0.05; mean±SD=39.24±1.23 vs. 32.58±3.91 weeks, respectively; Fig. 3). The average uterine EMG MTD interval was significantly lower for G1 as compared to G2 (P<0.05; mean±SD=0.20±0.16 vs. 30.77±21.51 days, respectively; Fig. 4). Average FD was significantly higher for G1 compared to G2 (P<0.05; mean±SD=1.27±0.03 vs. 1.25±0.02, respectively; Fig. 5). A highly significant negative correlation for gestational age and MTD interval (P<0.001; R=−0.768) was found. However, FD demonstrated a significant negative correlation with the MTD interval (P<0.05; R=−0.457; Fig. 6a, b), but had no significant correlation of any kind with gestational age (P>0.05; R=0.165). ROC analysis, using a golden standard, or endpoint, of 24 h (Fig. 7) was used to find a measure of the predictive quality of the data. From this, we also calculated sensitivity (sens), specificity (spec), positive predictive value (PPV), and negative predictive value (NPV) for various cutoffs of FD (Table 1).
a Contraction events, as measured mechanically by tocodynamometer (bottom trace), and as recorded simultaneously by non-invasive trans-abdominal uterine EMG (top trace—unfiltered signal). The electrical activity is actually responsible for uterine muscle contractions. Therefore, analysis of the uterine electrical signals should give a more precise measure of the uterine state, and the evolution of the parturition, than measurement with tocodynamometer, which can only measure, crudely, the force of the contraction and number of contractions/unit-time. b This time the EMG data (top trace) was properly filtered in the uterine-specific range (0.34–1.00 Hz) in order to show the bursts of uterine activity. Note the exceptional correspondence between the EMG measurement and the tocodynamometer readings for contractions when the EMG signal is properly filtered
a A significant negative correlation between FD and MTD (P<0.05; R=−0.457) was seen. As delivery becomes imminent, the fractal dimension of the associated uterine electrical activity increases. Note that the 14 labor patients’ data are represented by triangles, while the 13 antepartum patients’ data-points are marked by circles. b Since the 14 labor-patients all delivered within 24 h, it is necessary to expand the y-axis in order to visually discern their individual FD versus MTD plots
Receiver–operator characteristics (ROC) curve for the 24-h endpoint, showing: area under the curve (Area), Z-value, standard error (St. Err.) and the one-tailed P value. The results suggest that FD may be a good predictor. However, future studies should incorporate more patients to increase the accuracy of the ROC analysis, especially if a head-to-head comparison between FD and other analytic methods is to be properly conducted
4 Discussion
The significant difference between FD for labor patients versus antepartum patients indicates that the wavelet-decomposition-generated fractal dimension method is capable of discerning between those patients who will soon deliver and those who will not. The higher FD value of the labor patients indicates a more highly active electrical signal, corresponding to the increased electrical activity and frequencies associated with a well-developed uterus that is electrochemically prepared for intense labor contractions to expel the developed fetus. The negative correlation between FD and MTD is expected, since the shift to greater uterine electrical activity should occur as the patient gets closer to actual delivery at term. The lack of any significant correlation between FD and gestational age is perhaps a little surprising, especially considering the strong relation between gestational age and MTD; however, this simply verifies that increased electrical activity, hence transition to labor, and ultimately to delivery, are not necessarily dependent upon the patient’s gestation, and this has been known for some time.
As stated previously, in recent studies we successfully quantified uterine EMG signals using power spectrum analysis, a method generally considered most applicable to linear phenomena [17]. But many biological electrical processes are thought to be non-linear in origin [2, 21] to either a greater or lesser degree. The prior success of the power-spectrum approach to quantifying uterine electrical activity could possibly be attributed to the fact that only a narrow band of frequencies (the uterine-specific range) was examined, which may have approximated conditions for linearity in the surface electrical signals examined, or it may be that the specific frequency range of uterine electrical activity is governed primarily by linear processes. In any case, it should probably be emphasized that the power-spectrum technique has not only proven itself useful for our own work in classification of pregnant human patients based on uterine electrical frequency changes; it has also been used effectively for years to observe spectral characteristics and changes in various other biological (non-linear?) processes and species [18, 24–26, 31]. The general rule is that when distinct power peaks in the spectral representation are observed, even in the midst of apparent broadband “noise” at other frequencies, power-spectrum classification of the phenomenon is usually appropriate [1]. This has been the case with the uterine electrical data observed so far [17]. However, it could be that a number of non-linear methods are indeed suitable for uterine electrical data. Based on the results of the present work, fractal analysis apparently is one such promising analytic method; evidently there are at least some non-linear components in the trans-abdominally acquired uterine electrical signals studied that can be characterized with wavelet/fractal methods.
