Abstract
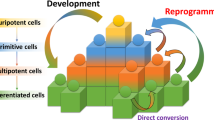
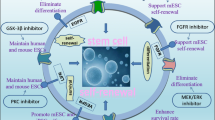
Direct reprogramming technology has emerged as an outstanding technique for the generation of induced pluripotent stem cells (iPSCs) and various specialized cells directly from somatic cells of different species. Reprogramming techniques conventionally use viral vectors encoding transcription factors to induce fate conversion. However, the introduction of transgenes limits the therapeutic applications of the reprogrammed cells. To overcome safety-related concerns, small molecules offer some advantages over the existing methods for the control of gene expression and induction of cell fate conversion. Technical advances in optimizing concentrations, durations, structures, and combinations of small molecules make chemical reprogramming a safe and feasible method. This review provides a concise overview of cutting-edge findings regarding chemical-only reprogramming as one of the integration-free approaches to iPSC generation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Anokye-Danso F, Trivedi C M, Juhr D, Gupta M, Cui Z, Tian Y, Zhang Y, Yang W, Gruber P J, Epstein J A, Morrisey E E (2011). Highly efficient miRNA-mediated reprogramming of mouse and human somatic cells to pluripotency. Cell Stem Cell, 8(4): 376–388
Babos K, Ichida J K (2015). Small molecules take a big step by converting fibroblasts into neurons. Cell Stem Cell, 17(2): 127–129
Ban H, Nishishita N, Fusaki N, Tabata T, Saeki K, Shikamura M, Takada N, Inoue M, Hasegawa M, Kawamata S, Nishikawa S (2011). Efficient generation of transgene-free human induced pluripotent stem cells (iPSCs) by temperature-sensitive Sendai virus vectors. Proc Natl Acad Sci USA, 108(34): 14234–14239
Cahan P, Li H, Morris S A, Lummertz da Rocha E, Daley G Q, Collins J J (2014). Cell net: Network biology applied to stem cell engineering. Cell, 158(4): 903–915
Chen J K, Liu H, Liu J, Qi J, Wei B, Yang J Q, Liang H Q, Chen Y, Chen J, Wu Y R, Guo L, Zhu J Y, Zhao X J, Peng T R, Zhang Y X, Chen S, Li X J, Li D W, Wang T, Pei D Q (2013). H3K9 methylation is a barrier during somatic cell reprogramming into iPSCs. Nat Genet, 45 (1): 34–U62
Cheng L, Gao L, Guan W, Mao J, Hu W, Qiu B, Zhao J, Yu Y, Pei G (2015). Direct conversion of astrocytes into neuronal cells by drug cocktail. Cell Res, 25(11): 1269–1272
Cheng L, Hu W, Qiu B, Zhao J, Yu Y, Guan W, Wang M, Yang W, Pei G (2014). Generation of neural progenitor cells by chemical cocktails and hypoxia. Cell Res, 24(6): 665–679
Chou B K, Gu H, Gao Y, Dowey S N, Wang Y, Shi J, Li Y, Ye Z, Cheng T, Cheng L (2015). A facile method to establish human induced pluripotent stem cells from adult blood cells under feeder-free and xeno-free culture conditions: a clinically compliant approach. Stem Cells Transl Med, 4(4): 320–332
Chou B K, Mali P, Huang X, Ye Z, Dowey S N, Resar L M, Zou C, Zhang Y A, Tong J, Cheng L (2011). Efficient human iPS cell derivation by a non-integrating plasmid from blood cells with unique epigenetic and gene expression signatures. Cell Res, 21(3): 518–529
Cyranoski D 2014. Japanese woman is first recipient of next-generation stem cells. Nature Publishing Group.
