Abstract
Background
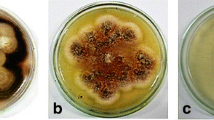
The microbes living in planta termed ‘endophytes’ is bestowed with the potential to produce bioactive substances. The aim of this investigation was focused on the isolation and molecular identification of the fungal endophytes from Zingiber nimmonii (J. Graham) Dalzell., an endemic medicinal plant species of the ‘Western ghats’, a hotspot location in southern India and characterization of the secondary metabolites responsible for the antioxidant and DNA protective capacity using chromatography and mass spectrometry techniques.
Methods
Endophytic fungi were isolated and identified by sequencing the Internal Transcribed Spacer (ITS). The secondary metabolites were extracted with ethyl acetate and evaluated for the total phenolic, flavonoid and antioxidant capacities. The isolates with potential antioxidative property were further analyzed for the DNA protection ability and the presence of bioactive phenolic compounds by High Performance Liquid Chromatography (HPLC) and Electrospray Ionization-Mass Spectroscopy/Mass Spectroscopy (ESI-MS/MS) techniques.
Results
Endophytic fungi belonging to 11 different taxa were identified. The total phenolic content of the extracts ranged from 10.8±0.7 to 81.6±6.0 mg gallic acid equivalent/g dry extract. Flavonoid was present in eight extracts in the range of 5.2± 0.5 to 24.3±0.9 mg catechin equivalents/g dry extract. Bipolaris specifera, Alternaria tenuissima, Aspergillus terreus, Nectria haematococca and Fusarium chlamydosporum extracts exhibited a potentially high antioxidant capacity. Characterization of the extracts revealed an array of phenolic acids and flavonoids. N. haematococca and F. chlamydosporum extracts contained quercetin and showed DNA protection ability.
Conclusion
This study is the first comprehensive report on the fungal endophytes from Z. nimmonii, as potential sources of antioxidative and DNA protective compounds. The study indicates that Z. nimmonii endophytes are potential sources of antioxidants over the plant itself.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ausubel F M, Brent R, Kingston R E, Moore D D, Seidman J G, Smith J A, Struhl K (1994). Current protocols in molecular biology. Wiley, New York
Barros L, Ferreira M J, Queirós B, Ferreira C F R, Baptista P (2007). Total phenols, ascorbic acid, ß-carotene and lycopene in Portuguese wild edible mushrooms and their antioxidant activities. Food Chem, 103(2): 413–419
Bussaban B, Lumyong S, Lumyong P, Mckenzie E H C, Hyde K D (2001). Endophytic fungi from Amomum siamense. Can J Microbiol, 47(10): 943–948
Çelik H, Arinç E (2010). Evaluation of the protective effects of quercetin, rutin, resveratrol, naringenin and trolox against idarubicininduced DNA damage. J Pharm Pharm Sci, 13(2): 231–241
Cheng MJ, Wu MD, Chen J J, Cheng Y C, Hsieh MT, Hsieh S Y, Yuan G F, Su Y S (2014). Secondary metabolites from the endophytic fungus Annulohypoxylon stygium BCRC 34024. Chem Nat Compd, 50(2): 237–241
Das A K, Singh V (2016). Antioxidative free and bound phenolic constituents in botanical fractions of Indian specialty maize (Zea mays L.) genotypes. Food Chem, 201: 298–306
Deng C M, Liu S X, Huang C H, Pang J Y, Lin Y C (2013). Secondary metabolites of a mangrove endophytic fungus Aspergillus terreus (No.GX7–3B) from the South China Sea. Mar Drugs, 11(7): 2616–2624
Duthie S J, Collins A R, Duthie G G, Dobson V L (1997). Quercetin and myricetin protect against hydrogen peroxide-induced DNA damage strand breaks and oxidised pyrimidines in human lymphocytes. Mutat Res, 393(3): 223–231
Finose A, Gopalakrishnan V K (2014). Antioxidant potential of Zingiber nimmonii (J. Graham) Dalzell. Int J Pharm PharmSci, 6(6): 50–52
Gamble J S (1928). Flora of the Presidency of Madras. Vol. III, Bishen Singh Mahenra Pal Singh, Dehra Dun, India, pp. 1487–1489
Huang W Y, Cai Y Z, Hyde K D, Corke H, Sun M (2007b). Endophytic fungi from Nerium oleander L (Apocynaceae): main constituents and antioxidant activity. World J Microbiol Biotechnol, 23(9): 1253–1263
Huang W Y, Cai Y Z, Xing J, Corke H, Sun M (2007a). A Potential antioxidant resource: endophytic fungi from medicinal plants. Econ Bot, 61(1): 14–30
Jasim B, Anisha C, Rohini S, Kurian J M, Jyothis M, Radhakrishnan E K (2014). Phenazine carboxylic acid production and rhizome protective effect of endophytic Pseudomonas aeruginosa isolated from Zingiber officinale. World J Microbiol Biotechnol, 30(5): 1649–1654
Jing P, Zhao S J, Jian W J, Qian B J, Dong Y, Pang J (2012). Quantitative studies on structure- DPPH scavenging activity relationships of food phenolic acids. Molecules, 17(12): 12910–12924
Karamac M, Kosiñska A, Pegg R B (2005). Comparison of radicalscavenging activities for selected phenolic acids. Pol J Food NutrSci, 14/55(2): 165–170
Kavitha P G, Kiran A G, Dinesh Raj R, Sabu M, Thomas G (2010). Amplified fragment length polymorphism analyses unravel a striking difference in the intraspecific genetic diversity of four species of four species of genus Zingiber Boehm. from the Western Ghats, South India. Curr Sci, 98(2): 242–246
Lee J C, Kim H R, Kim J, Jang Y S (2002). Antioxidant activity of ethanol extract of the stem of Opuntiaficus-indica var. saboten. J Agric Food Chem, 50(22): 6490–6496
Liu X, Dong M, Chen X, Jiang M, Lv X, Yan G (2007). Antioxidant activity and phenolics of an endophytic Xylaria sp. from Ginkgo biloba. Food Chem, 105(2): 548–554
Maldonado P D, Rivero-Cruz I, Mata R, Pedraza-Chaverrí J (2005). Antioxidant activity of A-type proanthocyanidins from Geranium niveum (Geraniaceae). J Agric Food Chem, 53(6): 1996–2001
Nalini MS, Sunayana N, Prakash HS (2014). Endophytic fungal diversity in medicinal plants of Western Ghats, India. Int J Biodiv, doi:org/10.1155/2014/494213.