Even long ago, it was recognized that linear modeling of the abdominal-surface electrical signals, generated from the uterus and acquired by finite-size electrodes, was probably best at only a good approximation of the actual process [22], although some have defended the use of linear models of the surface EMG, at least for non-uterine muscle [23]. For most muscles, the issue is complicated even further by the possibility of non-stationarity of the signals even during a single contraction, owing to the fact that such contractions are generally only approximately isokinetic [12, 16, 23]. It is presumed that this is also the case for uterine contractions as well. However, since the number of muscle cells involved in producing external electrical signals is very large, some have contended that non-stationarity would not greatly affect the power-spectral shape or frequency content after all, but only its relative size, and this would be especially true if using short-time Fourier transform (STFT) for quantification [12, 16, 23]. Though most of those studies considered formulations based on striated and innervated muscle, it may be that their fundamental principles largely apply for uterine smooth muscle as well.
The present 27-patient study was essentially a proof-of-concept investigation. The patients who were included were either already clearly in labor or they were clearly non-labor. Now that this initial work is completed, it remains to be seen whether or not non-linear analytic methods are superior to the power spectrum approach for quantification of uterine electrical data, and ultimately, to the classification of pregnant patients or prediction of labor and delivery. Future work, using a greater number of patients (as in our previous investigations of the power-spectrum [17]), and using “rule-out” patients (for which a clinical determination of labor vs. non-labor is not so certain), might also involve utilizing Lyapunov exponents for the uterine electrical burst analysis, for example. A head-to-head comparison of the predictive capability of the non-linear method against spectral or other parameters by the way of ROC curves applied to the same patient data set for analysis could then be performed. Only in this way could one confidently demonstrate that one method is superior to another. To do otherwise may invite premature and erroneous conclusions about how the different methods “stack-up” against each other.
It is possible, however, that it will be found in the end that the application of the tandem of linear and non-linear analytic techniques ultimately ends up producing the most complete and useful description about the state of the uterus. With such work, the non-invasive uterine EMG-monitoring method could allow for highly accurate forecasting of labor and delivery in pregnant women, and thus would enable clinicians the capability to apply much better treatment for, and exercise superior management of, pregnant patients than with any currently available tools [7, 8].
References
Abarbanel HDI (1996) Analysis of observed chaotic data. Springer, Berlin Heidelberg New York esp. see pp 11, 14, 31, 37, 198–202
Bassingthwaighte JB, Liebovitch LS, West BJ (1994) Fractal physiology. Oxford University Press, New York
Buhimschi C, Garfield RE (1998) Uterine activity during pregnancy and labor assessed by simultaneous recordings from the myometrium and abdominal surface in the rat. Am J Obstet Gynecol 178:811–822
Devedeux D, Marque C, Mansour S, Germain G, Duchene J (1993) Uterine electromyography: a critical review. Am J Obstet Gynecol 169:1636–1653
Figueroa JP, Honnebier MB, Jenkins S, Nathanielsz PW (1990) Alteration of 24-h rhythms in the myometrial activity in the chronically catheterized pregnant rhesus monkey after 6-hours shift in the light-dark cycle. Am J Obstet Gynecol 163:648–654
Garfield RE, Yallampalli C (1994) Structure and function of uterine muscle. In: Chard T, Grudzinskas JG (eds) The uterus. Cambridge reviews in human reproduction. Cambridge University Press, Cambridge, UK, pp 54–93, 40–81
Garfield RE, Buhimschi C (1998) Control and assessment of the uterus and cervix during pregnancy and labour (Sep–Oct). Hum Reprod Update 4(5):673–695
Garfield RE et al (1998) Instrumentation for the diagnosis of term and pre-term labour. J. Perinat Med 26:413–436
