Abstract
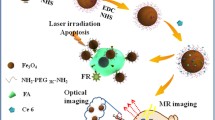
Construction of multifunctional/multimodality nanoparticles for cancer diagnosis and therapy has become an attractive area of investigation. In this report, we designed a multimodality nanoprobe for cell labeling, and can be detectable by both magnetic resonance and near infrared (NIR) fluorescence imaging. Multiple hydrophobic superparamagnetic iron oxide (SPIO) nanocrystals are self-assembled into nanocomposites in water phase with the help of partially alkylated hyperbranched polycation, polyethylenimine (PEI), which already conjugated with the indocyanine dye Cy5.5 and can be used for cell imaging under NIR fluorescence imaging. The amphiphilic PEI/SPIO nanocomposites have a strong T 2 relaxivity. The iron uptake process in MCF-7/Adr displays a time dependent behavior. Confocal laser scanning microscopy reveals that the nanoprobes are internalized into the cytoplasm of MCF-7/Adr after 24 h labeling. Both MR and NIR fluorescence imaging showed strong image contrast against unlabeled cells. Under a clinical MRI scanner, labeled cells in gelatin phantom present much darker images than controlled ones. The T 2 relaxation rate of the labeled cells is 98.2 s−1, significantly higher than that of the control ones of 2.3 s−1. This study provides an important alternative to label MCF-7/Adr at optimized low dosages with high efficiency, and may be useful to label other biologically important cells and track their behaviors in vivo.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Brigger I, Dubernet C, Couvreur P. Nanoparticles in cancer therapy and diagnosis. Adv Drug Deliv Rev, 2002, 54: 631–651
Ferrari M. Cancer nanotechnology: Opportunities and challenges. Nat Rev Cancer, 2005, 5: 161–171
Peer D, Karp J M, Hong S, et al. Nanocarriers as an emerging platform for cancer therapy. Nat Nanotechnol, 2007, 2: 751–760
Shuai X T, Ai H, Nasongkla N, et al. Micellar carriers based on block copolymers of poly(e-caprolactone) and poly(ethylene glycol) for doxorubicin delivery. J Control Release, 2004, 98: 415–426
Wood K C, Azarin S M, Arap W, et al. Tumor-targeted gene delivery using molecularly engineered hybrid polymers functionalized with a tumor-homing peptide. Bio Chem, 2008, 19: 403–405
Kozlowska D, Foran P, MacMahon P, et al. Molecular and magnetic resonance imaging: The value of immunoliposomes. Adv Drug Delivery Rev, 2009, 61: 1402–1411
Bulte J W M, Kraitchman D L. Iron oxide MR contrast agents for molecular and cellular imaging. NMR Biomed, 2004, 17: 484–499
Janib S M, Moses A S, MacKay J A. Imaging and drug delivery using theranostic nanoparticles. Adv Drug Delivery Rev, 2010, 62: 1052–1063
Rudin M, Weissleder R. Molecular imaging in drug discovery and development. Nat Rev Drug Discov, 2003, 2: 123–131
Willmann J K, van Bruggen N, Dinkelborg L M, et al. Molecular imaging in drug development. Nat Rev Drug Discovery, 2008, 7: 591–607
Ai H. Layer-by-layer capsules for magnetic resonance imaging and drug delivery. Adv Drug Delivery Rev, 2011, 63: 772–788
Ai H, Flask C, Weinberg B, et al. Magnetite-loaded polymeric micelles as ultrasensitive magnetic-resonance probes. Adv Mater, 2005, 17: 1949–1956
Wang Y X, Hussain S M, Krestin G P. Superparamagnetic iron oxide contrast agents: Physicochemical characteristics and applications in MR imaging. Eur Radiol, 2001, 11: 2319–2331
Thorek D L J, Chen A, Czupryna J, et al. Superparamagnetic iron oxide nanoparticle probes for molecular imaging. Ann Biomed Eng, 2006, 34: 23–38
Taktak S, Sosnovik D, Cima M J, et al. Multiparameter magnetic relaxation switch assays. Anal Chem, 2007, 79: 8863–8869
Liu G, Wang Z, Lu J, et al. Low molecular weight alkyl-polycation wrapped magnetite nanoparticle clusters as MRI probes for stem cell labeling and in vivo imaging. Biomaterials, 2011, 32: 528–537
Liu G, Xia C, Wang Z, et al. Magnetic resonance imaging probes for labeling of chondrocyte cells. J Mater Sci-Mater Med, 22: 601–606
Chen X Y, Conti P S, Moats R A. In vivo near-infrared fluorescence imaging of integrin αvβ3 in brain tumor xenografts. Cancer Res, 2004, 64: 8009–8014
Becker A, Hessenius C, Licha K, et al. Receptor-targeted optical imaging of tumors with near-infrared fluorescent ligands. Nat Biotechnol, 2001, 19: 327–331
Weissleder R, Tung C H, Mahmood U, et al. In vivo imaging of tumors with protease-activated near-infrared fluorescent probes. Nat Biotechnol, 1999, 17: 375–378
Veiseh O, Sun C, Gunn J, et al. Optical and MRI multifunctional nanoprobe for targeting gliomas. Nano Lett, 2005, 5: 1003–1008
Talanov V S, Regino C A S, Kobayashi H, et al. Dendrimer-based nanoprobe for dual modality magnetic resonance and fluorescence imaging. Nano Lett, 2006, 6: 1459–1463
Sun S H, Zeng H, Robinson D B, et al. Monodisperse MFe2O4 (M = Fe, Co, Mn) nanoparticles. J Am Chem Soc, 2004, 126: 273–279
Wang Q, Su H, Xia C, et al. Amphiphilic dextran/magnetite nanocomposites as magnetic resonance imaging probes. Chin Sci Bull, 2009, 54: 2925–2933
Kramer M, Perignon N, Haag R, et al. Water-soluble dendritic architectures with carbohydrate shells for the templation and stabilization of catalytically active metal nanoparticles. Macromolecules, 2005, 38: 8308–8315
Cheng Z, Levi J, Xiong Z M, et al. Near-infrared fluorescent deoxyglucose analogue for tumor optical imaging in cell culture and living mice. Bioconj Chem, 2006, 17: 662–669
Zhiyong W, Gang L, Jiayu S, et al. Self-assembly of magnetite nanocrystals with amphiphilic polyethylenimine: Structures and applications in magnetic resonance imaging. J Nanosci Nanotechnol, 2009, 9: 378–385
Wilson D L, Kump K S, Eppell S J, et al. Morphological restoration of atomic-force microscopy imaging. Langmuir, 1995, 11: 265–272
Kircher M F, Allport J R, Graves E E, et al. In vivo high resolution three-dimensional imaging of antigen-specific cytotoxic T-lymphocyte trafficking to tumors. Cancer Res, 2003, 63: 6838–6846
Song H T, Choi J S, Huh Y M, et al. Surface modulation of magnetic nanocrystals in the development of highly efficient magnetic resonance probes for intracellular labeling. J Am Chem Soc, 2005, 127: 9992–9993
Ntziachristos V, Bremer C, Weissleder R. Fluorescence imaging with near-infrared light: New technological advances that enable in vivo molecular imaging. Eur Radio, 2003, 13: 195–208
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Wang, D., Su, H., Liu, Y. et al. Near-infrared fluorescent amphiphilic polycation wrapped magnetite nanoparticles as multimodality probes. Chin. Sci. Bull. 57, 4012–4018 (2012). https://doi.org/10.1007/s11434-012-5073-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-012-5073-7