Davies S G, Kennewell P D, Russell A J, Seden P T, Westwood R, Wynne G M (2015). Stemistry: The control of stem cells in situ using chemistry. J Med Chem, 58(7): 2863–2894
Durruthy-Durruthy J, Briggs S F, Awe J, Ramathal C Y, Karumbayaram S, Lee P C, Heidmann J D, Clark A, Karakikes I, Loh K M, Wu J C, Hoffman A R, Byrne J, Reijo Pera R A, Sebastiano V (2014). Rapid and Efficient Conversion of Integration-Free Human Induced Pluripotent Stem Cells to GMP-Grade Culture Conditions. PLoS ONE, 9(4): e94231
Ebrahimi B (2015a). Reprogramming barriers and enhancers: strategies to enhance the efficiency and kinetics of induced pluripotency. Cell Regen (Lond), 4(1): 1–12
Ebrahimi B (2015b). Reprogramming of adult stem/progenitor cells into iPSCs without reprogramming factors. Journal of Medical Hypotheses and Ideas, 9(2): 99–103
Ebrahimi B (2016). Biological computational approaches: new hopes to improve (re)programming robustness, regenerative medicine and cancer therapeutics. Differentiation, doi: 10.1016/j.diff.2016.03.001
Eriksson P S, Perfilieva E, Bjork-Eriksson T, Alborn A M, Nordborg C, Peterson D A, Gage F H (1998). Neurogenesis in the adult human hippocampus. Nat Med, 4(11): 1313–1317
Esteban MA, Wang T, Qin B, Yang J, Qin D, Cai J, Li W, Weng Z, Chen J, Ni S, Chen K, Li Y, Liu X, Xu J, Zhang S, Li F, He W, Labuda K, Song Y, Peterbauer A, Wolbank S, Redl H, Zhong M, Cai D, Zeng L, Pei D (2010). Vitamin C enhances the generation of mouse and human induced pluripotent stem cells. Cell Stem Cell, 6(1): 71–79
Foster KW, Liu Z, Nail C D, Li X, Fitzgerald T J, Bailey S K, Frost A R, Louro I D, Townes T M, Paterson A J, Kudlow J E, Lobo-Ruppert S M, Ruppert J M (2005). Induction of KLF4 in basal keratinocytes blocks the proliferation-differentiation switch and initiates squamous epithelial dysplasia. Oncogene, 24(9): 1491–1500
Fu Y, Huang C, Xu X, Gu H, Ye Y, Jiang C, Qiu Z, Xie X (2015). Direct reprogramming of mouse fibroblasts into cardiomyocytes with chemical cocktails. Cell Res, 25(9): 1013–1024
Fusaki N, Ban H, Nishiyama A, Saeki K, Hasegawa M (2009). Efficient induction of transgene-free human pluripotent stem cells using a vector based on Sendai virus, an RNA virus that does not integrate into the host genome. Proc Jpn Acad, Ser B, Phys Biol Sci, 85(8): 348–362
Goh P A, Caxaria S, Casper C, Rosales C, Warner T T, Coffey P J, Nathwani A C (2013). A systematic evaluation of integration free reprogramming methods for deriving clinically relevant patient specific induced pluripotent stem (iPS) cells. PLoS ONE, 8(11): e81622
González F, Boué S, Belmonte J C I (2011). Methods for making induced pluripotent stem cells: reprogramming à la carte. Nat Rev Genet, 12(4): 231–242
He S, Guo Y, Zhang Y, Li Y, Feng C, Li X, Lin L, Guo L, Wang H, Liu C, Zheng Y, Luo C, Liu Q, Wang F, Sun H, Liang L, Li L, Su H, Chen J, Pei D, Zheng H (2015). Reprogramming somatic cells to cells with neuronal characteristics by defined medium both in vitro and in vivo. Cell Regen (Lond), 4(1): 1–9
Higuchi A, Ling Q D, Kumar S S, Munusamy M A, Alarfaj A A, Chang Y, Kao S H, Lin K C, Wang H C, Umezawa A (2015). Generation of pluripotent stem cells without the use of genetic material. Lab Invest, 95(1): 26–42
Hochedlinger K, Yamada Y, Beard C, Jaenisch R (2005). Ectopic expression of Oct-4 blocks progenitor-cell differentiation and causes dysplasia in epithelial tissues. Cell, 121(3): 465–477
