Abstract
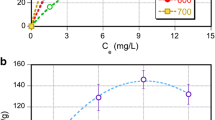
The effect of microstructure and surface chemistry on nicotine adsorption capacity of almond-shell-based activated carbon was studied. Almond-shell-based activated carbon was reactivated or modified with steam, ZnCl2 and NaHSO4 separately. The surface area and pore structure parameters of activated carbon were determined from the adsorption-desorption isotherm of nitrogen at 77 K. Boehm titration and FTIR were explored to determine the surface oxygen groups. The results revealed that nicotine adsorption from solution on carbons depend on the micropores and phenolic groups. A strong correlation between the amounts of adsorbed nicotine and the phenolic groups was observed, whereas the higher amounts of carboxylic groups decrease the nicotine adsorption capacity on activated carbons.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Lipowicz P J, Piadé J J. Evaporation and subsequent deposition of nicotine from minstream cigarette smoke in a denuder tube. J Aerosol Sci, 2004, 435: 33–45
Chen Z, Zhang L, Tang Y, et al. Adsorption of nicotine and tar from the mainstream smoke of cigarettes by oxidized carbon nanotubes. Appl Surf Sci, 2006, 252: 2933–2937
Rakić V, Damjanović L, Rac V, et al. The adsorption of nicotine from aqueous solution on different zeolite structure. Water Res, 2010, 44: 2047–2057
Lazarevic N, Adnadjevic B, Jovanovic J. Adsorption of nicotine from aqueous solution onto hydrophobic zeolite type USY. Appl Surf Sci, 2011, 257: 8017–8023
Yang J L, Zhou J B. Adsorption of nicotine from aqueous solution by activated carbons prepared from Chinese fir sawdust. Acta Phys Chim Sin, 2013, 29: 377–384
Demiral H, Demiral İ, Karabacakoğlu B, et al. Production of activated carbon from olive bagasse by physical activation. Chem Eng Res Des, 2011, 89: 206–213
Nowicki P, Wachowska H, Pietrzak R. Active carbons prepared by chemical activation of plum stones and their application in removal of NO2. J Hazardous Mater, 2010, 181: 1088–1094
Yang H, Yan R, Chen H, et al. Characteristics of hemicellulose, cellulose and lignin pyrolysis. Fuel, 2007, 86: 1781–1788
Arami-Niya A, Daud W M A W, Mjalli F S. Comparative study of the textural characteristics of oil palm shell activated carbon produced by chemical and physical activation for methane adsorption. Chem Eng Res Des, 2011, 89: 657–664
Mourão P A M, Laginhas C, Custódio F, et al. Influence of oxidation process on the adsorption capacity of activated carbons from lignocellulosic precursors. Fuel Process Technol, 2011, 92: 241–246
Shangguan J, Li C, Miao M, et al. Surface characterization and SO2 removal activity of activated semi-coke with heat treatment. New Carbon Mater, 2008, 23: 37–43
Wang H, Kang J, Liu H, et al. Preparation of organically functionalized silica gel as adsorbent for copper ion adsorption. J Environ Sci, 2009, 21: 1473–1479
Hanafiah M A K M, Ngah W S W, Zolkafly S H, et al. Acid Blue 25 adsorption on base treated Shorea dasyphylla sawdust: Kinetic, isotherm, thermodynamic and spectroscopic analysis. J Environ Sci, 2012, 24: 261–268
Dandekar A, Baker R T K, Vannice M A. Characterization of activated carbon, graphitized carbon fibers and synthetic diamond power using TPD and DRIFTS. Carbon, 1998, 36: 1821–1831
Ishizaki C, Marti I. Surface oxide structures on a commercial activated carbon. Carbon, 1981, 19: 409–412
Wang C P, Wang B L, Liu J T, et al. Adsorption of Cd(II) from acidic aqueous solutions by tourmaline as a novel material. Chin Sci Bull, 2012, 57: 3218–3225
Su W, Zhou Y, Wei L, et al. Effect of microstructure and surface modification on the hydrogen adsorption capacity of active carbons. New Carbon Mater, 2007, 22: 135–140
Skodras G, Diamantopoulou I, Sakellaropoulos G P. Role of activated carbon structural properties and surface chemistry in mercury adsorption. Desalination, 2007, 210: 281–286
Salame I I, Bandosz T J. Role of surface chemistry in adsorption of phenol on activated carbon. J Colloid Interf Sci, 2003, 264: 307–312
Figueiredo J L, Mahata N, Pereira M F R, et al. Adsorption of phenol on supercritically activated carbon fibres: Effect of texture and surface chemistry. J Colloid Interf Sci, 2011, 357: 210–214
Daifullah A A M, Girgis B S. Impact of surface characteristics of activated carbon on adsorption of BTEX. Colloid Surf A: Physicochem Eng Aspects, 2003, 214: 181–193
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Yang, J., Fang, F. & Zhou, J. Effect of microstructure and surface chemistry in liquid-phase adsorptive nicotine by almond-shell-based activated carbon. Chin. Sci. Bull. 58, 3715–3720 (2013). https://doi.org/10.1007/s11434-013-6040-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-013-6040-7