Goldberger AL (1997) Fractal variability versus pathologic periodicity: complexity loss and stereotypy in disease. Perspect Biol Med 40:543–561
Goldberger A (1999) Nonlinear dynamics, fractals, and chaos theory: implications for neuroautonomic heart rate control in health and disease. Bolis CL, Licinio J (eds) The Autonomic nervous system. World Health Organization, Geneva
Goldenberg RL, Cliver SP, Bronstein J, Cutter GR, Andrews WW, Mennemeyer ST (1994) Bed rest in pregnancy. Obstet Gynecol 84:131–136
Karlsson JS, Gerdle B, Akay M (2001) Analyzing surface myoelectric signals recorded during isokinetic contractions. IEEE Eng Med Biol November/December:97–105
Kobayashi M, Musha T (1982) 1/f fluctuation of heartbeat period. IEEE Trans Biomed Eng 29:456–457
Kuriyama H, Csapo A (1967) A study of the parturient uterus with the microelectrode technique. Endocrinology 80:748–753
Linhart J, Olson G, Goodrum L, Rowe T, Saade G, Hankins G (1990) Pre-term labor at 32 to 34 weeks’ gestation: effect of a policy of expectant management on length of gestation. Am J Obstet Gynecol 178:S179
Maclsaac DT, Parker PA, Scott RN, Englehart KB, Duffley C (2001) Influence of dynamic factors on myoelectric parameters. IEEE Eng Med Biol November/December:82–89
Maner W, Garfield RE, Maul H, Olson G, Saade G (2003) Predicting term and pre-term delivery in humans using transabdominal uterine electromyography. Obstet Gynecol 101(6):1254–1260
Mansour S, Devedeux D, Germain G, Marque C, Duchene J (1996) Uterine EMG spectral analysis and relationship to mechanical activity in pregnant monkeys. Med Biol Eng Comput 34(2):115–121
Marsh DJ, Osborn JL, Cowley AW (1990) 1/f fluctuations in arterial pressure and regulation of renal blood flow in dogs. Am J Physiol 258:F1394–F1400
Marshall JM (1962) Regulation of the activity in uterine muscle. Physiol Rev 42:213–227
Nagarajan R, Eswaran H, Wilson JD, Murphy P, Lowery C, Preibl H (2003) Analysis of uterine contractions: a dynamical approach. J Maternal Fetal Neonatal Med 14:8–21
Nagel J, Schaldach M (1980) Recording of uterine activity from the abdominal lead EMG, in fetal and neonatal physiological measurements. In: Rolfe P (ed) Pitman Medical Limited, Tunbridge Wells pp 177–182
Pan ZS, Zhang Y, Parker PA (1989) Motor unit power spectrum and firing rate. Med Biol Eng Comput 27:14–18
Sheridan TB, Meyer JE, Roy SH, Decker KS, Yanagishima T, Yoichi K (1991) Physiological and psychological evaluations of driver fatigue during long term driving. In: International congress and exposition of the engineering society for advancing mobility land sea air and space, Feb 25–March 1
Stief CG, Kellner B, Hartung C, Hauck E, Schlote N, Truss M, Hinrichs H, Jonas U (1997) Computer-assisted evaluation of the smooth-muscle electromyogram of the corpora cavernosa by fast Fourier transformation. Eur Urol 31(3):329–334
Struijk PC, Ursem NTC, Mathews J, Clark EB, Keller BB, Wladimiroff JW (2001) Power spectrum analysis of heart rate and blood flow velocity variability measured in the umbilical and uterine arteries in early pregnancy: a comparative study. Ultrasound Obstet Gynecol 17(4):316–321
Szeto H, Chen PY, Decena JA, Cheng YI, Wu Dun-L, Dwyer G (1992) Fractal properties of fetal breathing dynamics regulatory interactive comp physiol. Am J Physiol 263 (32):R141–R147
Tezuka N, Ali M, Chwalisz K, Garfield RE (1995) Changes in transcripts encoding calcium channel subunits of rat myometrium during pregnancy. Am J Physiol 269:C1008–C1017
U.S. Preventive Services Task Force (1989) Guide to clinical preventive services: an assessment of the effectiveness of 169 interventions. Williams & Wilkins, Baltimore
Wolfs GMJA, Van Leeuwen (1979) Electromyographic observations on the human uterus during labor. Acta Obstet Gynecol Scand Suppl 90:1–61
Yamada K, Isotani T, Irisawa S, Yoshimura M, Tajika A, Yagyu T, Saito A, Kinoshita T (2004) EEG Global field power spectrum changes after a single dose of atypical antipsychotics in healthy volunteers. Brain Topogr Summer 16(4):281–285
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maner, W.L., MacKay, L.B., Saade, G.R. et al. Characterization of abdominally acquired uterine electrical signals in humans, using a non-linear analytic method. Med Bio Eng Comput 44, 117–123 (2006). https://doi.org/10.1007/s11517-005-0011-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11517-005-0011-3