Hou P, Li Y, Zhang X, Liu C, Guan J, Li H, Zhao T, Ye J, Yang W, Liu K, Ge J, Xu J, Zhang Q, Zhao Y, Deng H (2013). Pluripotent stem cells induced from mouse somatic cells by small-molecule compounds. Science, 341(6146): 651–654
Hu W, Qiu B, Guan W, Wang Q, Wang M, Li W, Gao L, Shen L, Huang Y, Xie G, Zhao H, Jin Y, Tang B, Yu Y, Zhao J, Pei G (2015). Direct conversion of normal and alzheimer’s disease human fibroblasts into neuronal cells by small molecules. Cell Stem Cell, 17(2): 204–212
Ichida J K, Blanchard J, Lam K, Son E Y, Chung J E, Egli D, Loh K M, Carter A C, Di Giorgio F P, Koszka K, Huangfu D, Akutsu H, Liu D R, Rubin L L, Eggan K (2009). A small-molecule inhibitor of tgf- Beta signaling replaces sox2 in reprogramming by inducing nanog. Cell Stem Cell, 5(5): 491–503
Jung D W, Kim W H, Williams D R (2014). Reprogram or reboot: small molecule approaches for the production of induced pluripotent stem cells and direct cell reprogramming. ACS Chem Biol, 9(1): 80–95
Kaji K, Norrby K, Paca A, Mileikovsky M, Mohseni P, Woltjen K (2009). Virus-free induction of pluripotency and subsequent excision of reprogramming factors. Nature, 458(7239): 771–775
Kim D, Kim C H, Moon J I, Chung Y G, Chang M Y, Han B S, Ko S, Yang E, Cha K Y, Lanza R, Kim K S (2009). Generation of human induced pluripotent stem cells by direct delivery of reprogramming proteins. Cell Stem Cell, 4(6): 472–476
Koyanagi-Aoi M, Ohnuki M, Takahashi K, Okita K, Noma H, Sawamura Y, Teramoto I, Narita M, Sato Y, Ichisaka T, Amano N, Watanabe A, Morizane A, Yamada Y, Sato T, Takahashi J, Yamanaka S (2013). Differentiation-defective phenotypes revealed by largescale analyses of human pluripotent stem cells. Proc Natl Acad Sci USA, 110(51): 20569–20574
Lee A S, Tang C, Rao M S, Weissman I L, Wu J C (2013). Tumorigenicity as a clinical hurdle for pluripotent stem cell therapies. Nat Med, 19(8): 998–1004
Lee J, Sayed N, Hunter A, Au K F,WongWH, Mocarski E S, Pera R R, Yakubov E, Cooke J P (2012). Activation of Innate Immunity Is Required for Efficient Nuclear Reprogramming. Cell, 151(3): 547–558
Li W, Jiang K, Ding S (2012a). Concise review: A chemical approach to control cell fate and function. Stem Cells, 30(1): 61–68
Li W, Jiang K, Wei W, Shi Y, Ding S (2012a). Chemical approaches to studying stem cell biology. Cell Res, 23(1): 81–91
Li W, Li K, Wei W, Ding S (2012b). Chemical approaches to stem cell biology and therapeutics. Cell Stem Cell, 13(3): 270–283
Li W, Tian E, Chen Z X, Sun G, Ye P, Yang S, Lu D, Xie J, Ho T V, Tsark W M, Wang C, Horne D A, Riggs A D, Yip M L, Shi Y (2012b). Identification of Oct4-activating compounds that enhance reprogramming efficiency. Proc Natl Acad Sci USA, 109(51): 20853–20858
Li X, Zuo X, Jing J, Ma Y, Wang J, Liu D, Zhu J, Du X, Xiong L, Du Y, Xu J, Xiao X, Wang J, Chai Z, Zhao Y, Deng H (2015). Smallmolecule- driven direct reprogramming of mouse fibroblasts into functional neurons. Cell Stem Cell, 17(2): 195–203
Li Y, Zhang Q, Yin X, Yang W, Du Y, Hou P, Ge J, Liu C, Zhang W, Zhang X, Wu Y, Li H, Liu K, Wu C, Song Z, Zhao Y, Shi Y, Deng H (2011). Generation of iPSCs from mouse fibroblasts with a single gene, Oct4, and small molecules. Cell Res, 21(1): 196–204
