Abstract
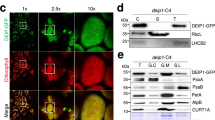
Deg1, a thylakoid lumen-localized protease, retains both chaperone and protease activities. The in vivo function of Deg1 has been shown to be involved not only in PSII assembly but also in the degradation of PSII reaction center protein D1. Here we used the transgenic plants with reduced Deg1 to examine whether the lumen-localized proteins are also the substrates of Deg1 in vivo. Our results showed that the transgenic plants accumulated degradation products of the PsbO protein while the levels of full-length PsbO were not affected. The PsbO degradation products could be efficiently degraded by the recombinant Deg1. These results suggest that Deg1 is involved in the degradation of the PsbO degradation fragments, but not in the initial cleavage event itself.
Similar content being viewed by others
References
Gottesman S. Proteases and their targets in Escherichia coli. Annu Rev Genet, 1996, 30: 465–506
Pallen M, Wren B. The HtrA family of serine proteases. Mol Microbiol, 1997, 26: 209–221
Clausen T, Southan C, Ehrmann M. The HtrA family of proteases: Implications for protein composition and cell fate. Mol Cell, 2002, 10: 443–455
Lipinska B, Zylicz M, Georgopoulos C. The HtrA (DegP) protein, essential for Escherichia coli survival at high temperatures, is an endopeptidase. J Bacteriol, 1990, 172: 1791–1797
Spiess C, Beil A, Ehrmann M. A temperature-dependent switch from chaperone to protease in a widely conserved heat shock protein. Cell, 1999, 97: 339–347
Huesgen P F, Schuhmann H, Adamska I. The family of Deg proteases in cyanobacteria and chloroplasts of higher plants. Physiol Plant, 2005, 123: 413–420
Kieselbach T, Funk C. The family of Deg/HtrA proteases: From Escherichia coli to Arabidopsis. Physiol Plant, 2003, 119: 337–346
Adam Z, Rudella A, van Wijk K J. Recent advances in the study of Clp, FtsH and other proteases located in chloroplasts. Curr Opin Plant Biol, 2006, 9: 234–240
Sakamoto W. Protein degradation machineries in plastids. Annu Rev Plant Biol, 2006, 57: 599–621
Kato Y, Sakamoto W. Protein quality control in chloroplasts: A current model of D1 protein degradation in the photosystem II repair cycle. J Biochem, 2009, 146: 463–469
Kapri-Pardes E, Naveh L, Adam Z. The thylakoid lumen protease Deg1 is involved in the repair of photosystem II from photoinhibition in Arabidopsis. Plant Cell, 2007, 19: 1039–1047
Sun X W, Ouyang M, Guo J K, et al. The thylakoid protease Deg1 is involved in photosystem II assembly in Arabidopsis thaliana. Plant J, 2010, 62: 179–355
Hauβühl K, Andersson B, Adamska I. A chloroplast DegP2 protease performs the primary cleavage of the photodamaged D1 protein in plant photosystem II. EMBO J, 2001, 20: 713–722
Sun X W, Peng L W, Guo J K, et al. Formation of DEG5 and DEG8 complexes and their involvement in the degradation of photodamaged photosystem II reaction center D1 protein in Arabidopsis thaliana. Plant Cell, 2007, 19: 1347–1361
Sun X W, Fu T J, Chen N, et al. The stromal chloroplast Deg7 protease participates in the repair of PSII after photoinhibition in Arabidopsis. Plant Physiol, 2010, 152: 1263–1273
Chassin Y, Kapri-Pardes E, Sinvany G, et al. Expression and characterization of the thylakoid lumen protease DegP1 from Arabidopsis. Plant Physiol, 2002, 130: 857–864
Peng L W, Ma J F, Chi W, et al. Low PSII accumulation1 is involved in the efficient assembly of photosystem II in Arabidopsis thaliana. Plant Cell, 2006, 18: 955–969
Schuster G, Timberg R, Ohad I. Turnover of thylakoid photosystem II proteins during photoinhibition of Chlamydomonas reinhardtii. Eur J Biochem, 1988, 177: 403–410
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Li, J., Sun, X. & Zhang, L. Deg1 is involved in the degradation of the PsbO oxygen-evolving protein of photosystem II in Arabidopsis . Chin. Sci. Bull. 55, 3145–3148 (2010). https://doi.org/10.1007/s11434-010-4042-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-010-4042-2