Nongalleima K, Dey A, Lokesh D, Singh C B, Thongam B, Sunitibala D H, Indira D S (2013). Endophytic fungus isolated from Zingiber zerumbet (L.) Sm. inhibits free radicals and cyclooxygenase activity. Int J Pharm Tech Res, 5(2): 301–307
Ohkawa H, Ohishi N, Yagi K (1979). Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem, 95(2): 351–358
Onyema O O, Farombi E O, Emerole G O, Ukoha A I, Onyeze G O (2006). Effect of vitamin E on monosodium glutamate induced hepatotoxicity and oxidative stress in rats. Indian J Biochem Biophys, 43(1): 20–24
Oyaizu M (1986). Studies on product of browning reaction prepared from glucose amine. J Nutr, 44: 307–315
Pérez-Magariño S, Revilla I, González-SanJosé M L, Beltrán S (1999). Various applications of liquid chromatography-mass spectrometry to the analysis of phenolic compounds. J Chromatogr A, 847(1–2): 75–81
Pizarro J G, Folch J, De La Torre A V, Verdaguer E, Junyent F, Jordan J, Pallas M, Camins A (2009). Oxidative stress-induced DNA damage and cell cycle regulation in B65 dopaminergic cell line. Free Radic Res, 43(10): 985–994
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Evans C R (1999). Antioxidant activity applying and improved ABTS radical cation decolorization assay. Free Radic Biol Med, 26(9–10): 1231–1237
Sabulal B, Dan M, John J A, Kurup R, Pradeep N S, Valsamma R K, George V (2006). Caryophyllene-rich rhizome oil of Zingiber nimmonii from South India: chemical characterization and antimicrobial activity. Phytochemistry, 67(22): 2469–2473
Samaga P V, Rai V R (2016). Diversity and bioactive potential of endophytic fungi from Nothapodytes foetida, Hypericum mysorense and Hypericum japonicum collected from Western Ghats of India. Ann Microbiol, 66(1): 229–244
Schulz B, Guske S, Dammann U, Boyle C (1998). Endophyte host interactions II. Defining symbiosis of the endophyte host interaction. Symbiosis, 25: 213–227
Strobel G, Daisy B (2003). Bio prospecting for microbial endophytes and their natural products. Microbiol Mol Biol Rev, 67(4): 491–502
Strobel G, Yang X, Sears J, Kramer R, Sidhu R S, Hess W M (1996). Taxol from Pestalotiopsis microspora, an endophytic fungus of Taxus wallachiana. Microbiology, 142(2): 435–440
Sun J, Liang F, Bin Y, Li P, Duan C (2007). Screening non-colored phenolics in red wines using liquid chromatography/ultraviolet and mass spectrometry/mass spectrometry libraries. Molecules, 12(3): 679–693
Tan R X, Zou W X (2001). Endophytes: a rich source of functional metabolites. Nat Prod Rep, 18(4): 448–459
Tejesvi M V, Mahesh B, Nalini M S, Prakash H S, Kini K R, Subbiah V, Shetty H S (2005). Endophytic fungal assemblages from inner bark and twig of Terminalia arjuna W. & A. (Combretaceae). World J Microbiol Biotechnol, 21(8–9): 1535–1540
Thannickal V J, Fanburg B L (2000). Reactive oxygen species in cell signalling. Am J Physiol Lung Cell Mol Physiol, 279: L1005–L1028
Tiwari S, Singh S, Pandey P, Saikia S K, Negi A S, Gupta S K, Pandey R, Banerjee S (2014). Isolation, structure determination, and antiaging effects of 2,3-pentanediol from endophytic fungus of Curcuma amada and docking studies. Protoplasma, 251(5): 1089–1098
Tuma D J, Casey C A (2003). Dangerous byproducts of alcohol breakdown—focus on adducts. Alcohol Res Health, 27: 285–290
Wilson D (1995). Fungal endophytes: out of sight but should not be out of mind. Oikos, 68(2): 379–384
Yashavantha Rao H C, Rakshith D, Satish S (2015). Antimicrobial properties of endophytic actinomycetes isolated from Combretum latifolium Blume, a medicinal shrub from Western Ghats of India. Front Biol, 10(6): 528–536
Acknowledgements
The University Grants Commission–Major Research Project and the facilities utilized from Institution of Excellence, University of Mysore, are thankfully acknowledged. Thanks to Dr. Shylaja Dharmesh, Senior Scientist, Central Food Technological Research Institute, Mysore for the inputs in the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Das, M., Prakash, H.S. & Nalini, M.S. Antioxidative properties of phenolic compounds isolated from the fungal endophytes of Zingiber nimmonii (J. Graham) Dalzell.. Front. Biol. 12, 151–162 (2017). https://doi.org/10.1007/s11515-016-1441-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11515-016-1441-z