Lin T, Ambasudhan R, Yuan X, Li W, Hilcove S, Abujarour R, Lin X, Hahm H S, Hao E, Hayek A, Ding S (2009). A chemical platform for improved induction of human iPSCs. Nat Methods, 6(11): 805–808
Lin T, Wu S (2015). Reprogramming with Small Molecules instead of Exogenous Transcription Factors. Stem Cells Int, 2015: 794632
Long Y, Wang M, Gu H, Xie X (2015). Bromodeoxyuridine promotes full-chemical induction of mouse pluripotent stem cells. Cell Res, 25(10): 1171–1174
Lu X, Zhao T (2013). Clinical Therapy Using iPSCs: Hopes and Challenges. Genomics Proteomics Bioinformatics, 11(5): 294–298
Ma H, Morey R, O’Neil R C, He Y, Daughtry B, SchultzMD, Hariharan M, Nery J R, Castanon R, Sabatini K, Thiagarajan R D, Tachibana M, Kang E, Tippner-Hedges R, Ahmed R, Gutierrez NM, Van Dyken C, Polat A, Sugawara A, Sparman M, Gokhale S, Amato P, Wolf P, Ecker D, Laurent L C, Mitalipov S (2014). Abnormalities in human pluripotent cells due to reprogramming mechanisms. Nature, 511: 177–183
Masuda S, Wu J, Hishida T, Pandian G N, Sugiyama H, Izpisua Belmonte J C (2013). Chemically induced pluripotent stem cells (CiPSCs): a transgene-free approach. J Mol Cell Biol, 5(5): 354–355
Morris S A, Cahan P, Li H, Zhao AM, San Roman A K, Shivdasani R A, Collins J J, Daley G Q (2014). Dissecting Engineered Cell Types and Enhancing Cell Fate Conversion via CellNet. Cell, 158(4): 889–902
Ohnishi K, Semi K, Yamamoto T, Shimizu M, Tanaka A, Mitsunaga K, Okita K, Osafune K, Arioka Y, Maeda T, Soejima H, Moriwaki H, Yamanaka S, Woltjen K, Yamada Y (2014). Premature Termination of Reprogramming In Vivo Leads to Cancer Development through Altered Epigenetic Regulation. Cell, 156(4): 663–677
Okano H, Nakamura M, Yoshida K, Okada Y, Tsuji O, Nori S, Ikeda E, Yamanaka S, Miura K (2013). Steps toward safe cell therapy using induced pluripotent stem cells. Circ Res, 112(3): 523–533
Okita K, Ichisaka T, Yamanaka S (2007). Generation of germlinecompetent induced pluripotent stem cells. Nature, 448(7151): 313–317
Okita K, Nakagawa M, Hyenjong H, Ichisaka T, Yamanaka S (2008). Generation of mouse induced pluripotent stem cells without viral vectors. Science, 322(5903): 949–953
Okita K, Yamakawa T, Matsumura Y, Sato Y, Amano N, Watanabe A, Goshima N, Yamanaka S (2013). An efficient nonviral method to generate integration-free human-induced pluripotent stem cells from cord blood and peripheral blood cells. Stem Cells, 31(3): 458–466
Okita K, Yamanaka S (2011). Induced pluripotent stem cells: opportunities and challenges. Philos Trans R Soc Lond B Biol Sci, 366(1575): 2198–2207
Onder T T, Kara N, Cherry A, Sinha A U, Zhu N, Bernt K M, Cahan P, Marcarci B O, Unternaehrer J, Gupta P B, Lander E S, Armstrong S A, Daley G Q (2012). Chromatin-modifying enzymes as modulators of reprogramming. Nature, 483(7391): 598–602
Pandian G N, Nakano Y, Sato S, Morinaga H, Bando T, Nagase H, Sugiyama H (2012). A synthetic small molecule for rapid induction of multiple pluripotency genes in mouse embryonic fibroblasts. Sci Rep, 2: 544
Pennarossa G, Maffei S, Campagnol M, Tarantini L, Gandolfi F, Brevini T A (2013). Brief demethylation step allows the conversion of adult human skin fibroblasts into insulin-secreting cells. Proc Natl Acad Sci USA, 110(22): 8948–8953
Piao Y, Hung S S, Lim S Y, Wong R C, Ko M S (2014). Efficient generation of integration-free human induced pluripotent stem cells from keratinocytes by simple transfection of episomal vectors. Stem Cells Transl Med, 3(7): 787–791
