Abstract
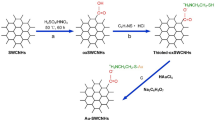
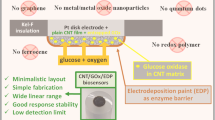
Carbon nanotubes (CNTs) and reduced graphene oxide (rGO) nanosheets were utilized to construct glucose biosensors in combination with gold nanowire arrays (AuNWAs), and microfluidic injection analysis driven by gravity force was used to investigate the performances of as-prepared glucose biosensors. The results demonstrated glucose biosensors based on carbon nanomaterials/AuNWAs presented excellent performance at low working potential of −0.2 V versus Ag/AgCl (3 mol/L KCl), such as high sensitivity, good anti-interference ability and high throughput (45 h−1). The glucose biosensor based on glucose oxidase (GOx)–CNT–AuNWAs showed a wide linear range from 100 to 3,000 µmol/L with a sensitivity of 4.12 µA/cm2 mmol/L. Furthermore, the linear range and sensitivity of GOx–rGO–AuNWAs-based glucose biosensor were 50–4,000 µmol/L and 8.59 µA/(cm2 mmol/L), respectively, which were better than those of glucose biosensor based on GOx–CNT–AuNWAs, suggesting rGO nanosheets in combination with AuNWAs being a good platform for the construction of glucose biosensors.






Similar content being viewed by others
References
Srinivasan V, Pamula VK, Fair RB (2004) Droplet-based microfluidic lab-on-a-chip for glucose detection. Anal Chim Acta 507:145–150
Wang JX, Sun X, Wei A et al (2006) Zinc oxide nanocomb biosensor for glucose detection. Appl Phys Lett 88:233106
Wang J (2008) Electrochemical glucose biosensors. Chem Rev 108:814–825
Wang HC, Wang XS, Zhang XQ et al (2009) A novel glucose biosensor based on the immobilization of glucose oxidase onto gold nanoparticles-modified Pb nanowires. Biosens Bioelectron 25:142–146
Pradhan D, Niroui F, Leung KT (2010) High-performance, flexible enzymatic glucose biosensor based on ZnO nanowires supported on a gold-coated polyester substrate. ACS Appl Mater Interfaces 2:2409–2412
Shi WT, Ma ZF (2010) Amperometric glucose biosensor based on a triangular silver nanoprisms/chitosan composite film as immobilization matrix. Biosens Bioeletron 26:1098–1103
Xu GQ, Adeloju SB, Wu YC et al (2012) Modification of polypyrrole nanowires array with platinum nanoparticles and glucose oxidase for fabrication of a novel glucose biosensor. Anal Chim Acta 755:100–107
Tang J, Wang YC, Li J et al (2014) Sensitive enzymatic glucose detection by TiO2 nanowire photoelectrochemical biosensors. J Mater Chem A 2:6153–6157
Lin YH, Lu F, Tu Y et al (2004) Glucose biosensors based on carbon nanotube nanoelectrode ensembles. Nano Lett 4:191–195
Lim SH, Wei J, Lin JY et al (2005) A glucose biosensor based on electrodeposition of palladium nanoparticles and glucose oxidase onto Nafion-solubilized carbon nanotube electrode. Biosens Bioeletron 20:2341–2346
Liu GD, Lin YH (2006) Amperometric glucose biosensor based on self-assembling glucose oxidase on carbon nanotubes. Electrochem Commun 8:251–256
Li G, Liao JM, Hu GQ et al (2005) Study of carbon nanotube modified biosensor for monitoring total cholesterol in blood. Biosens Bioelectron 20:2140–2144
Dey RS, Raj CR (2010) Development of an amperometric cholesterol biosensor based on graphene–Pt nanoparticle hybrid material. J Phys Chem C 114:21427–21433
Bo Y, Yang HY, Hu Y et al (2011) A novel electrochemical DNA biosensor based on graphene and polyaniline nanowires. Electrochim Acta 56:2676–2681
Wang J, Musameh M, Lin YH (2003) Solubilization of carbon nanotubes by Nafion toward the preparation of amperometric biosensors. J Am Chem Soc 125:2408–2409
Wang J, Musameh M (2003) Carbon nanotube/teflon composite electrochemical sensors and biosensors. Anal Chem 75:2075–2079
Liu Y, Wang MK, Zhao F et al (2005) The direct electron transfer of glucose oxidase and glucose biosensor based on carbon nanotubes/chitosan matrix. Biosens Bioelectron 21:984–988
Shan CS, Yang HF, Song JF et al (2009) Direct electrochemistry of glucose oxidase and biosensing for glucose based on grapheme. Anal Chem 81:2378–2382
Kang XH, Wang J, Wu H et al (2009) Glucose oxidase-graphene-chitosan modified electrode for direct electrochemistry and glucose sensing. Biosens Bioelectron 25:901–905
Wang Y, Shao YY, Matson DW et al (2010) Nitrogen-doped graphene and its application in electrochemical biosensing. ACS Nano 4:1790–1798
Jose MV, Marx S, Murata H et al (2012) Direct electron transfer in a mediator-free glucose oxidase-based carbon nanotube-coated biosensor. Carbon 50:4010–4020
Nien PC, Chen PY, Hsu CY et al (2011) On-chip glucose biosensor based on enzyme entrapment with pre-reaction to lower interference in a flow injection system. Sens Actuator B Chem 157:64–71
Lankelma J, Nie ZH, Carrilho E et al (2012) Paper-based analytical device for electrochemical flow-injection analysis of glucose in urine. Anal Chem 84:4147–4152
Yu JH, Wang SM, Ge L et al (2011) A novel chemiluminescence paper microfluidic biosensor based on enzymatic reaction for uric acid determination. Biosens Bioelectron 26:3284–3289
Cui JW, Adeloju SB, Wu YC (2014) Integration of a highly ordered gold nanowires array with glucose oxidase for ultra-sensitive glucose detection. Anal Chim Acta 809:134–140
Hummers WS, Offeman RE (1958) Preparation of graphitic oxide. J Am Chem Soc 80:1339
Stankovich S, Dikin DA, Piner RD et al (2007) Synthesis of graphene-based nanosheets via chemical reduction of exfoliated graphite oxide. Carbon 45:1558–1565
Jiang XY, Wu YH, Mao XY et al (2011) Amperometric glucose biosensor based on integration of glucose oxidase with platinum nanoparticles/ordered mesoporous carbon nanocomposite. Sens Actuator B Chem 153:158–163
Choi BG, Im J, Kim HS et al (2011) Flow-injection amperometric glucose biosensors based on graphene/Nafion hybrid electrodes. Electrochim Acta 56:9721–9726
Jang HD, Kim SK, Chang H et al (2012) A glucose biosensor based on TiO2–graphene composite. Biosens Bioelectron 38:184–188
Unnikrishnan B, Palanisamy S, Chen SM (2013) A simple electrochemical approach to fabricate a glucose biosensor based on graphene–glucose oxidase biocomposite. Biosens Bioelectron 39:70–75
Nenkova R, Ivanova D, Vladimirova J et al (2010) New amperometric glucose biosensor based on cross-linking of glucose oxidase on silica gel/multiwalled carbon nanotubes/polyacrylonitrile nanocomposite film. Sens Actuator B Chem 148:59–65
Zeng Q, Cheng JS, Liu XF et al (2011) Palladium nanoparticle/chitosan-grafted graphene nanocomposites for construction of a glucose biosensor. Biosens Bioelectron 26:3456–3463
Lineweaver H, Burk D (1934) The determination of enzyme dissociation constants. J Am Chem Soc 56:658–666
Kandimalla VB, Tripathi VS, Ju HX (2006) A conductive ormosil encapsulated with ferrocene conjugate and multiwall carbon nanotubes for biosensing application. Biomaterials 27:1167–1174
Manso J, Mena ML, Yáñez-Sedeño P et al (2007) Electrochemical biosensors based on colloidal gold-carbon nanotubes composite electrodes. J Electroanal Chem 603:1–7
Zou HJ, Xiang CL, Sun LX et al (2008) Glucose biosensor based on electrodeposition of platinum nanoparticles onto carbon nanotubes and immobilizing enzyme with chitosan–SiO2 sol–gel. Biosens Bioelectron 23:1010–1016
Ali MA, Srivastava S, Solanki PR et al (2013) Highly efficient bienzyme functionalized nanocomposite-based microfluidics biosensor platform for biomedical application. Sci Rep 3:2661
Acknowledgments
This work was supported by the National Natural Science Foundation of China (51272063 and 51402081), the Natural Science Foundation of Anhui Province (1408085QB42), the Fundamental Research Funds for the Central Universities (JZ2014HGBZ0010) and the Science and Technology Planning Project of Tongling (2015CXZX02).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Qin, Y., Cui, J., Zhang, Y. et al. Integration of microfluidic injection analysis with carbon nanomaterials/gold nanowire arrays-based biosensors for glucose detection. Sci. Bull. 61, 473–480 (2016). https://doi.org/10.1007/s11434-016-1013-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-016-1013-2