Sayed N, Wong W T, Ospino F, Meng S, Lee J, Jha A, Dexheimer P, Aronow B J, Cooke J P (2015). Transdifferentiation of human fibroblasts to endothelial cells: role of innate immunity. Circulation, 131(3): 300–309
Schlaeger T M, Daheron L, Brickler T R, Entwisle S, Chan K, Cianci A, DeVine A, Ettenger A, Fitzgerald K, Godfrey M, Gupta D, McPherson J, Malwadkar P, Gupta M, Bell B, Doi A, Jung N, Li X, Lynes MS, Brookes E, Cherry A B C, Demirbas D, Tsankov AM, Zon L I, Rubin L L, Feinberg A P, Meissner A, Cowan C A, Daley G Q (2015). A comparison of non-integrating reprogramming methods. Nat Biotechnol, 33(1): 58–63
Shi Y, Desponts C, Do J T, Hahm H S, Scholer H R, Ding S (2008). Induction of pluripotent stem cells from mouse embryonic fibroblasts by Oct4 and Klf4 with small-molecule compounds. Cell Stem Cell, 3(5): 568–574
Shu J, Wu C, Wu Y, Li Z, Shao S, Zhao W, Tang X, Yang H, Shen L, Zuo X, Yang W, Shi Y, Chi X, Zhang H, Gao G, Shu Y, Yuan K, He W, Tang C, Zhao Y, Deng H (2013). Induction of pluripotency in mouse somatic cells with lineage specifiers. Cell, 153(5): 963–975
Silva M, Daheron L, Hurley H, Bure K, Barker R, Carr A J, Williams D, Kim HW, French A, Coffey P J, Cooper-White J J, Reeve B, Rao M, Snyder E Y, Ng K S, Mead B E, Smith J A, Karp J M, Brindley D A, Wall I (2015). Generating iPSCs: Translating Cell Reprogramming Science into Scalable and Robust Biomanufacturing Strategies. Cell Stem Cell, 16(1): 13–17
Soldner F, Hockemeyer D, Beard C, Gao Q, Bell G W, Cook E G, Hargus G, Blak A, Cooper O, Mitalipova M, Isacson O, Jaenisch R (2009). Parkinson’s disease patient-derived induced pluripotent stem cells free of viral reprogramming factors. Cell, 136(5): 964–977
Stadtfeld M, Apostolou E, Ferrari F, Choi J, Walsh R M, Chen T, Ooi S S, Kim S Y, Bestor T H, Shioda T, Park P J, Hochedlinger K, (2012). Ascorbic acid prevents loss of Dlk1-Dio3 imprinting and facilitates generation of all-iPS cell mice from terminally differentiated B cells. Nat Genet, 44: 398–405, S391–392
Stadtfeld M, Nagaya M, Utikal J, Weir G, Hochedlinger K (2008). Induced pluripotent stem cells generated without viral integration. Science, 322(5903): 945–949
Su J B, Pei D Q, Qin B M (2013). Roles of small molecules in somatic cell reprogramming. Acta Pharmacol Sin, 34(6): 719–724
Sugiura M, Kasama Y, Araki R, Hoki Y, Sunayama M, Uda M, Nakamura M, Ando S, Abe M (2014). Induced Pluripotent Stem Cell Generation-Associated Point Mutations Arise during the Initial Stages of the Conversion of These Cells. Stem Cell Rep, 2(1): 52–63
Takahashi K, Tanabe K, Ohnuki M, Narita M, Sasaki A, Yamamoto M, Nakamura M, Sutou K, Osafune K, Yamanaka S (2014). Induction of pluripotency in human somatic cells via a transient state resembling primitive streak-like mesendoderm. Nat Commun, 5: 3678
Takahashi K, Yamanaka S (2006). Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell, 126(4): 663–676
Tomaru Y, Hasegawa R, Suzuki T, Sato T, Kubosaki A, Suzuki M, Kawaji H, Forrest A R R, Hayashizaki Y, Consortium F, Shin J W, Suzuki H (2014). A transient disruption of fibroblastic transcriptional regulatory network facilitates trans-differentiation. Nucleic Acids Res, 42(14): 8905–8913
Valamehr B, Robinson M, Abujarour R, Rezner B, Vranceanu F, Le T, Medcalf A, Lee T T, Fitch M, Robbins D, Flynn P (2014). Platform for Induction and Maintenance of Transgene-free hiPSCs Resembling Ground State Pluripotent Stem Cells. Stem Cell Rep, 2(3): 366–381
Vidal S E, Amlani B, Chen T, Tsirigos A, Stadtfeld M (2014). Combinatorial Modulation of Signaling Pathways Reveals Cell- Type-Specific Requirements for Highly Efficient and Synchronous iPSC Reprogramming. Stem Cell Rep, 3(4): 574–584
Wang T, Chen K, Zeng X, Yang J, Wu Y, Shi X, Qin B, Zeng L, Esteban M A, Pan G, Pei D (2011). The histone demethylases Jhdm1a/1b enhance somatic cell reprogramming in a vitamin-C-dependent manner. Cell Stem Cell, 9(6): 575–587
Warren L, Manos P D, Ahfeldt T, Loh Y H, Li H, Lau F, Ebina W, Mandal P K, Smith Z D, Meissner A, Daley G Q, Brack A S, Collins J J, Cowan C, Schlaeger T M, Rossi D J (2010). Highly efficient reprogramming to pluripotency and directed differentiation of human cells with synthetic modified mRNA. Cell Stem Cell, 7(5): 618–630
Woltjen K, Michael I P, Mohseni P, Desai R, Mileikovsky M, Hamalainen R, Cowling R, Wang W, Liu P, Gertsenstein M, Kaji K, Sung H K, Nagy A (2009). piggyBac transposition reprograms fibroblasts to induced pluripotent stem cells. Nature, 458(7239): 766–770
Xu J, Du Y, Deng H (2015). Direct Lineage Reprogramming: Strategies, Mechanisms, and Applications. Cell Stem Cell, 16(2): 119–134
Ye J, Ge J, Zhang X, Cheng L, Zhang Z, He S, Wang Y, Lin H, Yang W, Liu J, Zhao Y, Deng H (2016). Pluripotent stem cells induced from mouse neural stem cells and small intestinal epithelial cells by small molecule compounds. Cell Res, 26(1): 34–45
Yu C, Liu K, Tang S, Ding S (2014). Chemical approaches to cell reprogramming. Curr Opin Genet Dev, 28: 50–56
Yu J, Chau K F, VodyanikMA, Jiang J, Jiang Y (2011). Efficient feederfree episomal reprogramming with small molecules. PLoS ONE, 6(3): e17557
Yu J, Hu K, Smuga-Otto K, Tian S, Stewart R, Slukvin I I, Thomson J A (2009). Human induced pluripotent stem cells free of vector and transgene sequences. Science, 324(5928): 797–801
Zhang L, Yin J C, Yeh H, Ma N X, Lee G, Chen X A, Wang Y, Lin L, Chen L, Jin P, Wu G Y, Chen G (2015). Small Molecules Efficiently Reprogram Human Astroglial Cells into Functional Neurons. Cell Stem Cell, 17(6): 735–747
Zhao Y, Zhao T, Guan J, Zhang X, Fu Y, Ye J, Zhu J, Meng G, Ge J, Yang S, Cheng L, Du Y, Zhao C, Wang T, Su L, Yang W, Deng H (2015). A XEN-like State Bridges Somatic Cells to Pluripotency during Chemical Reprogramming. Cell, 163(7): 1678–1691
Zhou H, Wu S, Joo J Y, Zhu S, Han DW, Lin T, Trauger S, Bien G, Yao S, Zhu Y, Siuzdak G, Scholer H R, Duan L, Ding S (2009). Generation of induced pluripotent stem cells using recombinant proteins. Cell Stem Cell, 4(5): 381–384
Zhou Y y, Zeng F (2013). Integration-free Methods for Generating Induced Pluripotent Stem Cells. Genomics Proteomics Bioinformatics, 11(5): 284–287
Zhu S, Li W, Zhou H, Wei W, Ambasudhan R, Lin T, Kim J, Zhang K, Ding S (2010). Reprogramming of human primary somatic cells by OCT4 and chemical compounds. Cell Stem Cell, 7(6): 651–655
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ebrahimi, B. Chemical-only reprogramming to pluripotency. Front. Biol. 11, 75–84 (2016). https://doi.org/10.1007/s11515-016-1396-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11515-016-1396-0